Sep 08, 2025
Total lipid content analysis for coral samples
- Keyla Plichon1,
- Paola Furla1
- 1Université Côte d'Azur

Protocol Citation: Keyla Plichon, Paola Furla 2025. Total lipid content analysis for coral samples. protocols.io https://dx.doi.org/10.17504/protocols.io.81wgbx681lpk/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: July 29, 2023
Last Modified: September 08, 2025
Protocol Integer ID: 85681
Keywords: Coral, lipid content, 96-well plate, total lipid content analysis for coral sample, total lipidic content from frozen coral sample, total lipid content analysis, frozen coral sample, coral sample, lipid content, total lipidic content, animal tissue surface, extraction protocol
Abstract
This protocol aims to extract and quantify total lipidic content from frozen coral samples. This method is adapted from the extraction protocol of Rowan et al., 2020 and the sulfo-phospho-vanillin protocol on 96-well plate published by Cheng et al., 2011. Lipid content is normalized by the animal tissue surface determined by the aluminum technique described by Hoegh-Guldberg, 1988. The method was developed with the guidance of Dr. Mohamed Mehiri and Dr. Louis-Félix Nothias at Université Côte d'Azur.
Guidelines
The protocol takes at least three days to be achieved, depending on the amount of samples.
Materials
Materials:
- Liquid scintillation vial with attached cap (20 mL capacity)
- Glass syringes (10 mL capacity)
- Glass conical centrifuge tubes with phenolic cap (15 mL capacity)
- Disposable glass culture tubes (5 mL capacity)
- Glass bottles (volume capacity depends on the samples treated each day)
- Glass separatory funnels (30-50 mL capacity)
- Glass beakers (volume capacity depends on the samples treated each day)
- Aluminum paper sheets
- 96-well plates with lid
- Multi-channel pipette (50-300 μL)
Equipment:
- -80 °C freezer
- Weighing balance accurate to 4 decimal places
- Freeze Dryer
- Fume hood
- Sonicator
- Digital clock
- Centrifuge
- Heat plate large enough to hold a water bath
- Microplate absorbance reader
Compounds:
- Liquid nitrogen
- Milli-Q water
- Chloroform (100%)
- Methanol (100%)
- Butylated hydroxytoluene
- KCl
- Cholesterol
- Concentrated phosphoric acid (100-80%)
- Vanillin
- Concentrated sulfuric acid (100%)
Troubleshooting
Safety warnings
This procedure uses hazardous chemicals
- Read the SDS forms for each chemical in the procedure.
- Use Viton gloves (0.7 mm) throughout the procedure.
- Wear a lab coat and safety glasses throughout the procedure.
- Dispose of all chemical waste in appropriately labeled containers.
- Perform all the extraction and quantification steps in a fume hood
- Use 96-well plates with lid
Before start
During extraction, all the material used should be made of glass. All glassware should be washed and rinsed in 100% chloroform and allowed to dry in the fume hood before use.
Sample preparation
Weigh and rinse the coral subsamples.
Make sure to keep the sample in liquid nitrogen, and cut a subsample of 1 cm2 with the help of a mortar and a pestle, making sure not to grind it. Rinse the subsample for 5 minutes in MilliQ water to remove salts. Place the subsample in the freeze-drying vial. Return the vial to the freezer for later.
Dehydrate the subsamples.
In the late afternoon, turn on the freeze dryer, load subsamples into it, start the vacuum, and let them dry overnight. The protocol can be stopped here if necessary.
Lipid extraction
Remove subsamples from the freeze dryer.
Turn off the freeze-dryer and quickly tighten the screw caps to the vial to avoid moisture entering the subsample. Keep samples at -80°C until analysis.
Prepare solvents
Three solvents are needed throughout the procedure. On each extraction day, prepare:
- the 2:1 (v/v) chloroform:methanol + 50 mg/L butylated hydroxytoluene (BHT) extraction solvent
Table 1. Guide for extraction solvent volumes, depending upon the number of samples.
- a stock container with 100% chloroform and a stock container with 100% methanol
Table 2. Guide for 100% chloroform and 100% methanol volumes, depending upon the number of samples.
These solvents will be used for lipid extraction and rinsing.
Prepare a potassium chloride, 0.88% solution (daily).
Dissolve the KCl powder in Milli-Q water. This solution will be used for purification.
