Jul 07, 2025
Wedge sampling of pars petrosa (os temporale) for ancient DNA extraction
- Raphaela Stahl1,
- Lena Semerau1,
- Eleftheria Orfanou1,
- Marie Himmel2,
- Franziska Aron3,
- Wolfgang Haak1
- 1Department of Archaeogenetics, Max Planck Institute for Evolutionary Anthropology;
- 2Department of Archaeogenetics, Max Planck Institute for the Science of Human History;
- 3Friedrich-Schiller Universität Jena

Protocol Citation: Raphaela Stahl, Lena Semerau, Eleftheria Orfanou, Marie Himmel, Franziska Aron, Wolfgang Haak 2025. Wedge sampling of pars petrosa (os temporale) for ancient DNA extraction. protocols.io https://dx.doi.org/10.17504/protocols.io.261gen8wog47/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: June 08, 2022
Last Modified: July 07, 2025
Protocol Integer ID: 64178
Keywords: ancient DNA, petrous pyramid, sampling, non-invasive method, archaeogenetics, archaeology, wedge, pars petrosa, bone powder from the pars petrosa, wedge sampling of pars petrosa, ancient dna extraction, ancient dna extraction this protocol, dna extraction, disarticulated ossis temporali, bone powder, wedge into the bone, ossis temporali, pars petrosa, bone, high amounts of adna, dna, os temporale, dense parts around the cochlea, wedge sampling
Abstract
This protocol describes how to obtain bone powder from the pars petrosa of disarticulated ossis temporalis, specifically from the dense parts around the cochlea, in a more precise way by cutting a wedge into the bone before drilling.
The pars petrosa has been shown to consistently yield high amounts of aDNA (Pinhasi et al. 2015 PLoS One, doi: 10.1371/journal.pone.0129102).
Guidelines
Working in an Ancient DNA Laboratory
-All steps of the protocol should take place in a clean room facility specifically designed for ancient DNA.
-The researcher performing lab work should wear correspondingly suitable lab-wear, such as:
- full-body suit with hood (e.g. Tyvek)
- hairnet
- face mask
- three pairs of clean gloves
- clean shoes
- protective glasses
- Sample processing should be carried out in separated work benches with integrated UV irradiation (e.g. Dead Air PCR work bench)
- Surfaces and equipment should be regularly decontaminated with e.g. bleach solution or Thermofisher's DNA AWAY (or similar) and irradiated with UV.
Please see the following for more detailed guidance:
Llamas, B. et al., 2017. From the field to the laboratory: Controlling DNA contamination in human ancient DNA research in the high-throughput sequencing era. STAR: Science & Technology of Archaeological Research, 3(1), pp.1–14. Available at: https://doi.org/10.1080/20548923.2016.1258824.
Materials
Lab materials
Safe-Lock Tubes 2 ml Biopur (preferably packed individually)EppendorfCatalog #0030121597
DNA AWAY® 4000 mlCarl RothCatalog #X996.2
Weighing Paper MN 226 block of 100 sheets 9 x 115 cmMacherey-NagalCatalog #186002
Precision forceps 18/10 steel extra sharp bent points without ridges L=105mmVWR International (Avantor)Catalog #232-0008
Weighing pans ROTILABO® blue antistatic 330 ml 140 mm 140 mmCarl RothCatalog # 2159.2
Lab equipment
PCR work bench (e.g. AirClean Dead Air PCR Werkbank, 48´´)
UV irradiation box or cross linker (e.g. Vilber Lourmat Bio-Link BLX-254)
Air extraction/vacuuming unit
Drill Lab Handpiece (e.g. K-POWERgrip™ Installation Lab Handpieces from Kavo Dental Excellence; SKU: 10022916)
Rounded dental drill bit (e.g. NT1 from Kahla; SKU: H1-016 HP)
Round diamond saw blades (e.g. Diamond Discs Superflex, 22 mm from Kahla, SKU 806 104 358 514 220)
Tooth plier (e.g. mf-dental.de, Zahnzange, OK Prämolaren, Anatom. Griff, A127)
Balance (e.g. Ohaus Adventurer balance AX1502)
Anti-static instrument (e.g. Zerostat 3 from Zerostat; SKU: SAFAZ108812)
Aluminium foil (lab grade or sterile preferred)
Paper towels
Polyethylene clear plastic bags
Marking or masking tape
Camera
General Reagents
Solution of household bleach (2-6% NaClO, then diluted to a working solution concentration of 0.2-0.5% NaClO)
Thermofisher DNA AWAY
UV-irradiated and deionised tap water
Troubleshooting
Safety warnings
Reagents
Household bleach solution (2-6%) diluted to a working concentration of 0.2-0.5 % NaClO in total.
