Feb 01, 2026
Zymo Zyppy Plasmid Miniprep - CHEM 384
Forked from Zymo Plasmid Miniprep - Classic - CHEM 584
This protocol is a draft, published without a DOI.
- JC P1
- 1Brigham Young University

Protocol Citation: JC P 2026. Zymo Zyppy Plasmid Miniprep - CHEM 384. protocols.io https://protocols.io/view/zymo-zyppy-plasmid-miniprep-chem-384-hqdxb5s7p
Manuscript citation:
https://files.zymoresearch.com/protocols/_d4015_d4016_d4054_zr_plasmid_miniprep.pdf
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
This is based on the manufacturers protocol.
Created: January 29, 2026
Last Modified: February 01, 2026
Protocol Integer ID: 241815
Keywords: efficient isolation of plasmid dna, zr plasmid miniprep, high yield plasmid dna, zymo plasmid miniprep, plasmid dna, quality plasmid dna, dna ligation, restriction endonuclease digestion, dna, endotoxin, modified alkaline lysis protocol, alkaline lysis protocol, complete cell lysi, spin lln column, zymo zyppy plasmid miniprep, zyppy plasmid miniprep kit, unique fast spin column
Abstract
The Zyppy Plasmid Miniprep kit is designed for efficient isolation of plasmid DNA from E. coli cell lysates using a procedure that is simple, rapid, user-friendly, and reliable. It features a modified alkaline lysis protocol together with a unique Fast Spin column to yield high-quality plasmid DNA in minutes.
Guidelines
1. The following procedures are carried out at room temperature. All centrifugation steps should be performed between 11,000 - 16,000 x g.
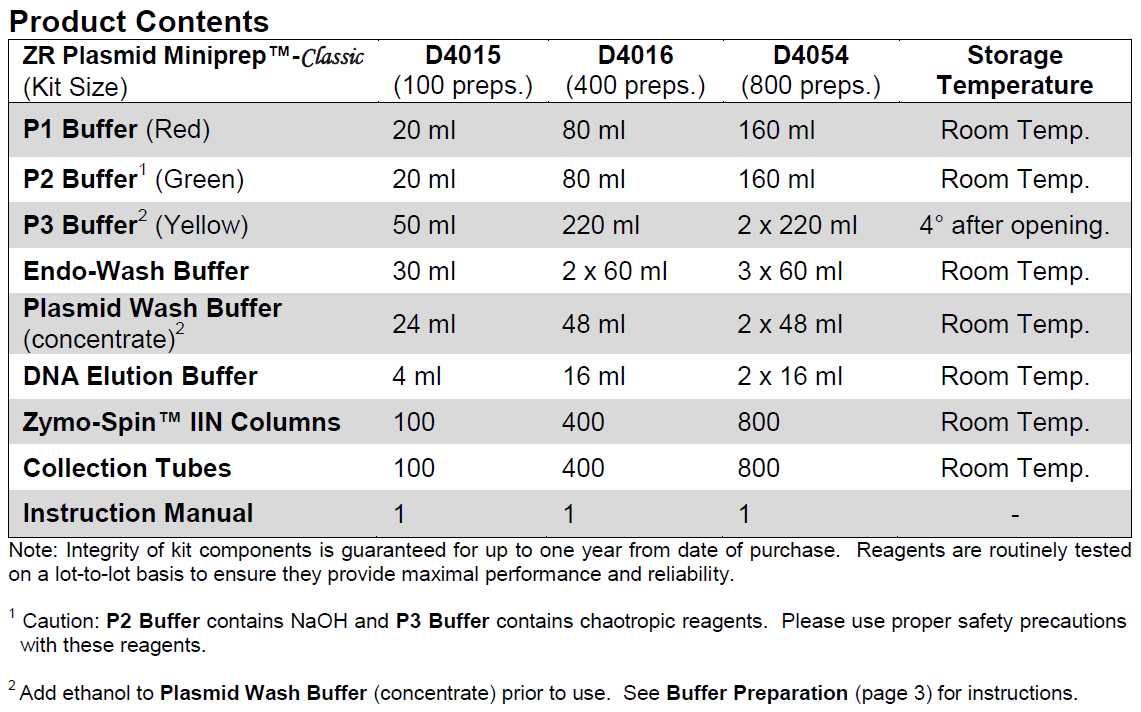
Materials