Table 3. Guide for potassium chloride volumes, depending upon the number of samples.
Extract lipids.
Pour 10 mL of the chloroform:methanol solution into the glass extraction vial containing the freeze-dried sample. Record the time of solvent addition. Sonicate five times for 10s. Keep the vial on ice during sonication and make pauses between if needed, as it will cause heat. Store the vial in the dark for one hour at room temperature.
1h
Filter subsamples.
After one hour, pour the supernatant containing the lipid extract from the extraction vial into a glass centrifuge tube. Using a glass syringe, dispense 2 mL of the extraction solvent into the extraction vial to collect the remaining lipids and pour the rinse into the centrifuge tube. Centrifuge the sample and pour the lipid extract into a clean glass vial or a separatory funnel.
3000 rpm, Room temperature, 00:05:00
5m
Purify lipid extracts.
Pour the lipid extract into a separatory funnel, the stopcock in the closed position. Add 4 ml of the 0.88 % KCl solution and place the stopper. Gently invert the funnel three times, making sure to release pressure.
Drain lipid extract.
Remove the stopper and drain the lower organic phase into a pre-weight clean vial, making sure not to let pass the aqueous (white) phase. Rinse the remaining aqueous with 1 mL of 100% chloroform and repeat the purification step.
Add methanol.
Add 1 mL of 100% methanol to the organic phase, the extract should now be transparent, otherwise, an additional 1 mL of methanol can be added.
Evaporate solvent.
Leave the vial containing the lipid extracts under the hood to let evaporate the solvent overnight between 37 °C and 40 °C. Do not overheat the extract, as heat may alter the lipids.
Figure 1. Organic phase of samples after purification.
Lipid quantifcation sulfo-phospho-vanillin method
Prepare a stock of cholesterol solution and prepare a standard range.
Prepare a stock of a 2 mg/ml cholesterol solution diluted in the extraction solvent. Dilute the stock solution to obtain a range of standards of 0, 0.125, 0.25, 0.5, 1, 1.25 & 1.5 mg/mL. The cholesterol solution should be prepared daily.
Prepare a stock of 17% phosphoric acid.
Dilute the concentrated (100% or 80%) phosphoric acid solution in Milli-Q water.
Prepare a stock of 0.2 mg/mL vanillin reagent.
Dilute the vanillin in the 17% phosphoric acid solution. This solution corresponds to the reagent solution.
Table 4. Guide for reagent solution volumes depending upon the number of samples.
Prepare a stock of concentrated (100%) sulfuric acid.
Keep a small amount of sulfuric acid at room temperature.
Table 5. Guide for sulfuric acid 100% volumes depending upon the number of samples.
Resuspend lipid extract.
Add 4 mL of the extraction solvent to the dried lipids' vial.
Prepare samples.
Pour 150 μL of standard or sample into clean glass tubes (two per sample for duplicates and to speed up the process), and let evaporate the solvent in a water bath at 90°C for 30 min.
Add the sulfuric acid.
After completely evaporating the solvent, cool the tubes on ice for 5 minutes. Add 150 μL of sulfuric acid and place the samples back in a water bath at 90 °C for 20 min.
Cool the tubes.
Place the tubes in an ice bath for 2 min.
Transfer the samples into a 96-well plate.
Transfer 100 μL of each sample.
