Feb 10, 2026
Version 2
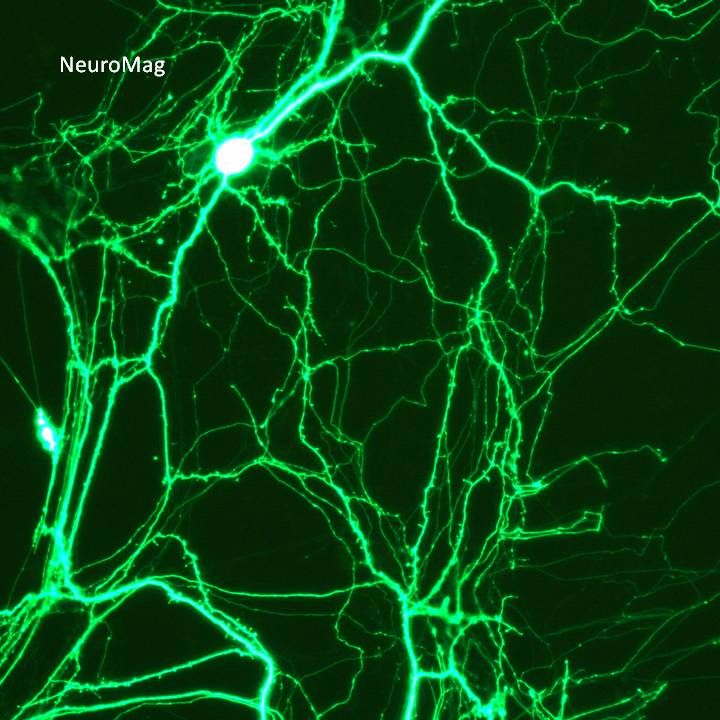
Transfection of Mature Primary Neurons (≥10 DIV) using Magnetofection with NeuroMag V.2
- Cedric Sapet1
- 1OZ Biosciences

External link: https://ozbiosciences.com/neurons/52-50-neuromag-neuron-transfection-reagent.html#/22-capacity-200_l
Protocol Citation: Cedric Sapet 2026. Transfection of Mature Primary Neurons (≥10 DIV) using Magnetofection with NeuroMag. protocols.io https://dx.doi.org/10.17504/protocols.io.261ge196wv47/v2Version created by Cedric Sapet
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: February 10, 2026
Last Modified: February 10, 2026
Protocol Integer ID: 242965
Keywords: magnetofection, primary neurons, transfection, long-term culture, NeuroMag, gene delivery, hippocampal neurons, cortical neurons, Day in Vitro, magnetofectiontm with neuromagtm, gene delivery into primary neuron, magnetic nanoparticles with nucleic acid, transfection of mature primary neuron, complexing magnetic nanoparticle, using neuromag reagent, using magnetofectiontm, mediated gene delivery, neuromag reagent, applicable to primary neuron, neuromagtm, mature primary neuron, dna onto cell, efficient transfection with high viability, achieving efficient transfection, primary neuron, conventional transfection method, spinal neuron, magnetofection with neuromag, using magnetofection, neuromag
Abstract
Post-mitotic neurons are highly sensitive to chemical and physical perturbation and conventional transfection methods show poor efficiency and high toxicity particularly in mature cultures where synaptic networks are functionally established. This protocol describe Magnetofection-mediated gene delivery into primary neurons cultured for 10-22 days in vitro (DIV) using NeuroMag reagent. By complexing magnetic nanoparticles with nucleic acids, magnetofection concentrates DNA onto cells within minutes achieving efficient transfection with high viability. This method is applicable to primary neurons (hippocampal, cortical, cerebellar, spinal neurons...) and is compatible with fluorescent imaging, electrophysiology as well as any biochemical analysis.
Attachments
Image Attribution
OZBiosciences
Guidelines
■ This protocol represent standard protocol that should be used as a starting point. Optimal conditions (DNA amount and NeuroMag ratio) may vary depending on nucleic acid, cell type, size and condition of culture and DIV.
■ Once prepared complexes should be added to the cells within 1 hour.
■ For NeuroMag volumes <1 µL we recommend to prepare a dilution in H2O only ; discard remaining dilution after use.
■ NeuroMagTM is efficient for co-transfections: calculate NeuroMag ratio related to the total plasmid quantity.
Materials
■ Neuron Culture
Poly-L-lysine (Sigma, P-1520)
classic culture medium: MEM supplemented with 15% (vol/vol), Nu-serum, 2% (vol/vol) B27, 0.015 M HEPES (pH 7.2), 0.45% (wt/vol), D-glucose, 1 mM sodium pyruvate, 2 mM L-glutamine and 10 IU ml-1, penicillin–streptomycin.
long term culture medium: MEM supplemented with 10% (vol/vol) Nu-serum, 15 mM HEPES (pH 7.2), 0.45% (wt/vol) D-glucose, 1 mM sodium pyruvate, 2 mM L-glutamine and 10 IU ml-1 penicillin–streptomycin
feeding medium: MEM supplemented with 15 mM, HEPES, 0.45% (wt/vol) D-glucose, 1 mM sodium pyruvate, 2 mM L-glutamine, and 2% (vol/vol) B27.
■ Transfection
Nucleic Acid construct
OptiMEM
Troubleshooting
Problem
Non-optimal neronal rowth
Solution
- check the cell density, pH of the culture medium (7.2) and % of CO2
- supplement cell culture with Nu-serum and B27
Problem
Astrocyte overgrowth
Solution
vary the concentration of Nu-serum
Problem
cells are very sensitive to transfection
Solution
medium can be changed right after the magnetofection procedure: keep the cells onto the magnetic plate and replace transfection medium with fresh pre-warm complete culture medium
Problem
low transfection efficiency
Solution
■ Vary dose of nucleic acid used, ratio of NeuroMag to nucleic acid, cell density, and incubation time.
