Oct 24, 2023
Ten(10)X-compatible Combinatorial Indexing ATAC sequencing (txci-ATAC-seq)
- Hao Zhang1,
- Ryan Mulqueen2,
- Andrew Adey2,
- Darren Cusanovich1
- 1University of Arizona, Department of Cellular and Molecular Medicine, Tucson, AZ;
- 2Oregon Health & Science University

Protocol Citation: Hao Zhang, Ryan Mulqueen, Andrew Adey, Darren Cusanovich 2023. Ten(10)X-compatible Combinatorial Indexing ATAC sequencing (txci-ATAC-seq). protocols.io https://dx.doi.org/10.17504/protocols.io.dm6gp3o68vzp/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: October 22, 2023
Last Modified: October 24, 2023
Protocol Integer ID: 89713
Keywords: Single-cell, ATAC-seq, Chromatin accessibility, Combinatorial indexing, Molecular hashing, compatible combinatorial indexing atac, cell atac, nuclei across multiple sample, atac, combinatorial indexing, seq technique, nuclei, microfluidic barcoding, seq, tn5, based microfluidic barcoding
Funders Acknowledgements:
NIH/NIGMS
Grant ID: R35GM137896
NIH/NIGMS
Grant ID: R35GM124704
NIH/NIMH, BICCN
Grant ID: RF1MH128842
Abstract
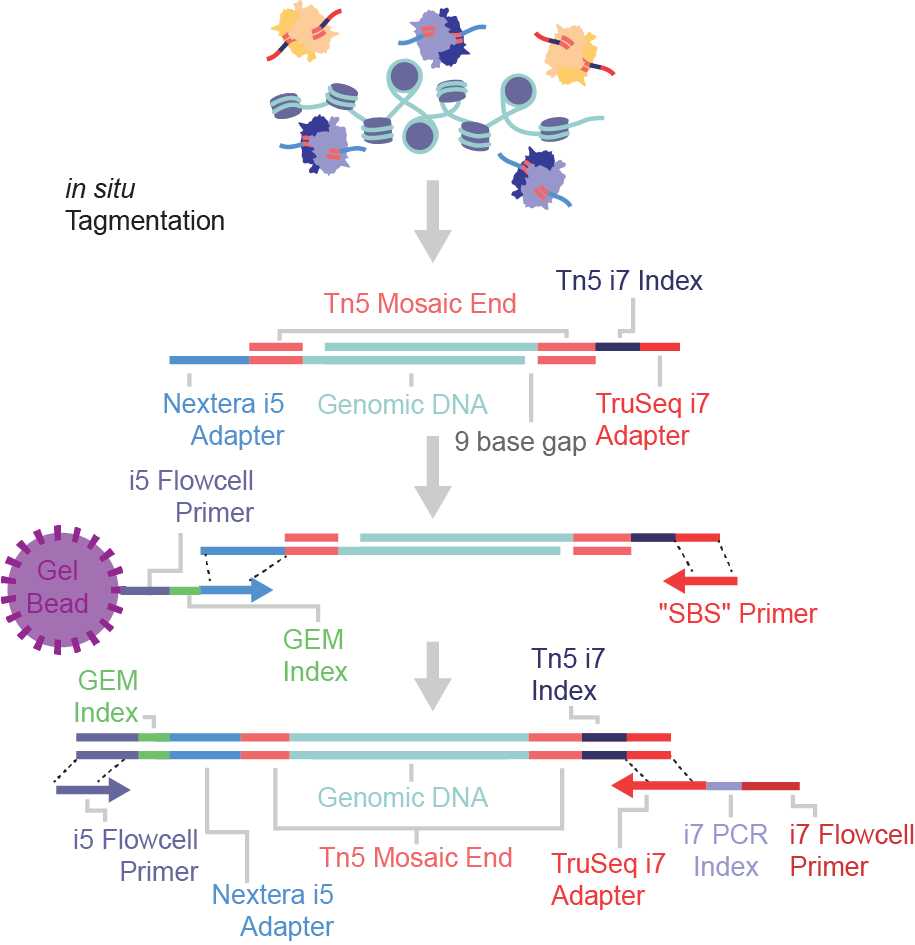
The txci-ATAC-seq method is a large-scale single-cell ATAC-seq technique that combines the Tn5-based pre-indexing with the 10X Chromium-based microfluidic barcoding. This molecular hashing strategy enables the profiling of up to 200,000 nuclei across multiple samples in a single emulsion reaction.
