Aug 28, 2025
Water and sediment sampling in the sea to discover marine bacteria
- Karla Sofia Millan Lopez1,
- Elva Escobar1,
- Liliana Pardo-López1
- 1UNAM

Protocol Citation: Karla Sofia Millan Lopez, Elva Escobar, Liliana Pardo-López 2025. Water and sediment sampling in the sea to discover marine bacteria. protocols.io https://dx.doi.org/10.17504/protocols.io.x54v9rw9zv3e/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: January 17, 2025
Last Modified: August 28, 2025
Protocol Integer ID: 118646
Keywords: Sediment cores, Sea water, Ship sampling, marine bacteria this protocol, marine bacteria, sediment sampling in the sea, sediment sampling, sediment sample, sediment samples on board, oceanographic vessel at sea, oceanographic vessel, sea, obtaining water, sample
Funders Acknowledgements:
CONACYT PhD Scholarship
Grant ID: 957581
LANCO-CONAHCyT
Grant ID: APOYOSLNC-2023-80
DGAPA-PAPIIT
Grant ID: IG200223
Abstract
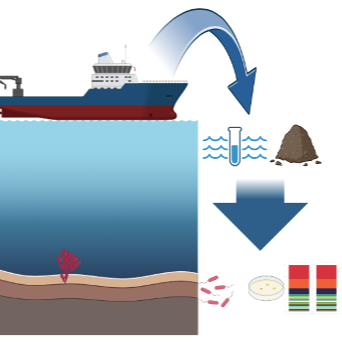
This protocol aims to show the strategy for obtaining water and sediment samples on board an oceanographic vessel at sea. The samples obtained can be used to identify marine bacteria associated with water and sediment samples.
Image Attribution
Marine Biotechnology Laboratory - IBT-UNAM
Guidelines
Samples must be processed immediately after collection.
Store samples cold after sampling.
Materials
Carousel equipped with Niskin bottles
Multinucleator
Spatulas
Glass containers
Container with liquid nitrogen
Protocol materials
Parafilm
Durapore™ PVDF Membrane Filters: 0.22μ Pore Size, 0.22 μ Pore Size; Plain; White; Dia.: 25mmThermo FisherCatalog #GVWP02500
Falcon Tube (50 mL)Fischer Scientific
Troubleshooting
Health and safety
Note
Make sure you have the necessary equipment to work on deck (overalls, suede gloves, helmet, safety booths, googles or sunglasses). Maintaining adequate hydration is crucial, as deck operations are quite exhausting.
Arriving at the sampling point
When arriving at the sampling station equipments are deployed following the cruise plan. Each station is referred to by is geographical position and depth.
Note
The sampling operations were carried out aboard UNAM's Research Vessel Justo Sierra.
Upon reaching the designated sampling station, a SBE 9plus-Seabird Conductivity, Temperature, and Depth (CTD) profiler was deployed until reaching an approximate height of 50 meters above the seabed.
To record launch, bottom, and ascent information in an electronic and physical log, as well as to include any additional annotations.
The equipment is recovered on deck and the next equipment in the sequence is prepared to be deployed.
The PNF-300 fluorometer (Wet Labs) is deployed, and the graph is generated from the fluorescence profile of the water column. This is done to identify the maximum peak.
Be sure to remove the caps from the fluorometer sensors.
The fluorometer is recovered and the plugs are placed back on the sensors.
Seafloor is determined using a Kongsberg EM302 Multibeam Echosounder system.
Sampling in the marine environment: Sea water
The identification of specific zones of interest is to be based on depth measurements following site characterization
Note
The extent of this process is subject to considerable variation, due to the depth of the site. For instance, the process may take up to 2 hours at depths of 3,800 meters. The equipment is deployed and recovered at a rate of 1m/sec, however stabilization of the equipment for a few seconds is required before the acquisition of water samples
Water sampling is carried out using a SBE 32 – Seabird Carousel equipped with the capacity to activate 12 10-liter Niskin PVC bottles. The carousel is descended with the bottles open and at each of the previously defined sampling depths the bottles are closed remotely.
The first bottle to close is the deepest and is labeled bottom. That bottle is marked as background.
During the carousel's ascent through the water column, the bottles are sealed at the required depth, for example 1500 meters, 1000 meters, 500 meters and at the surface. The time, coordinates and bottle number are recorded in the logbook.
Water collection on the deck of the ship
Once all the bottles are closed, the carousel is transported to the deck of the ship and secured.