■ Avoid any physical shock that could activate neurons
Safety warnings
■ Use medium without any supplement (growth factor, antibiotics, serum...) for complex preparation.
Before start
Bring NeuroMag and DNA suspension at room temperature before start
Glass coverslips preparation
Prepare a 1 mg/mL poly-L-lysine solution in water.
Prepare glass coverslips.
Place coverslips in 1% (vol/vol) nitric acid overnight. Overnight
Rinse coverslips vigorously.
Sonicate 3 times for 30 min in distilled water, 3 times in 70% (vol/vol) ehtanol and 2 times in 95 % (vol/vol) ethanol.
Sterilize at 160°c for 1h.
Note
Coverslips can be stored at room temperature under sterile conditions for few months.
Coat coverslips with poly-L-lysine diluted 1:100 and incubate overnight. Overnight
Rinse 3 times in PBS
Note
Rigourous procedure of washing and coating are crucial for neuron culturing. Residual poly-L-lysine is toxic for neurons.
Preparation of the cells of interest
Dissect and collect the tissues of interest in accordance with institutional and national guidelines for animal experimentation.
Dissociate neurons using papain or trypsin at 37°C and stop the action by washing twice with DMEM.
Dilute dissociated neurons at the desired concentration and plate coverslips for 1h at 5% CO2, 37°C.
Transfer coverslip in 12-well plate and add 2 mL culture medium.
Grow the cells under standard culture conditions (5% CO2, 37°C) until transfection experiment.
Long-term culture neurons
For long term transfection a high cellular density is necessary: as a starting point, 800.000 cells per 35 mm dish can be used.
Note
culture medium should be replaced gradually with feeding medium containing B27.
Until DIV 4, use classic culture medium ( MEM supplemented with 15% (vol/vol), Nu-serum, 2% (vol/vol) B27, 0.015 M HEPES (pH 7.2), 0.45% (wt/vol), D-glucose, 1 mM sodium pyruvate, 2 mM L-glutamine and 10 IU ml-1, penicillin–streptomycin).
Start feeding neurons at DIV 5 (MEM supplemented with 15 mM, HEPES, 0.45% (wt/vol) D-glucose, 1 mM sodium pyruvate, 2 mM L-glutamine, and 2% (vol/vol) B27).
Change 50% of the medium every 3 days.
Replace 50% of the culture medium with fresh medium 24H before Magnetofection.
MagnetofectionTM with NeuroMagTM
40m
Complexes preparation
Vortex NeuroMag and place the appropriate amount in a microtube (standard procedure: 3.5µL per µg DNA).
Refer to table 1 below to optimize NeuroMag volume depending on the cell format.
Prepare nucleic acid suspension by diluted DNA into OPTIMEM without any supplement.
Refer to table 1 below for DNA quantity depending on the cell vessel format.
| Format | DNA (µg) | Dilution vol (µL) | NeuroMag (µL) | Transfection vol (mL) | |
| 96-well | 0.5 | 50 | 0.5, 1, 1.5, 1.75 | 0.2 | |
| 24-well | 1 | 100 | 1, 2, 3, 3.5 | 0.5 | |
| 6-well | 4 | 200 | 4, 8, 12, 14 | 2 | |
| 60 mm dish | 10 | 300 | 10, 20, 30, 35 | 4 |
Table 1: recommended DNA and NeuroMag quantities
Add DNA solution to NeuroMag.
Mix by vigourous pipetting.
Incubate at room temperature for 20 min.
20m
Transfection
Note
For long-term cultures (>10 DIV) replace 50% of culture medium 24H before transfection.
Add complexes of NeuroMag/DNA onto cells drop by drop.
Gently rock the plate to ensure uniform distribution.
Place the cells onto magnetic plate.
Incubate under standard culture conditions for 20 min.
20m
Remove the magnetic plate.
Cultivate the cells under standard culture conditions until evaluation of the transgene expression (24 H to 7 days).
Representative protocol for transfection with 1 µg DNA and NeuroMag at ratio 3.5:1
Protocol references
■Buerli T, Pellegrino C, Baer K, Lardi-Studler B, Chudotvorova I, Fritschy JM, et al. Efficient transfection of DNA or shRNA vectors into neurons using magnetofection. Nat Protoc 2007;2(12):3090-101.
■ Sapet C, Laurent N, de Chevigny A, Le Gourrierec L, Bertosio E, Zelphati O, Béclin C. High transfection efficiency of neural stem cells with magnetofection. Biotechniques. 2011 Mar;50(3):187-9.
■ Charrier C, Joshi K, Coutinho-Budd J, Kim JE, Lambert N, de Marchena J, et al. Inhibition of SRGAP2 function by its human-specific paralogs induces neoteny during spine maturation. Cell. 2012 May 11;149(4):923-35.
■ Bartosz F. Grześkowiak, Yolanda Sánchez-Antequera, Edelburga Hammerschmid, Markus Döblinger, Dietmar Eberbeck, Anna Woźniak, Ryszard Słomski, Christian Plank & Olga Mykhaylyk. Nanomagnetic Activation as a Way to Control the Efficacy of Nucleic Acid Delivery. Pharm Res. 2014 DOI. 10.1007/s11095-014-1448-6