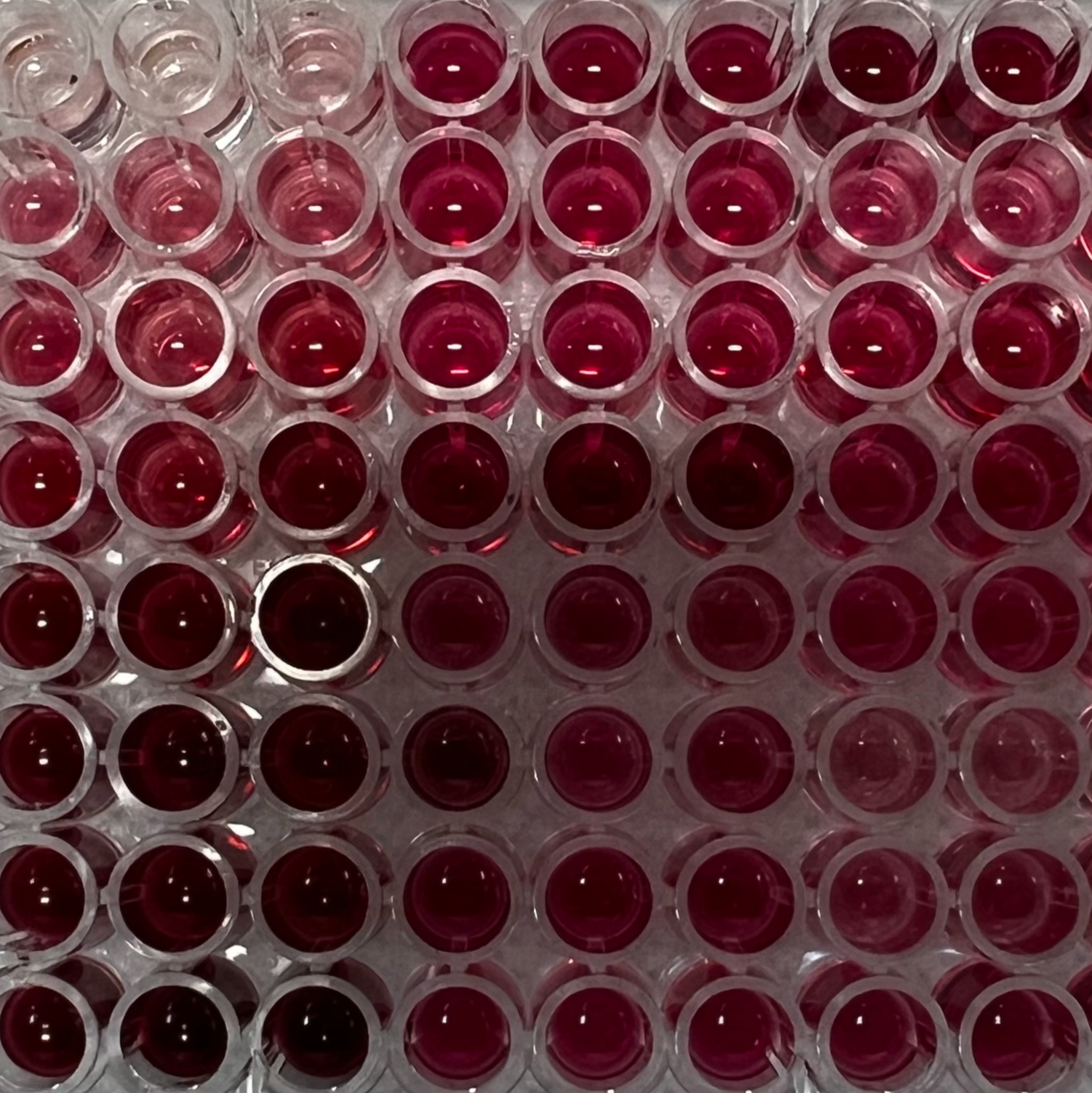
Figure 2. Disposition of standard range and samples in the 96-well plate. Measures are performed in triplicates.
Read the background absorbance in the spectrophotometer.
Determine the background at 540 nm.
Add the reagent.
Add 50 μL of vanillin-phosphoric acid solution into each well.
Incubate samples.
Incubate at 37°C for 15 min.
Figure 3. Standard range and samples after incubation at 37°C for 15 min.
Measure the absorbance at 540 nm.
Mix the samples just before reading absorbance.
Determine lipid content.
Subtract the background and the blank from each sample and standard value. Compute the standard curve and calculate the lipid concentration for each sample. Multiply by the total volume. Some samples must be diluted to ensure they are within the standard range.
Figure 4. Example of a standard curve for determining lipid concentration. Absorbance as a function of lipid concentration.
determining lipid concentration
Normalization
Prepare aluminum range.
Cut pieces of aluminum with different known surfaces and weigh them with a balance accurate to 4 decimal places to create a standard curve.
Figure 5. Example of a standard curve for surface determination. Aluminum paper sheet squares weight as a function of its surface.
The remaining samples can be kept at -80°C for further analyses.
Determine animal tissue coverage.
Cut pieces of aluminum and cover the skeleton surface that was covered with the animal tissue. Weigh the aluminum pieces and determine the corresponding surface with the standard curve.
Normalize by surface.
Divide the total lipid content by the surface of the sample.
Protocol references