Taking into account the order of the bottles assigned to each sampling point, the sample is taken.
The hoses connected to the nozzles of each Niskin bottle are carefully placed in 20-litre bottles previously labeled for collection.
Sea Water from the surface (~5 meters) and bottom (~3600 meters) depth is transported to the laboratory on board the vessel.
Filter approximately 15 L of Sea Water through 0.22 µm filters placed in a filtration unit system, sucked in with a vacuum pump.
Equipment
Vacuum Pump
NAME
Millipore
BRAND
WP6211560
SKU
LINK
115 V/60 Hz
SPECIFICATIONS
Durapore™ PVDF Membrane Filters: 0.22μ Pore Size, 0.22 μ Pore Size; Plain; White; Dia.: 25mmThermo FisherCatalog #GVWP02500
Equipment
PS reusable filtration unit
NAME
Wacol
BRAND
300-4000
SKU
LINK
Saturated filters with biomass are stored in sterile aluminum foil and wrapped inParafilm .
A fraction of the filters are stored at 4 °C for later use and another fraction is stored in liquid Nitrogen. These filters are transported to the laboratory.
Note
Make sure to keep nitrogen containers on the boat tightly closed and secured.
Sampling in the marine environment: Sediment
In multiple sediment sampling events the research vessel is repositioned at the same geographical position if drifting occurred.
Before launching the multi-core, a full protocol is followed to guarantee safety during its operation and facilitated the successful sample collection.
Note
The time dedicated to collecting sediment samples is subject to significant variation, with factors including the depth of the station, prevailing climatic conditions at the moment of deploymen, the performance during the descent, stabilization and ascent within the water column, the characteristics of the seafloor, and the retrieval of equipment.
The speed of the winch used for the descent of the multicorer depends on sea conditions. The multibeam system and from the penetration echosounder generate data that is then utilized to comprehend the nature of the seafloor where samples are being collected and to determine the probability of successfully extracting all cores.
The cable tension helps to identify the conditions during the descent of the equipment and the arrival of the multicorer on the seafloor.
Once the multicorer is retrieved and on deck, the equipment is secured before dettaching each core.
Note
If due to climatic conditions the sampling event was unsuccessful, additional sampling events are required in the same station an reposioning of the research vessel is needed.
Each core is documented and photographed on arrival before deattaching from the main frame. One by one each core is identified and deattached, core top and bottom are secured with rubber caps to avoid bottom water and/or sediment to be lost. Each core is placed and identified on a core rack for its later use. Depending on the use of each sample, cores can be processed either in a cold room, in the lab or on deck.

Placing the cores one by one in the core rack for their transport to the assigned processing area.
The processing of the cores can be performed by one or two persons depending on personal ability and experience. In cases where the core requires cutting in layers, the bottom can be retrieved and placed in an extruder. Otherwise no extruder is required. Bottom water can be of interest to some groups and preserving it properly should be considered in advance. Additional gadgets will be required depending on the nature of the study. In the event that the study is concerned with plastic, it is advisable to avoid the use of plastic tools, gloves, extruders, containers, hoses and rubber caps. Similarity, if metals are the focus of the of study, it is recommended to avoid all metals tools for cutting the sediments slices. Avoid transcontaminating from diverse sources in case organic matter or microbiology are the topic of research. Use filtered water to clean, sieve to avoid incorporating water column biota into the samples.
Place the core with the sample and remove the caps firmly to prevent water or sediment leakage that can incline the sediment surface and affect the layers generating bubbles with the borrom water. Maintaining a quality control of the sediment sample is essential for achieving optimal results.
The depth of each sediment layer required for a study is determined by the specific objectives of the research. This procedure can be facilitated by the use of precut rings or with sediment dispensed, thereby ensuring uniform cuts. Each layer should be placed in a prelabelled glass or plastic container or bag. The selection is based on the nature of the study in question, with the objetive of avoiding transcontamination and garantee the quality of the sample. In the case of deep sea samples, it is usually advisable to expedite processing to minimize the risk of warming and poling exposure to sunlight.
Here we show an example of section sampling, the sediment is stored in glass jars.
Sampled sections representation.
Each sample is documented and fixed and stored in the cold room or the laboratory aboard the ship.
To carry out a subsampling of the obtained sediment, obtaining a sample for storage at 4 °C in glass container with a lid and additional sample for storage in liquid nitrogen (30 g ) in sterile containersFalcon Tube (50 mL)Fischer Scientific .
Subsampling can be done withour perturbing the sediment layers by using hand held syringes without the tip.

The remaining sediment can be used by other team mebres, donated for art or eturned to the sea.
Cleaning the corers and the multicorer is among the best ocean practices to avoid transcontaminating samples and habitats from one location to another.
Laboratory storage
Samples are carefully recovered from liquid nitrogen stores.
Note
Handle liquid nitrogen carefully, it reaches extremely low temperatures.
To store samples at -80 °C in an ultra-freezer in the laboratory at the institute
Equipment
GLACIER
NAME
-86ºC ULTRALOW TEMPERATURE FREEZER
TYPE
NuAire
BRAND
NU-99728J
SKU
LINK

![Download 904801-06[498-All].Jpg](https://www.protocols.io/img/extensions/jpg.png)