Feb 11, 2026
Wholemount Immunofluorescence of Organoids (ver 3.0)
- Benjamin Sharpe1,
- Liliya Nazlamova1,2,
- David Johnston3,
- Tim Fenton1,4,
- Zoe Walters1,
- Tim Underwood1
- 1School of Cancer Sciences, Faculty of Medicine, University of Southampton;
- 2Early Cancer Institute, Department of Oncology, University of Cambridge;
- 3Biomedical Imaging Unit, Faculty of Medicine, University of Southampton;
- 4University Hospital Southampton NHS Foundation Trust

Protocol Citation: Benjamin Sharpe, Liliya Nazlamova, David Johnston, Tim Fenton, Zoe Walters, Tim Underwood 2026. Wholemount Immunofluorescence of Organoids (ver 3.0). protocols.io https://dx.doi.org/10.17504/protocols.io.yxmvm1zznv3p/v1
Manuscript citation:
This protocol is a modified version of the published Cell Reports Methods paper:
Sharpe, B.P. et al. (2024) ‘Patient-derived tumor organoid and fibroblast assembloid models for interrogation of the tumor microenvironment in esophageal adenocarcinoma’, Cell Reports Methods, 4(12), p. 100909. doi:10.1016/j.crmeth.2024.100909.
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: February 06, 2026
Last Modified: February 11, 2026
Protocol Integer ID: 242812
Keywords: Organoids, Wholemount immunofluorescence, Confocal microscopy, Clearing, 3D models, 3D cell culture, wholemount immunofluorescence of organoid, wholemount immunofluorescence, stained organoid, organoid, lightsheet microscope, cell culture, ki67 immuno, cleared sample, detection kit, imaging
Funders Acknowledgements:
Leverhulme Trust
Grant ID: RPG-2025-117
Royal College of Surgeons of England / Cancer Research UK
Grant ID: A23924
Abstract
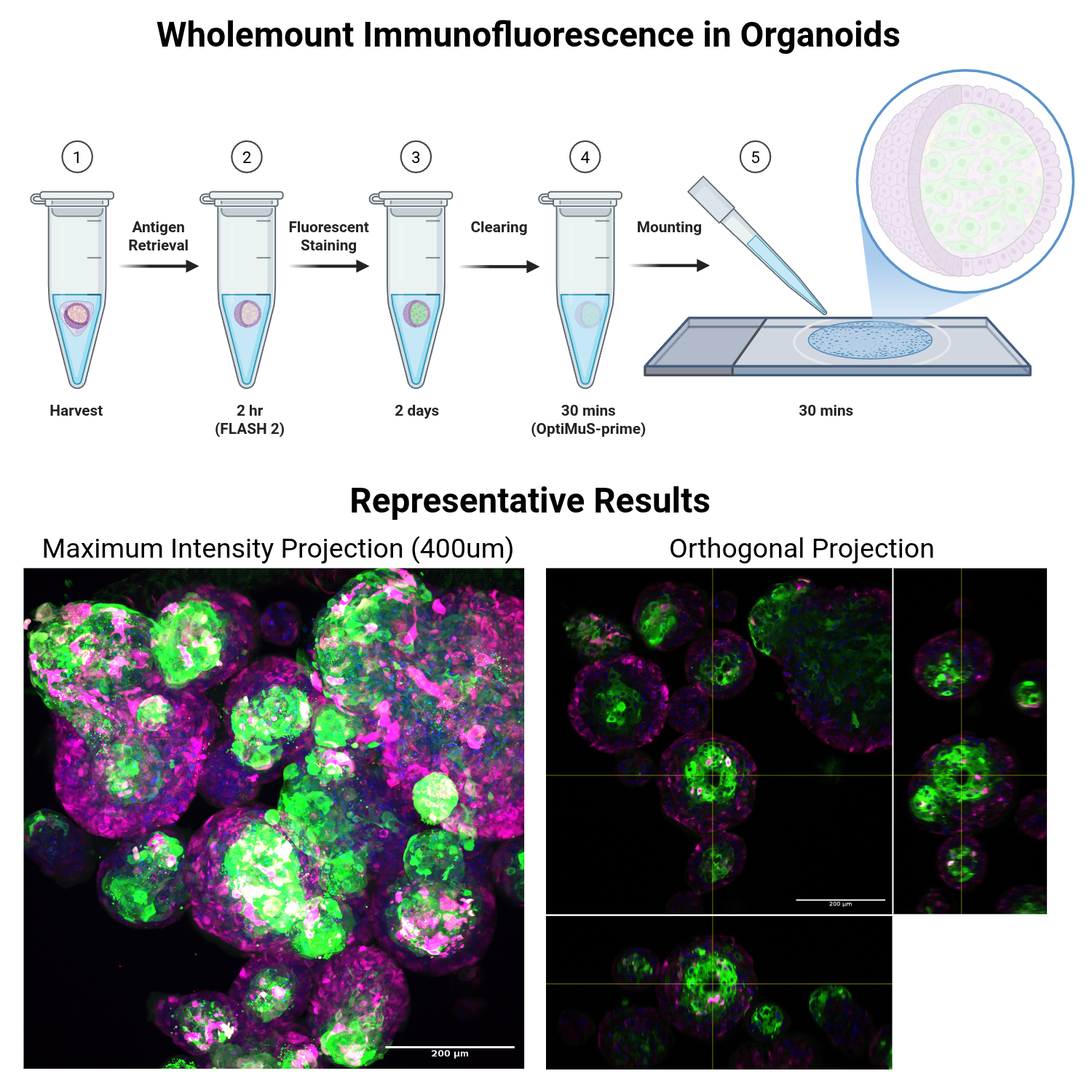
This protocol is for wholemount immunofluorescence of organoids, co-cultures or other 3-dimensional structures that can be grown in scaffolds. Stained organoids are then cleared and imaged with a laser-scanning confocal or lightsheet microscope depending on equipment availability and needs. Optionally, samples can also be cheaply counterstained for amines using the FLARE Protocol (Lee et al., Nature Protocols 2022) before immunostaining is performed. S-phase entry can also be visualised by EdU dosing during the cell culture, and later staining with click detection kits of your choice (the Click-iT EdU are incredibly bright and I prefer them to Ki67 immuno). Cleared samples can be washed after imaging and processed to paraffin if histological characterisation is required.
Image Attribution
Tonsil organoids generated by Dr Benjamin Sharpe (University of Southampton, UK). Figure schematics created with BioRender (https://BioRender.com).
Guidelines