- H290 May be corrosive to metals.
- H314 Causes severe skin burns and eye damage.
- H411 Toxic to aquatic life with long lasting effects.
- EUH206 Warning! Do not use together with other products. May release dangerous gases (chlorine).
Remove from surface after recommended incubation time with water-soaked tissue.


DNA AWAY
- H290 May be corrosive to metals.
- H314 Causes severe skin burns and eye damage.

Note: Both bleach solutions and DNA AWAY are used for decontamination. DNA AWAY is less corrosive than bleach and should be prefered for decontamination of sensitive equipments such as surfaces of electric devices.
Equipment
UV radiation
- UV radiation can damage eyes and can be carcinogenic in contact with skin. Do not look directly at unshielded UV radiation. Do not expose unprotected skin to UV radiation.
- UV emitters generate ozone during operation. Use only in ventilated rooms.


Usage of sharp tools
Always hold the sample with pliers while cutting and drilling to avoid injuries

Before start
Planning
The sampling procedure of any skeletal element for DNA extraction should be performed ideally in a dedicated sampling room. This should not happen in the buffer preparation room.
Sampling of each pars petrosa takes around 20-40 minutes, but this can vary depending on the nature of the sample.
Equipment
Make sure all necessary equipment is available (see Materials).
Documentation
This protocol is destructive, therefore for all samples consultation and permission with curators should be performed prior running this protocol.
All anatomical and morphological features of the pars petrosa should be documented (e.g. pre-sampling photos, CT-scans) before sampling.
If petrous bones from both sides are available, sample only from one side, in order not to cause alterations to the external morphology of both.
Workstation preparation
10m
Safety information
Everything that comes in contact with the sample needs to be decontaminated in order to avoid cross-contamination between samples. Change gloves regularly, especially between different samples.
Place a sheet of aluminium foil under the hood.
Place weighing paper, drill bits, and a pair of tooth pliers or forceps on a second sheet of aluminium foil on an easily accessible clean surface outside of the hood. Alternatively, you may place them within the hood but be sure to cover them with aluminum foil or a paper towel to protect them from dust produced during sampling.
You will also need:
- 2 ml Safe-Lock Biopure tubes
- Weighing trays/pans
- Clean and labelled sample bags according to the amount of samples
- Small pieces of aluminium foil for wrapping the bone (optional)
- Small tape strips to fix the aluminium foil while covering the bone (optional)
- Paper towels
- Diluted commercial bleach solution (1:10)
- UV-irradiated filtered/deionised water

Figure 1. Possible Set up of the workstation
10m
Preparation of sample
30m
Place a sheet of aluminium foil in the UV chamber.
Place the pars petrosa on the aluminium foil and place the labelled sample bag next to it
(to avoid mixing up samples in case you work on more than one).
UV- irradiate the bone for 00:15:00 on each side.
Safety information
If possible, avoid touching the bone directly. Use precision forceps or something similar to handle each bone individually.
Note
Tip: It is possible to UV irradiate multiple bones at once in the same UV chamber. Ensure each bone is on a separate bit of foil, and keep the corresponding labelled sample bag next to it to prevent sample mix up.
30m
Take a new 2 ml tube and label it with the sample ID on top of the lid and on the side of the tube. Tare the precision scale with the tube placed in the center.
Label a new sample bag with the sample ID to be used after sampling.