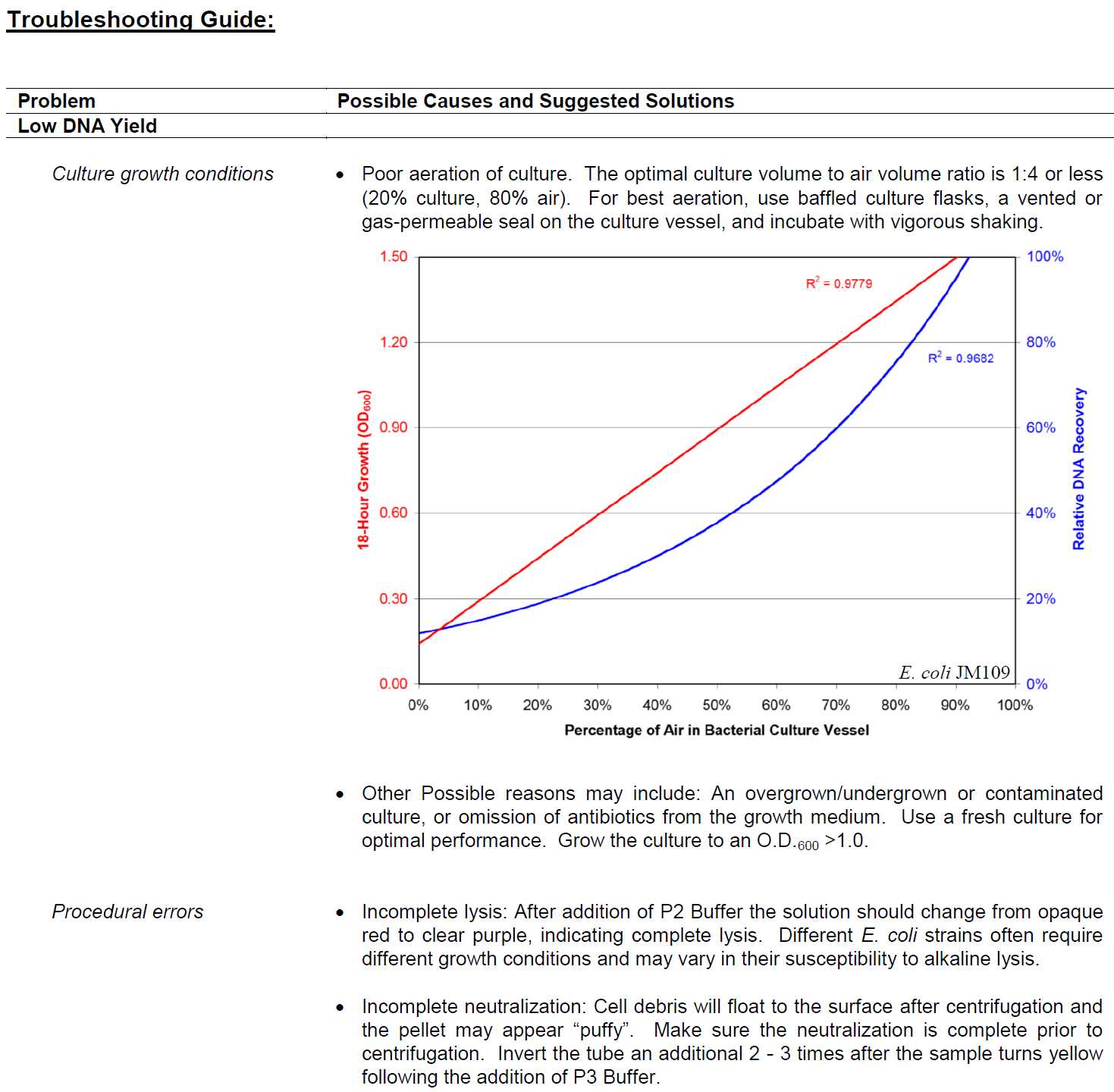
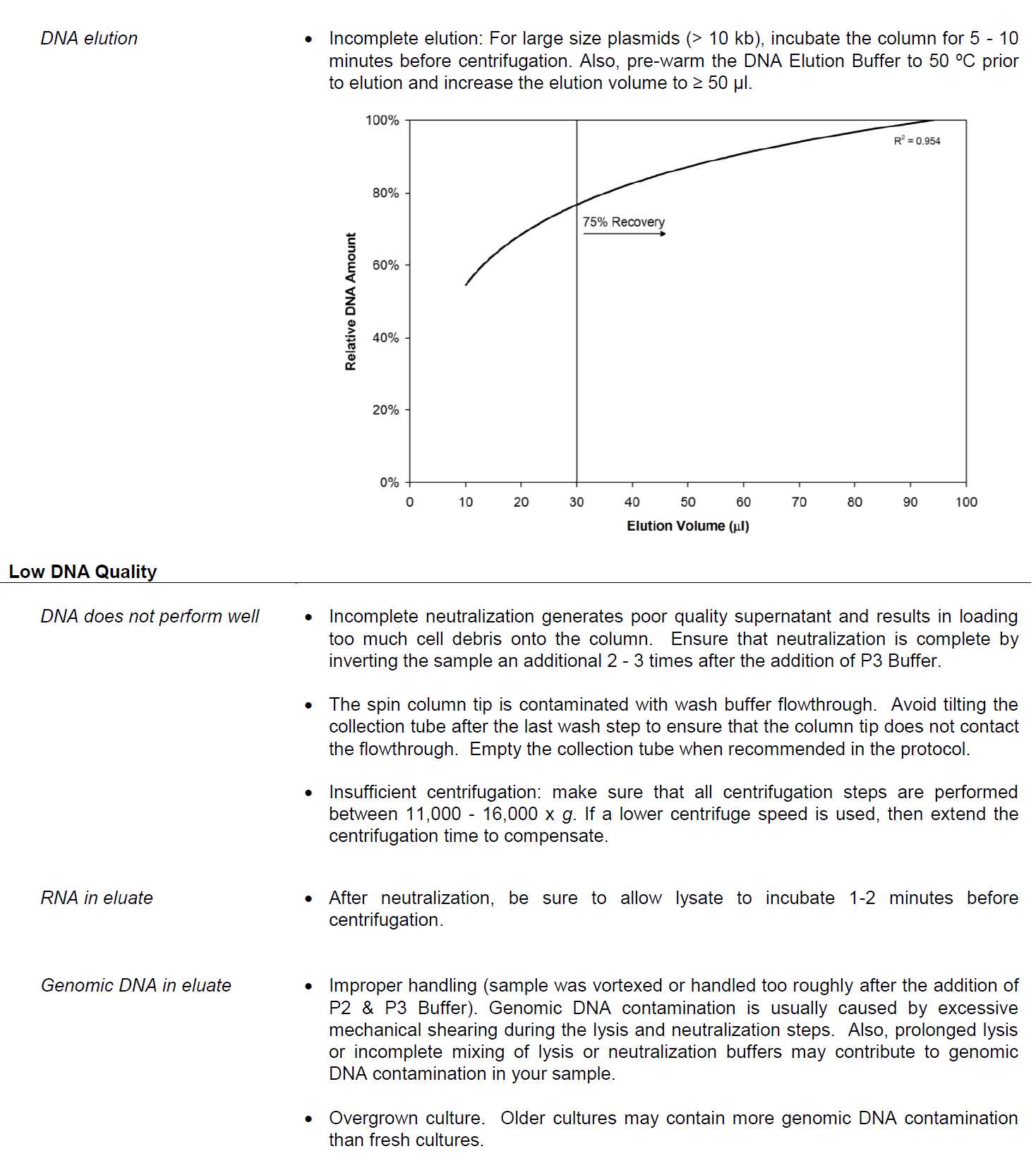
Troubleshooting
Zyppy MINIPREP
Using a UV-Vis spectrophotometer and a cuvette, measure the absorbance at 600 nm of your bacterial broth. Make sure to blank the spectrophotometer against clean fresh broth. Record this optical density (OD) measurement
For OD readings of less than 1, use 5 mL. For OD readings of 2 or more use 1mL. Centrifuge 1 mL - 5 mL of bacterial culture in a clear 1.5 ml tube at full speed for 15 - 20 seconds in a microcentrifuge. Discard supernatant.
Note
For OD readings of less than 1, use 5 mL. For OD readings of 2 or more use 1mL. Scale your volume depending on the OD of the broth. Depending on the volume of bacterial culture it may be necessary to repeat Step 1 several times. You can also centrifuge larger culture volumes (e.g., 5 mL) in a 15 mL centrifuge tube, then transfer to a microcentrifuge tube following step 2.
resuspend bacteria pellet in 600 uL of milliQ water and transfer to a 1.5 mL eppendorph tube
Add 100 µL of 7x lysis buffer (blue) to the tube with the resuspend pellet completely (i.e., by pipetting pellet up and down). Incubate for 1 minute with gentle mixing (inverting).
Add 350 µL of Neutralization Buffer (Yellow) and mix gently but thoroughly. Do not vortex. The sample will turn yellow when the neutralization is complete. Allow the lysate to incubate at room temperature for 1-2 minutes 00:02:00 .
Note
A green precipitate consisting of K·SDS and cell debris will form. A good way to mix is to shake the tube gently several times while it is inverted.