When handling organoids, always pre-coat tips and tubes with 1% BSA solution (or use protein low-bind tubes) to prevent organoids sticking to plastic. Note that for wash steps, ensure that gravity sedimentation or centrifugation are used, and this must be be optimised for each organoid system used to avoid crushing or distorting organoids.
Materials
- A rocker, nutator or equivalent
- Usual equipment for preparing stock solutions, e.g. magnetic stirrer and pH meter
- Suitable mounting frames to hold organoids (e.g. 3D printed/homemade) or ibidi chamber slides (no fuss, but expensive)
- Clear nail varnish
- Pasteur pipettes, 3mL (if handling big cultures), or 1mL tips cut at the ends to avoid shearing organoids
- A suitable laser-scanning confocal microscope for imaging (we use a Leica SP8). You could use lightsheet microscope or spinning disk confocal for quicker data generation (with caveats).
- 4% Paraformaldehyde (PFA), either premade frozen aliquots or snap-cap image-iT style PFA.
- Borate buffer
200mM (12.36g/L) boric acid in ddH2O, pH to 7.0.
- FLASH reagent 2 (make 10mL) - Messal et al., Nature Protocols 2021 (https://pubmed.ncbi.nlm.nih.gov/33247285/)
200mM borate buffer, pH 7.0
250g/L urea (2.5g per 10mL)
80g/L Zwittergent 3-10 (0.8g per 10mL)
Ensure pH = 7.0
- PBS
- Organoid Washing Buffer (OWB)
PBS
0.1% (v/v) Triton X-100
0.2% (w/v) bovine serum albumin
Store at 4C
- TE Buffer (pH 7.5) - 500mL
100 mM Tris (Thermo #J65594.A1, CAS 77-86-1, Mw = 121.14 g/mol) - 6.057g Tris
0.34 mM EDTA (Sigma #E5134-500G, CAS 6381-92-6, Mw = 372.24 g/mol) - 63.3mg EDTA
pH to 7.5
- OPTIMUS-Prime refractive-index matching (mounting) buffer as per Kim et al, Exp Mol Med 2025 (https://www.nature.com/articles/s12276-025-01550-w)
5mL TE buffer (pH 7.5)
75% (w/v) Iohexol (Sigma #D2158, CAS 66108-95-0) - 7.5g
10% (w/v) D-sorbitol (1g)
4M Urea - 2.402g Urea
Heat to 60C with occasional inversion to mix (be patient, there is a lot of iohexol in here to dissolve)
Once dissolved, make up to 10mL volume with TE buffer.
Store at 4C.
- Primary and Secondary Antibodies - choose fluorophores carefully to reduce spectral overlap
- Optional: Invitrogen Click-iT EdU Cell Proliferation Kit for Imaging (I use Alexafluor 555)
- Optional: MES Buffer (pH 6.0) - if using amine counterstain for morphology
25mL of ddH2O
586mg MES (2-(N-Morpholino)ethanesulfonic acid hydrate)
263mg NaCl
pH to 6 using 1M NaOH
Store at RT (~2 weeks or until precipitate appears, then discard and make fresh)
- Optional: AZDye-647-NHS-ester (2BScientific #FP-1121-1MG) - if using amine counterstain
Make a 5mg/mL stock solution in DMSO
Make small aliquots (1-5uL) and store at -20C
Troubleshooting
Safety warnings
Paraformaldehyde is harmful if inhaled and a suspected carcinogen. Always handle PFA in a fume hood and follow local waste disposal guidelines.