Safety information
To avoid contamination, tubes should only be opened inside the hood and remain closed except when the bone powder is added.
Take a weighing tray with a new pair of gloves.
Prepare a sheet of weighing paper by creasing it mid-way and place it under the area where you want to sample.
Note
Bone powder can be statically charged and difficult to transfer to tubes. By slightly folding the weighing paper, it makes it easier to move the bone powder from the weighing paper into the tube.
Sampling procedure
Wedge sampling of pars petrosa
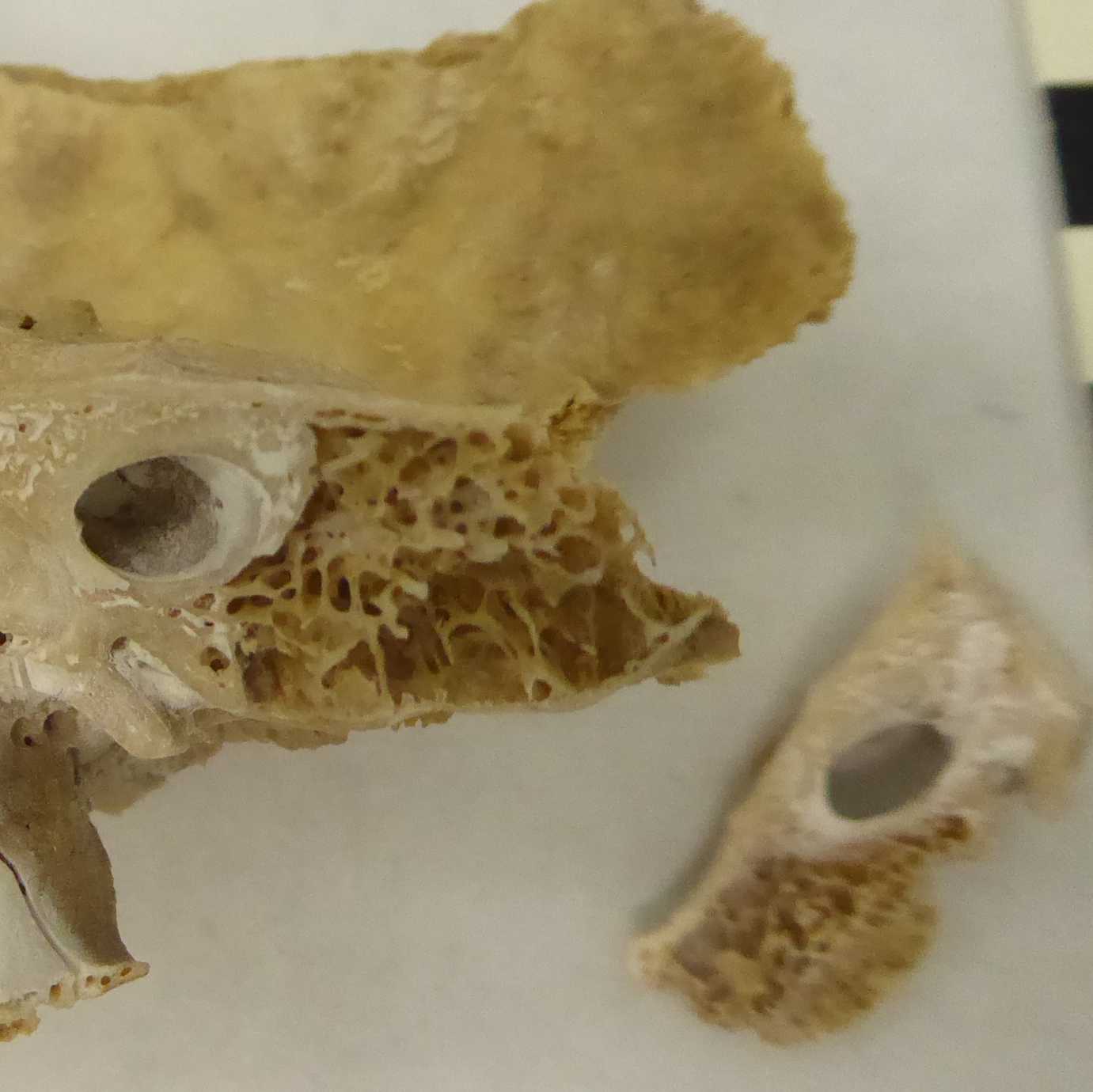
Familiarize yourself with the anatomy of the petrous bone (e.g., Pinhasi et al. 2015). Locate the dense parts around the cochlea (b & c) as shown below in Figure 2.