Materials
Loading Tn5
- Annealing Buffer:
| Reagent | Final Concentration | Per 10 ml | |
| 1M Tris-HCl, pH8.0 | 40 mM | 400 μl | |
| 5M NaCl | 50 mM | 100 μl | |
| H2O | 9.5 ml |
- Sequences of Tn5 linker oligos (The ‘N’ bases shown in the Tn5ME-B sequence represent the Tn5 barcodes):
| Linker Oligo | Sequence 5’ -> 3’ | |
| Tn5ME-A | TCGTCGGCAGCGTCAGATGTGTATAAGAGACAG | |
| Tn5ME-B | CGTGTGCTCTTCCGATCTNNNNNNNNAGATGTGTATAAGAGACAG | |
| Tn5MErev | [phos]CTGTCTCTTATACACATCT |
- Sequences of Tn5ME-B barcode:
| Row | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | |
| A | GAACCGCG | AGGTTATA | TCATCCTT | CTGCTTCC | GGTCACGA | AACTGTAG | GTGAATAT | ACAGGCGC | CATAGAGT | TGCGAGAC | GACGTCTT | AGTACTCC | |
| B | TGGCCGGT | CAATTAAC | ATAATGTG | GCGGCACA | CTAGCGCT | TCGATATC | CGTCTGCG | TACTCATA | ACGCACCT | GTATGTTC | CGCTATGT | TATCGCAC | |
| C | TCTGTTGG | CTCACCAA | TATTAGCT | CGCCGATC | TCTCTACT | CTCTCGTC | CCAAGTCT | TTGGACTC | GGCTTAAG | AATCCGGA | TAATACAG | CGGCGTGA | |
| D | ATGTAAGT | GCACGGAC | GGTACCTT | AACGTTCC | GCAGAATT | ATGAGGCC | ACTAAGAT | GTCGGAGC | CCGCGGTT | TTATAACC | GGACTTGG | AAGTCCAA | |
| E | ATCCACTG | GCTTGTCA | CAAGCTAG | TGGATCGA | AGTTCAGG | GACCTGAA | TGACGAAT | CAGTAGGC | AGCCTCAT | GATTCTGC | TCGTAGTG | CTACGACA | |
| F | TAAGTGGT | CGGACAAC | ATATGGAT | GCGCAAGC | AAGATACT | GGAGCGTC | ATGGCATG | GCAATGCA | GTTCCAAT | ACCTTGGC | CTTATCGG | TCCGCTAA | |
| G | GCTCATTG | ATCTGCCA | CTTGGTAT | TCCAACGC | CCGTGAAG | TTACAGGA | GGCATTCT | AATGCCTC | TACCGAGG | CGTTAGAA | CACGAGCG | TGTAGATA | |
| H | GATCTATC | AGCTCGCT | CGGAACTG | TAAGGTCA | TTGCCTAG | CCATTCGA | ACACTAAG | GTGTCGGA | TTCCTGTT | CCTTCACC | GCCACAGG | ATTGTGAA |
Isolation of nuclei from cell lines
Buffers to make beforehand
- Omni Resuspension Buffer (RSB; filter and store at 4℃):
| Reagent | Final Concentration | Per 50 ml | |
| 1M Tris-HCl, pH 7.5 | 10 mM | 500 µl | |
| 5M NaCl | 10 mM | 100 µl | |
| 1M MgCl2 | 3 mM | 150 µl | |
| H2O | 49.25 ml |
- Omni TD Buffer (filter and store at -20℃):
| Reagent | Final Concentration | Per 50 ml | |
| 1M Tris-HCI, pH 7.5 | 20 mM | 1 ml | |
| 1M MgCl2 | 10 mM | 0.5 ml | |
| Dimethyl Formamide | 20% | 10 ml | |
| Sterile water | 38.5 ml |
