Feb 27, 2018
The production of monocyte-derived dendritic cells from human iPSCs
- Lucas Armitage1,
- Mark Wallet1
- 1University of Florida

Protocol Citation: Lucas Armitage, Mark Wallet 2018. The production of monocyte-derived dendritic cells from human iPSCs. protocols.io https://dx.doi.org/10.17504/protocols.io.ng8dbzw
Manuscript citation:
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: February 27, 2018
Last Modified: March 28, 2018
Protocol Integer ID: 10496
Keywords: dendritic cells from human ipsc, derived dendritic cell, dendritic cell, induced pluripotent stem cell, monocyte, human ipsc, dc, ipsc, cell
Abstract
This protocol is a modification of a previously published method by Wilgenburg et al (PLoS One, 2013) to obtain monocytes and Dendritic cells (DCs) from induced Pluripotent Stem Cells (iPSCs) lines.
Troubleshooting
Before start
1. Order neccessary supplies
| Item | Catalog # | Vendor | |
| hES Medium | |||
| KO DMEM | 10829-018 | Gibco | |
| KO Serum replacement | 10828 | Gibco | |
| NE Amino Acids | M11-003 | PAA | |
| GlutaMAX | 35050 | Gibco | |
| 2-Mercaptoethanol | 31350-010 | Gibco | |
| bFGF | 4114-TC-01M | &D Systems | |
| Pen/Strep | P11-010 | PAA | |
| X-VIVO FACTORY base | |||
| X-VIVO 15 | BE04-418 | Lonza | |
| GlutaMAX | 35050 | Gibco | |
| 2-Mercaptoethanol | 31350-010 | Gibco | |
| Additional Growth Factors | |||
| M-CSF | 300-25 | PeproTech | |
| IL-3 | 200-03 | PeproTech | |
| Rock inhibitor: Y-27632 dihydrochloride | 1254 | Tocris | |
| GM-CSF | 300-03 | PeproTech | |
| IL-4 | 200-04 | PeproTech | |
| Other | |||
| Ultra-low attachment 6 well plates | 3471 | Corning from Appleton Woods/or Fisher Scientific Ltd | |
| hESC-Qualified Matrix Matrigel | 354277 | Corning | |
| mTeSR1 media | 05850 | Stem Cell Technologies |
2. Prepare solutions
X-VIVO Factory base medium
- 500mL X-VIVO 15
- 5mL Glutamax (100X)
- 500μL 2-ME (1000x)
FACTORY medium complete
- 25 mL of basic medium
- 250 μL MCSF / IL-3 cocktail
Stock MCSF = 20 μg/mL (final = 200 ng/mL)
Stock IL-3 = 5 μg/mL (final = 50 ng/mL)