H317 - May cause an allergic skin reaction
H318 - Causes serious eye damage
H351 - Suspected of causing cancer
Ethics statement
Appropriate ethics should be obtained prior to the use of animal or human tissue for organoid derivation and modelling.
Day 1: Harvest and Fix Organoids
2h 5m
Collect specimens (organoids, 3D cultures etc) as you normally would, taking best efforts to keep them intact. This varies depending on application and the type of culture.
**CRITICAL If you are handling organoids with tips, following the recommendation in Dekkers’ paper and coat them in sterile 1% BSA in PBS by pipetting 1mL a few times before you touch any organoids. Use protein low-bind (or BSA-precoated) tubes to collect them. This will stop them sticking, which WILL happen once they’re released from matrigel!
10m
For organoids that must be taken out of BME/Matrigel scaffolds, recommended harvest is on ice-cold Corning Cell Recovery solution, rocking, for up to 01:00:00 On ice .
1h
Collect organoids at the bottom of an eppendorf tube by gravity sedimentation or gentle centrifugation. Aspirate supernatant and fix in >1ml or 10 volumes of cold 4% PFA for 00:45:00 minutes at On ice (on ice).
Safety information
Warning: PFA is harmful if inhaled. Ensure PFA is added and removed in a fume hood.
45m
Centrifuge/sediment organoids. Wash with OWB (2x)00:05:00
Note
If staining is to be conducted on another day, the organoids can now be kept at 4 °C for short term storage, and the protocol can be resumed with part, or all, of the organoids at a later date.
10m
Antigen Retrieval
2h 20m
Make up FLASH reagent 2 freshly (I make up 10mL) and keep at 37 °C in a water bath until use. Add 2.5g urea and 0.8g Zwittergent-3-10 into 10mL 200 millimolar (mM) sodium borate buffer, 7.0
Note
If storing excess FLASH 2 buffer for later use, store remainder in the fridge and warm up to 37 °C again to re-dissolve.
10m
Aspirate excess wash buffer and add 1mL FLASH 2 reagent per tube. Pack tubes into a rocker or nutator with gentle agitation and incubate at 37 °C for 02:00:00 .
2h
Wash twice with PBS to remove excess FLASH 2 reagent (2x) 00:05:00
10m
Optional Counterstaining using NHS-Ester Dyes
2h 10m
Optional: At this point, organoids can be stained with AZDye-647-NHS-ester to detect cellular amines (mostly proteins). This visualises general morphological characteristics more distinctly than phalloidin. Add 0.5uL of dye stock per 1mL of MES buffer, pH 6.0. Incubate with rocking at room temperature for 02:00:00 , then wash twice with MES buffer 00:05:00 before proceeding.
2h 10m
Primary Antibody Staining
15h 45m
Wash with OWB, (3x) 00:10:00 with constant rotation.
30m
Prepare primary antibody solutions by diluting stock antibodies in OWB. You can mix e.g. a mouse and a rabbit primary antibody into the cocktail for multiplexing. You can get away with 200uL per tube as long as samples are mixed well, but more is better (recommended volume for large organoids is 500uL). Dilutions that work for standard IHC on paraffin normally work well here too in my experience, but further titration might be needed to reduce background staining depending on the antibody.
10m
Aspirate wash buffer carefully. A small amount of residual liquid is fine. Add antibody dilutions.
5m
Incubate on a rotator at room temperature Overnight for organoids or two days at 4 °C for large co-cultures. In our experience, room temperature incubations improve uniformity of labelling.
15h
Day 2-3: Secondary Antibody Staining and Counterstaining
16h 20m
Wash antibodies out with one brief rinse of OWB, then 3x10 min washes with constant rotation.
35m
Prepare secondary antibody solutions by diluting stock antibodies in OWB. You can mix e.g. an anti-mouse and an anti-rabbit secondary antibody into the cocktail for multiplexing.
Prepare 200-500uL per sample. Normally 1:500-1:1000 are reasonable dilutions for Molecular Probes alexafluor secondaries, but further titration might be needed depending on the antibody and the presence of background signal. At this point you can also add DAPI (350/405nm excitation) at 1:500 or methyl green (633nm excitation) at 1:500 for nuclear counterstaining.
Note
Ensure that the fluorophores are spectrally distinct to allow appropriate excitation and detection later with minimal bleedthrough, and that your fluorophores can be excited by the lasers on the microscope you choose to use (I recommend AlexaFluors 488, 568 and 633, the further apart the better).