Ideally you will target area c (orange) shown in Figure 2, which is located roughly halfway between the jugular fossa (bottom) and the petrous crest (top).
Citation
LINK
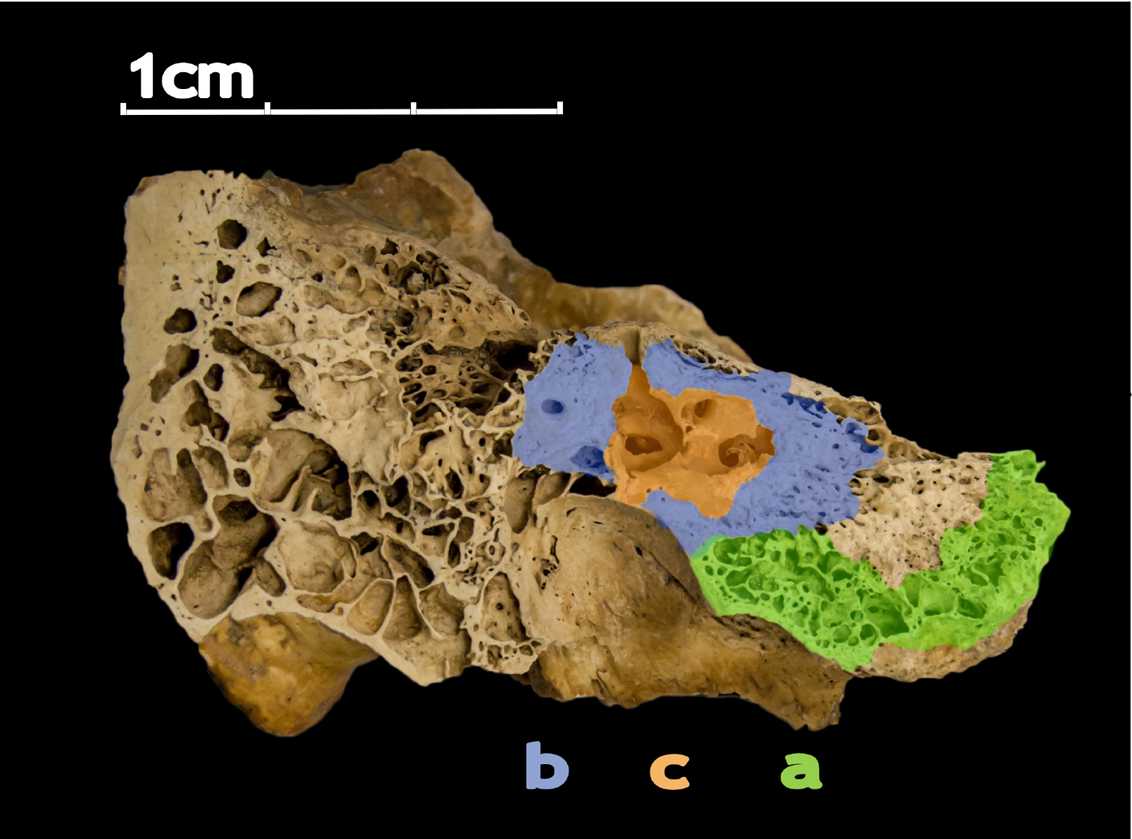
Figure 2. Dense parts around cochlea (b & c). Part c provides higher endogenous aDNA yields than part b and a.
(Pinhasi et al. 2015).