Centrifuge sample(s) at 16,000 x G for 2 minutes.
Place a Zymo-Spin™ IIN column in a Collection Tube and transfer the supernatant (~800uL) from Step 6 into the Zymo-Spin IIN column. When pipetting the supernatant, be careful not to disturb the yellow pellet to avoid transferring any cellular debris to the column.
If more than 800 uL of liquid is available repeat step 7 and 8 until all volume is passed through the column.
Centrifuge the Zymo-Spin IIN/Collection Tube assembly for 15 seconds.
Discard the flow-through in the Collection Tube, making sure the flow-through does not touch the bottom of the column. If bottom of the column is dirty clean gently with milliQ water. Return the Zymo-Spin™ IIN column to the Collection Tube.
Note
The capacity of the collection tube with the column inserted is 800 μl. Empty the collection tube whenever necessary to prevent contamination of the spin column with the flow-through.
Add 200 µL of Endo-Wash Buffer to the column and centrifuge for 30 seconds.
Add 400 µL of Zyppy Wash Buffer to the column. Centrifuge for 1 minute.
Transfer the column into a clean 1.5 ml microcentrifuge tube and then add 30 µL of Zyppy Elution Buffer to the column. Centrifuge for 30 seconds to elute the plasmid DNA.
Note
The DNA Elution Buffer contains 10 mM Tris·HCl, pH 8.5, 0.1 mM EDTA. If required, pure water can be used to elute the DNA. Add the DNA Elution Buffer directly to the center of the Zymo-Spin IIN column matrix to ensure optimal DNA elution.
Measure the concentration and purity of your purified DNA
TROUBLESHOOTING