- Freezing buffer stock solution (FB stock; filter and store at -20℃):
| Reagent | Final Concentration | Per 50 ml | |
| 1M Tris-HCI, pH 8.0 | 50 mM | 2.5 ml | |
| 1M Mg(OAc)2 | 5 mM | 0.25 ml | |
| 50% Glycerol | 25% | 25 ml | |
| 0.5M EDTA | 0.1 mM | 0.01 ml | |
| Sterile water | 22.24 ml |
Buffers to make on the day of the experiment
- RSB Lysis Buffer (200 µl per sample):
| Reagent | Final Concentration | Per 200 µl | |
| RSB | ~1x | 194 µl | |
| 10% Igepal-CA630 | 0.1% | 2 µl | |
| 1% Digitonin | 0.01% | 2 µl | |
| 10% Tween-20 | 0.1% | 2 µl |
- RSB Washing Buffer (1 ml per sample):
| Reagent | Final Concentration | Per 1 ml | |
| RSB | ~1x | 990 µl | |
| 10% Tween-20 | 0.1% | 10 µl |
- Freezing buffer working solution (FBW; 1 ml for every 3 millions of nuclei):
| Reagent | Final Concentration | Per 1 ml | |
| FB stock | ~1x | 975 µl | |
| 1M DTT | 5 mM | 5 µl | |
| Protease Inhibitors (Sigma P8340) | 2% (v/v) | 20 µl |
txci-ATAC-seq protocol
Buffers to make beforehand
- TMG washing buffer (50 ml):
| Reagent | Final Concentration | Per 50 ml | |
| 0.2M Tris-acetate pH 7.8 | 10 mM | 2.5 ml | |
| 1M Magnesium acetate | 5 mM | 0.25 ml | |
| 50% Glycerol | 10% | 10 ml | |
| Sterile water | 37.25 ml |
- Loading Buffer was made by mixing buffer 1 (5x) and buffer 2 below (5x):
1. Buffer1 (5x):
| Reagent | Final Concentration | Per 1000 μl | |
| 0.2M Tris-acetate pH 7.6 | 50 mM | 250 μl | |
| 1M Magnesium acetate | 25 mM | 25 μl | |
| Dimethyl Formamide | 50% | 500 μl | |
| H2O | 225 μl |
2. Buffer2 (5x):
| Reagent | Final Concentration | Per 1000 μl | |
| 100% Glycerol | 50% | 500 μl | |
| 5M NaCl | 100 mM | 20 μl | |
| 1M Tris-HCl, pH 7.5 | 50 mM | 50 μl | |
| 0.5M EDTA | 0.1 mM | 0.2 μl | |
| 1M DTT | 1 mM | 1 μl | |
| H2O | 428.8 μl |
3. Loading Buffer:
| Reagent | Final concentration | Per 250 μl | |
| 5x Buffer1 | 1x | 50 μl | |
| 5x Buffer2 | 1x | 50 μl | |
| H2O | 150 μl |
- Omni Resuspension Buffer (RSB; filter and store at 4C):
| Reagent | Final Concentration | Per 50 ml | |
| 1M Tris-HCl, pH 7.5 | 10 mM | 500 µl | |
| 5M NaCl | 10 mM | 100 µl | |
| 1M MgCl2 | 3 mM | 150 µl | |
| H2O | 49.25 ml |
- 10% (100 mg/mL) BSA:
Dissolve 1 g powdered Fraction V or molecular biology grade BSA in 10 mL of distilled H2O. Then, store in aliquots at −20°C.