To make this desired area more accessible for drilling, it is easier to turn the pars petrosa in a different angle (see Figure 2a). Locating the densest part of the bone around the cochlea is simplified by using the wedge cutting method for sampling. Hence the petrous bone is cut in half, while holding the bone fraction with a tooth plier at the temporal bone.
Figure 2a. Outside perspective of pars petrosa with temporal bone.
Therefore a round diamond saw blade is used to cut starting from the apex partis petrosae, following the margo superior partis petrosae and will stop doing so shortly after the internal acoustic opening (meatus acusticus internus). (Figure 2a)
See below a picture after cutting out this wedge. (Figure 3)
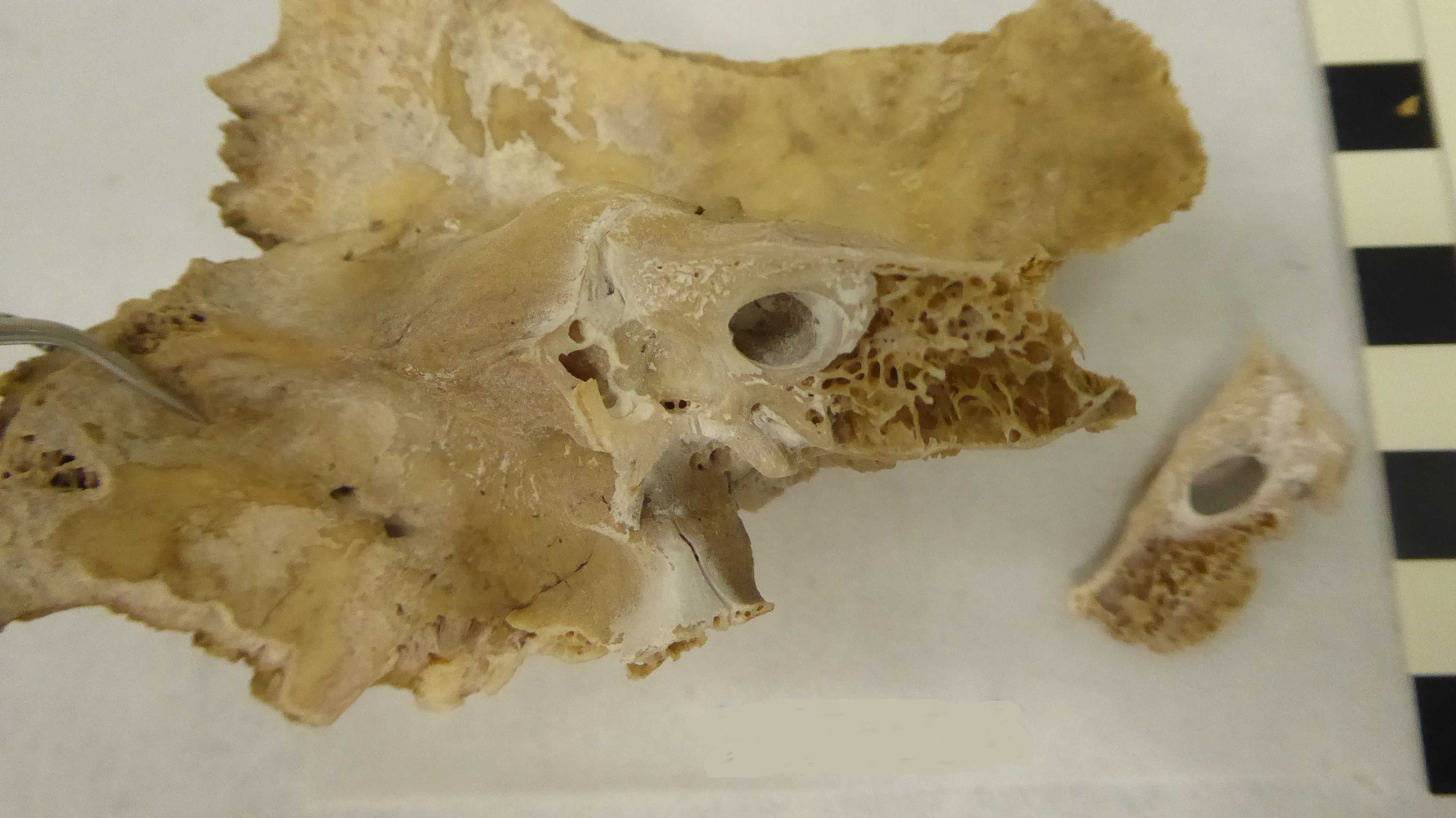
Figure 3. Pars petrosa with attached temporal bone after wedge cut.