- Pen/Strep
DC DIFFERENTIATION base
- 500 ml CellGenix GMP DC Medium (20801-0500) [Serum Free]
DC DIFFERENTIATION complete
- To a 15 mL aliquot of the base medium, add 50 ng/ml IL-4 and 50 ng/ml GM-CSF (150 ul of alliquotted and mixed cytokines each at 100X [5 ug/ml]) [Do not freeze thaw cytokines. Prepare single use aliquots in CellGenix GMP DC medium and store at -80C]
Sample
Start with a well of a 10 cm plate with iPS cells grown on Matrigel as follows:
A. Culture of iPSCs/ESCs on Matrigel (hES qualified) coated dishes in a 10 cm dish
Note
Cells require media changes every day starting on Day 1. The maintenance media is orange in color to begin with. The following is a guideline, but you can add more fresh media as necessary
Daily Maintenance
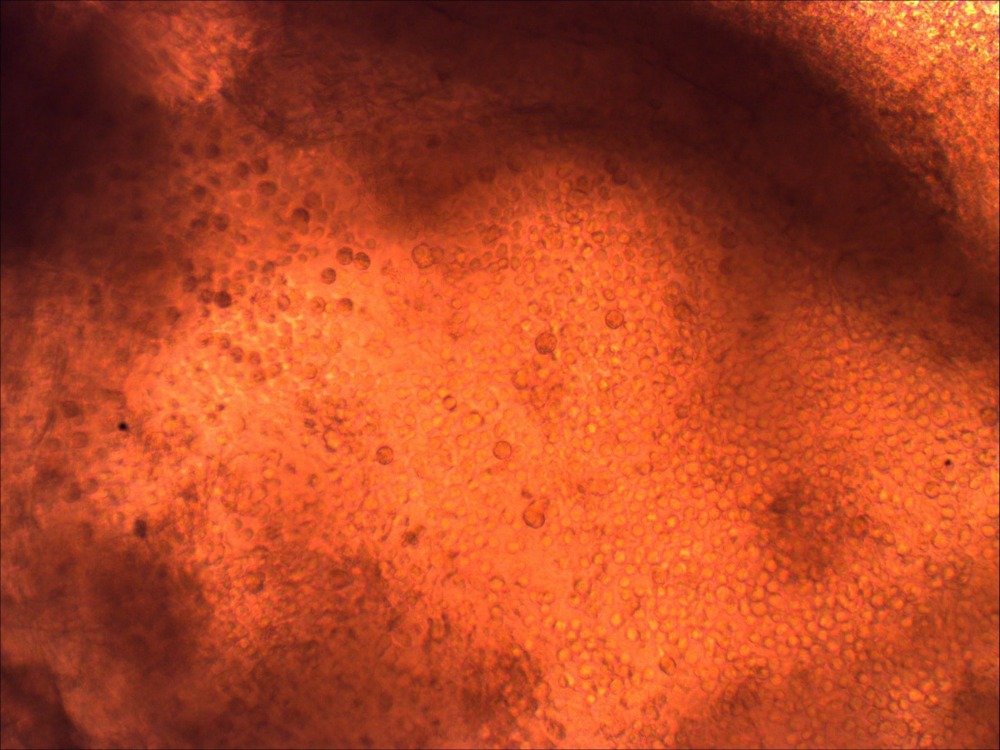
- Day 0, Passage iPSCs onto 10 cm Matrigel coated dish in 8 mL mTesR1 media **passage clumps sparsely so that they will have room to grow into large colonies by ~Day 10** (See Image 1)
- Day 1 Δ media, add 8 mL fresh media
- Day 2 Δ media, add 8 mL fresh media
- Day 3 Δ media, add 9 mL fresh media
- Day 4 Δ media, add 9 mL fresh media
- Day 5 Δ media, add 10 mL fresh media
- Day 6 Δ media, add 10 mL fresh media
- Day 7 Δ media, add 11 mL fresh media
- Day 8 Δ media, add 11 mL fresh media
- Day 9 Δ media, add 12 mL fresh media
Expected result
Image 1: iPSC colonies just before lifting for EB culture.
Setting up Embryoid Bodies (EBs)
Pre-treat iPSCs for 1 hr with 10 μM ROCK inhibitor (Y-27632) to reduce apoptosis of cells that occurs during the scraping step
Add 2 mL fresh media (+10 μM ROCK inhibitor) to each well of a 6-well non-adherent plate (to pre-wet the wells)
Use a cell lifter to gently dislodge the colonies, transfer colonies and media gently to a 15 mL conical tube to allow colonies to gravity settle. Aspirate media as soon as large colonies are settled (this will help remove small colonies or single cells)