Buffers to make on the day of the experiment
- PBSB (containing 0.04% BSA) (2 ml):
| Reagent | Final concentration | Per 2 ml | |
| PBS | ~1x | 1960 μl | |
| 20 mg/ml BSA | 0.4 mg/ml | 40 μl |
- RSB washing buffer (4 ml per sample):
| Reagent | Final Concentration | Per 4 ml | |
| RSB | ~1x | 3920 μl | |
| 10% Tween-20 | 0.1% | 40 μl | |
| 10% BSA | 0.1% | 40 μl |
- Loading buffer supplemented with SBS oligo (LBS):
| Reagent | Final Concentration | Per 150 μl | |
| Loading Buffer | ~1x | 140 μl | |
| 75 μM SBS Oligo | 5 μM | 10 μl |
Note: SBS oligo sequence (5’-3’) is CGTGTGCTCTTCCGATCT
- 300 μM DAPI solution:
| Reagent | Final Concentration | Per 200 μl | |
| 10.9 mM DAPI | 300 μM | 5.5 μl | |
| H2O | 194.5 μl |
Note: Add 1 μl of 300 uM DAPI to each 100 μl nuclei to make a final concentration of 3 μM for staining.
Troubleshooting
Loading Tn5
Resuspend Tn5ME-A, Tn5ME-B, and Tn5MErev in the annealing buffer to a final concentration of 100 μM.
Prepare annealed linker A: Mix one volume of Tn5ME-A with one volume of Tn5MErev in a PCR tube.
e.g. 100 μl Tn5ME-A + 100 μl Tn5Merev.
Prepare annealed linker B: Mix one volume of each barcoded Tn5ME-B with one volume of Tn5MErev on a 96-well plate.
e.g. 10 μl Tn5ME-B (Index A1) + 10 μl Tn5MErev.
Mix briefly by pipetting and run the following PCR program in a thermocycler for annealing oligos.
| Temperature | Time | |
| 95 ℃ | 5 min | |
| Slowly Cool down to 65 ℃ | -0.1 ℃/sec | |
| 65 ℃ | 5 min | |
| Slowly Cool down to 4 ℃ | -0.1 ℃/sec |
The annealed oligos can be kept at -20°C for long-term storage.
Add 1 μl of each annealed linker (A and B) to 20 μl of the Tn5 stock (0.3 mg/ml) on a 96-well plate with a unique annealed linker B in each well.
Mix briefly by pipetting, and then incubate at 23°C for 30 minutes in a thermomixer at 350 rpm.
Store at -20°C.
Isolation of nuclei from cell lines
Remove approximately 10x106 cells from culture.
Note
The nuclei isolation protocol was adapted from Corces MR, et al. 2017.
Citation
LINK
Pellet the cells at 500 RCF at 4°C for 5 min in a swinging-bucket centrifuge.
Aspirate supernatant.
Resuspend pellet in 200 µl RSB Lysis Buffer.
Incubate on ice for 3 minutes.
Add 1 ml RSB Washing Buffer.
Take 10 µl nuclei and dilute it with 40 µl of Omni TD buffer, then count the nuclei on a hemocytometer by adding 50 µl Trypan blue solution to the diluted nuclei (Note: we found that adding the RSB-resuspended nuclei straight to Trypan blue solution will cause inflation of nuclei, and diluting nuclei in Omni TD buffer before exposure to Trypan blue improves the nuclei integrity).
Pellet the remaining nuclei in RSB Washing Buffer at 500 RCF for 10 min at 4°C in a fixed-angle centrifuge.
Resuspend nuclei pellet in FBW at ~3 million nuclei/ml.
Snap-freeze nuclei in liquid nitrogen, and then transfer the cryovials to a liquid nitrogen dewar (or -80°C) for long-term storage.