To prevent residual dust/dirt from the outer surface and crevices to contaminate your freshly drilled bone powder, you can wrap the sample with aluminium foil and/or tape. Leave the part that you want to drill unwrapped. (Figure 4).

Figure 4. Wrapping the pars petrosa with aluminium foil.
Drill at low speed (and high torque) within the cochlear region in parallel to the auditory canal.
Collect clean bone powder on the weighing paper. Of note, it is possible that you will drill into internal canals and porosities that can be filled with soil, despite your bone being cut open for better visibility. If so, follow our Pro-Tip below.
Empty the drilling hole by carefully tipping it out onto the collection paper.
Note
Pro-Tip: If residual dirt/soil is mixed in with your collected clean bone powder, you can physically separate them by carefully tipping the edges of the weighing paper repeatedly. In doing so, the different weights and particle sizes will easily separate. Discard or set aside the dirt.
Safety information
Be careful when unwrapping the bone, the vibration of the drilling will loosen residual dirt/soil and bone powder, which will accumulate in the wrapping.
Weighing of bone powder
Transfer the clean powder bone into a labelled 2 ml Safe-Lock Biopure tube. The folded weighing paper will help guiding the powder into the tube.
Close the tube, wipe it with a damp paper towel and weigh the bone powder. Make sure that this exact tube was used before to tare the scale.
Safety information
Bone powder can become statically charged. In such case, consider using an anti-static gun and apply to the weighing paper and tube.
Note
Ideally you wish to collect around 30 mg to 50 mg of powder for extraction (if using our protocol for ancient DNA Extraction). If you have more than 50 mg then store it as back-up material in a separate tube.
Note down the weight in mg on the side of the tube. Please refrain from labeling the lid with this information, it'll be too hard to read afterwards. Of course labeling the lid with the sample ID is recommended.
Store the bone powder at-20 °C until further processing.
Take photos after sampling for documentation.
Put bone back into a new, UV irradiated, labelled bag.
Decontamination
Safety information
Everything that came in contact with the sample needs to be cleaned/decontaminated in order to avoid cross-contamination between samples.
Carefully clean the workspace and the equipment before sampling the next sample:
- Throw away disposable material such as aluminium foil and weighing tray/pan.
- Clean surfaces and tools such as pliers with bleach solution
- Use less aggressive DNA decontamination reagents (e.g. DNA away) for sensitive material such as electronic devices (e.g. handpiece of the drill, precision balance).
- Wipe off bleach with water-wet paper towels afterwards to prevent corrosion; air dry.
Drill bits and saw blades can be reused after careful cleaning:
- incubate them in bleach solution (1:10 dilution) for at least 2 minutes.
You can use a UV irradiated and bleached toothbrush for brushing the saw blade.
- Clean with UV irradiated water to remove all bleach and let it dry.
Note
Check conditions of drill bits after ~5 uses and replace by new ones if necessary.
Citations
Step 5.1
Ron Pinhasi, Daniel Fernandes, Kendra Sirak, Mario Novak, Sarah Connell, Songül Alpaslan-Roodenberg, Fokke Gerritsen, Vyacheslav Moiseyev, Andrey Gromov, Pál Raczky, Alexandra Anders, Michael Pietrusewsky, Gary Rollefson, Marija Jovanovic, Hiep Trinhhoang, Guy Bar-Oz, Marc Oxenham, Hirofumi Matsumura, Michael Hofreiter. Optimal Ancient DNA Yields from the Inner Ear Part of the Human Petrous Bone
https://doi.org/10.1371/journal.pone.0129102