Add 12 mL mTeSR1 + ROCK inhibitor to 15 mL conical tube with settled colonies, gently mix and distribute the volume equally among wells of the 6-well plate prepared above (total volume per well = 4 mL mTeSR1 + ROCK inhibitor (10 μM)
Change ¾ media on Day 2 (tip plate and allow EBs to settle to the bottom edge). Rather than using vacuum aspiration it is easier to remove media using a serological pipet. Replace with 4 mL fresh mTeSR1 + ROCK inhibitor per well
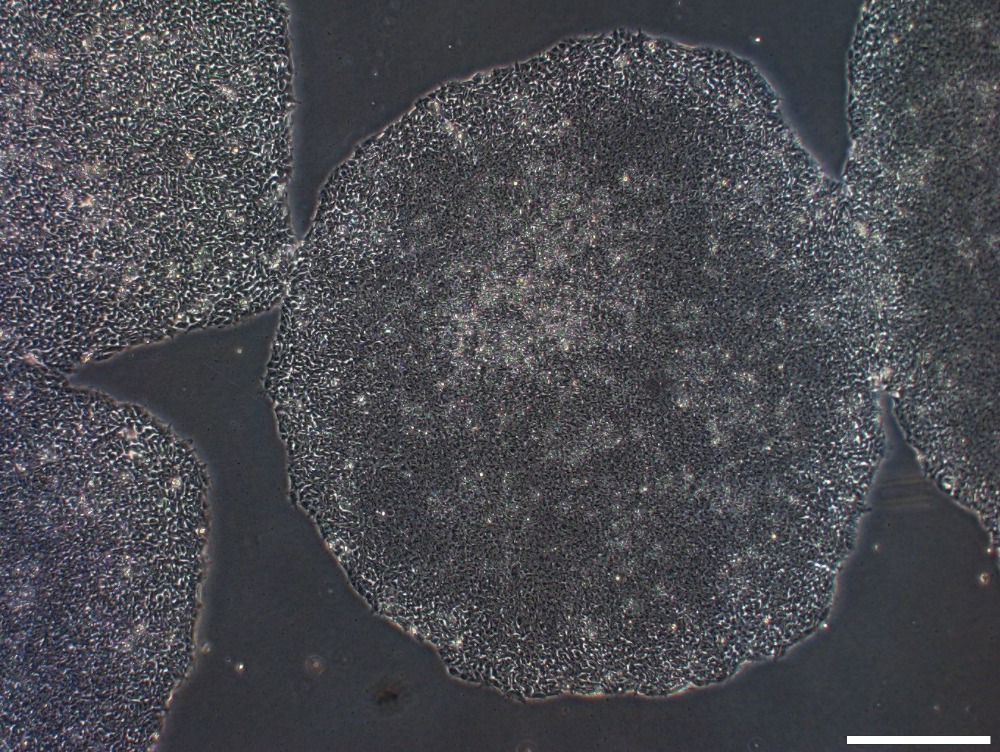
Collect EBs on Day 4. Transfer EBs and media to 15 mL conical tube(s). Gravity settle EBs, remove media, wash with plain XVIVO-15 media (no growth factors). (See Image 2 & 3 for examples of EBs)
Expected result
Image 2: EBs after 4 days culture [Note, this is an exceptionally good looking culture. Often, cystic regions will be less prominent, but differentiation still works]
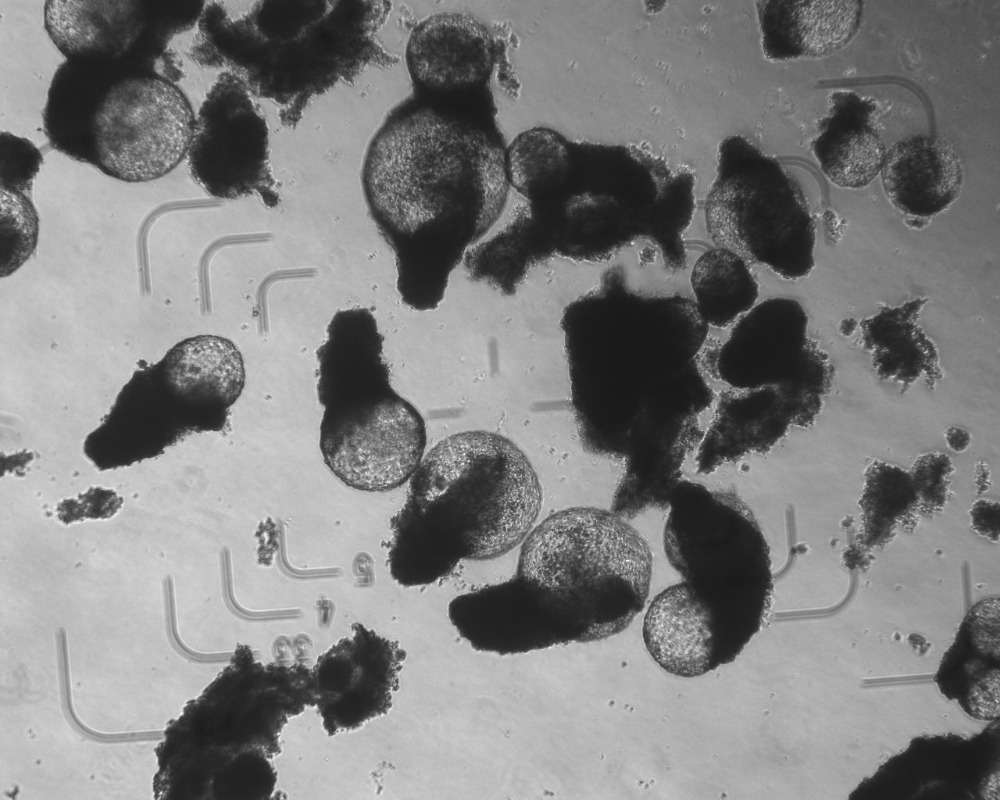
Expected result
Image 3: Less photogenic EBs that still yielded quality monocytes [Note the small debris because the wash step to remove small cells/debris had not been included in the protocol at this point].
Setting up FACTORIES
Estimate the total number of medium to large EBs formed (this is a judgement call that becomes easier only with experience).
Harvest EBs into a 50mL conical tube and allow to settle for a few mins. The large clumps (= good EBs) will sink, whilst individual/dead cells will remain floating. Remove supernatant, add 10ml factory base Repeat settling and wash with Factory base medium.
Resuspend EBs in 4 ml factory complete medium for every 20 EBs. Transfer 4 mL EBs + medium into each well of an adherent, regular tissue culture 6-well plate. Aim to end up with around 15 - 20 EBs per well (too many will inhibit monocyte production). If the original stem cell well was dense the EBs may be split between two or more wells.
Incubate for several days. If there is a lot of cell debris, then medium may need changing (warm medium, and always leave at least 1mL in the well), but otherwise do 50% approximately every 5 days or when the medium becomes yellow. Total volume can be increased to 6mL.