10m
Add secondary antibodies to specimen and incubate at room temperature with constant rotation Overnight
15h
Wash antibodies out with one brief rinse of OWB, then (3x)00:10:00 washes with constant rotation.
35m
EdU Detection
1h 50m
Optional: Proceed with EdU detection if a Click-iT EdU detection kit (or similar copper click reaction) is being used and cells have been dosed before fixation as below.
Note
Copper salts used in conventional click detection kits will interfere with some other reagents used in immunolabelling, such as phalloidin binding (phalloidin hates copper). Always do this reaction after other detection steps and optimise copper sulfate concentrations carefully if phalloidin is a necessity.
1h
Optional: Prepare click detection reaction according to manufacturer's directions, allowing 200-500uL of click reaction per tube. Incubate with the sample for 30 minutes at room temperature.
30m
Wash 2x with OWB 00:10:00 and proceed to clearing.
20m
Clearing and Mounting
55m
Remove as much OWB as possible, and add 200uL OPTIMUS-prime clearing buffer. Apply constant rotation for at least 30 minutes at room temperature, verifying that organoids are cleared with an epifluorescence microscope prior to imaging on a confocal microscope. Do not use excessive amounts of clearing buffer as organoids will be difficult to see at this point.
Note
Pause point: samples can be kept at 4C for short term storage prior to imaging.
35m
Mount samples using your preferred method. Pipetting up and down with a cut yellow tip will allow you to clearly see the organoids (they will be translucent but just about visible in the clearing solution when mixed, and usually at the bottom of the tube). I 3D print a ~0.4mm height square mounting frame in PLA, which I stick to glass slides using clear nail varnish. I then add all mounted organoids in, add a 22mm square coverslip, and seal the edges with more nail varnish. I have also had success with stacked adhesive reinforcement rings (as you would find in a folder), 50uL sample volume, and a circular 16mm coverslip, sealed with nail varnish. Ibidi chamber slides could also be used. Each lab will have different needs depending on their sample: consult with an imaging specialist if unsure what is suitable for the sample and microscope in question. Samples are now ready for imaging.
20m
Image on a point-scanning confocal, spinning-disk confocal or light sheet microscope as appropriate. I use a Leica SP8 laser-scanning confocal microscope with a 20x long working distance glycerol immersion objective. Long working distance objectives are important: ideally immersion objectives with immersion liquid that is close to the RI of the mounting media (OPTIMUS-Prime RI=1.47), but 63x can be used for high-resolution imaging with a lower volume/lower working distance.
Optional: Paraffin Processing
1d 1h
Following imaging, if processing for FFPE is desired, clearing buffer can be washed out with three changes of PBS, 10 minutes each.
30m
Embed samples in molten 2% (w/v) agarose in PBS (cooled to <60C), let cool, and cut to size where required. Store agarose blocks in a 5mL bijoux filled with 70% ethanol.
30m
Process to FFPE in standard histology casettes, using a tissue processor as standard.
1d
Protocol references
This protocol is a modified version of the published Cell Reports Methods paper:
Sharpe, B.P. et al. (2024) ‘Patient-derived tumor organoid and fibroblast assembloid models for interrogation of the tumor microenvironment in esophageal adenocarcinoma’, Cell Reports Methods, 4(12), p. 100909. doi:10.1016/j.crmeth.2024.100909.
This protocol includes recipes and reagents which have been published in other articles, as indicated below.
Organoid wash buffer (OWB):
van Ineveld, R.L. et al. (2020) ‘Single-cell resolution three-dimensional imaging of intact organoids’, Journal of Visualized Experiments, (160). doi:10.3791/60709.
Amine labelling using NHS-Ester chemistry:
Lee, M.Y. et al. (2022) ‘Fluorescent labeling of abundant reactive entities (flare) for cleared-tissue and super-resolution microscopy’, Nature Protocols, 17(3), pp. 819–846. doi:10.1038/s41596-021-00667-2.
Antigen retrieval using FLASH reagent 2:
Messal, H.A. et al. (2020) ‘Antigen retrieval and clearing for whole-organ immunofluorescence by flash’, Nature Protocols, 16(1), pp. 239–262. doi:10.1038/s41596-020-00414-z.
Refractive index-matching of stained organoids using OptiMuS-Prime:
Kim, K. et al. (2025) ‘A novel protein-preserving passive tissue clearing approach using sodium cholate and urea for whole-organ imaging’, Experimental & Molecular Medicine, 57(10), pp. 2292–2304. doi:10.1038/s12276-025-01550-w.
Acknowledgements
We would like to thank the Southampton Biomedical Imaging Unit for assistance with protocol development and microscopy, and the authors of published recipes included in this method (as indicated in references).