Note
EBs set up as factories in X-VIVO factory medium may not adhere to plates (most often the EBs adhere, but adherence is weak so medium exchange must be done very gently).
Harvesting monocytes for dendritic cell differentiation
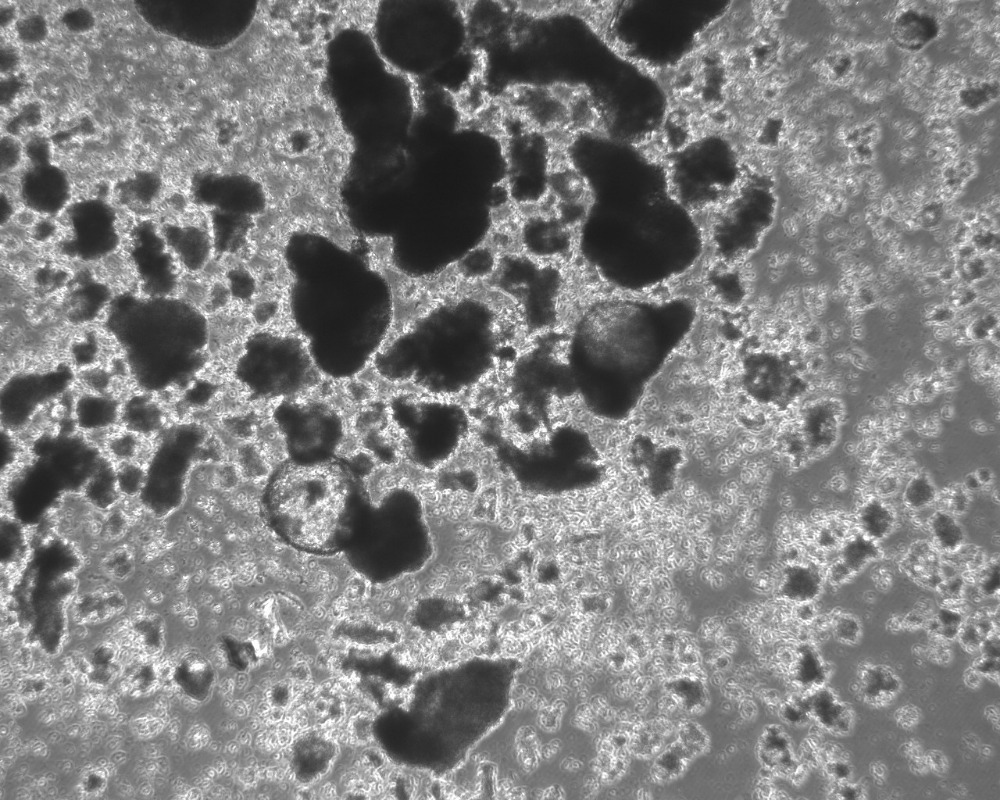
Monocytes will start to appear in supernatant after 10 – 20 days. (See Image 4) The monocyte harvest described below can be performed every 5 days for approximately 4-6 weeks. The first harvest yields the fewest monocytes, but yield tends to increase for harvests 2-5. Total yield from a 6-well plate of factories can reach about 2 million monocytes per harvest.
A. Gently swirl plate then harvest ~5ml of the supernatant (leaving 1ml behind) - be careful not to disrupt factory, and always use warm medium, otherwise factory may curl up. (Care is required for XVIVO factories since factories may not have adhered to plate).
B. Count cells, spin at 400g for 5mins, resuspend pellet in DC DIFFERENTIATION medium in low adherence plates (crucial that only low adherence plates are used here). Plate at 4 x 106 per well of 6 well plate, 4mL/well (or equivalent density for other well-sizes; 5 x 105 works well for 24 well plates).
C. Cells will grow loosley adherent or floating near the bottom in clusters.
D. Add 2 ml of DC DIFFERENTIATION medium at day 3.
E. Remove 2 ml of DC DIFFERENTIATION medium on day 5 and replace with 2 ml fresh DC DIFFERENTIATION medium.
F. At day 7 DCs are ready to use. Pipette gently to dislodge cells from plate and wash 2 X in PBS to remove GM-CSF and IL-4. DCs are usually cultured an additional 2-24 hrs in base GMP DC medium (without GM-CSF or IL-4) before initiation of functional experiments. At this point it is fine to plate on either low adherence plastic if you want to recover cells for flow cytometry or regular tissue culture plastic if you wish the cells to be adherent (for example, for T cell stimulation, we would plate on 96-well cell cluster plastic to allow the DCs to adhere for various treatments and then T cells would be seeded on top of the DCs).
Expected result
Image 4: monocyte production region within adherent EB after 10 days in complete factory medium [Note, the smooth round cells concentrated near the lower right are monocytes. When leaving these foci of monocyte generation and entering the surrounding medium, the monocytes are more vacuolated and often have small dendrites].