Isolation of nuclei from lung tissue
The following protocol can be used to isolate nuclei from lung tissues.
Citation
LINK
txci-ATAC-seq: Preparing nuclei
Take out flash-frozen nuclei (~3 million in 1 ml) from liquid nitrogen for each sample and thaw in a water bath at 37°C for about 1 min.
Add 3 ml RSB washing buffer to an empty 15 ml tube for each sample.
Transfer 1 ml nuclei stored in the freezing buffer to the 15 ml tube containing 3 ml RSB washing buffer.
Pellet the nuclei at 500 RCF for 10 min at 4°C.
Resuspend nuclei with 1 ml RSB washing buffer and then transfer to a 1.5 ml LoBind tube through Flowmi (40 micron).
Pellet the nuclei at 500 RCF for 5 min at 4°C in a fixed-angle centrifuge.
Resuspend nuclei with 100 μl of 1X PBSB for each sample.
Count nuclei with DAPI:
Add 1 μl of 300 μM DAPI to 100 ul nuclei;
Incubate on ice for 5 mins;
Add 10 μl stained nuclei to the countess slide to count nuclei.
txci-ATAC-seq: 96 barcoded Tn5 transposition
Prepare TD mix:
| Reagent | Final Concentration | Per Rxn | X120 rxn (in a 2ml tube) | |
| 2X Nextera TD Buffer* | 1X | 12.5 µl | 1500 | |
| 1% Digitonin | 0.01% | 0.25 µl | 30 | |
| 10% Tween-20 | 0.1% | 0.25 µl | 30 |
*Omni TD buffer can be used to replace the Illumina Nextera TD buffer.
Citation
LINK
Thaw a 96-well plate preloaded with 5 μl of barcoded Tn5 on ice. Mix by brief shaking at 1400 rpm for 30 seconds, spin for a minute at 2000 RCF at 4°C, and carefully unseal the aluminum foil seal.
Dilute nuclei to 2857 nuclei/μl in PBSB and then mix 7 μl diluted nuclei with 13 μl TD mix for each well.
Add 20 μl nuclei/TD mix mixture to each well of the 96-well plate containing 5 μl of barcoded Tn5 per well (total 25 μl).
Seal the plate using Bio-Rad Microseal B film.
Mix by shaking at 1000 rpm for one minute.
If liquid splashes to the seal, briefly spin at 500 RCF for 10 sec.
Incubate at 37°C for 60 min in a thermocycler block with a heated lid (47°C).
Thaw TMG washing buffer on ice.
Remove the plate from the thermocycler.
Briefly centrifuge at 500 RCF for 10 sec at 4°C.
Incubate the plate on ice for 5 min.
Pool nuclei in a LoBind 12-tube strip and then transfer them to a 15 ml conical tube preloaded with 400 ul of TMG.
Add 50 μl/well of TMG to the first row of the plate and pipette them throughout the whole plate to wash out the residual nuclei remaining in the plate.
After washing the last row of the plate, the TMG was transferred to the same conical tube that was used to collect the barcoded nuclei.
Centrifuge nuclei at 500 RCF for 10 min at 4°C.
Remove most of the supernatant.
Resuspend nuclei with 500 μl of TMG, then transfer to a 1.5 ml LoBind Tube through Flowmi.
Centrifuge at 500 RCF for 5 min at 4°C.
Remove most of the supernatant and resuspend the nuclear pellet with 30 μl of LBS.
Count nuclei with a hemocytometer.
Take the volume of solution containing the desired number of nuclei and dilute it with the LBS to make a total of 15 μl.
Use the 15 μl dilution as input into the 10X Chromium droplet generator – follow Step 2, page 24 of the Chromium Single Cell ATAC kit instructions (10x Document CG000209 Rev D) to complete the assay.
txci-ATAC-seq: Modification of 10X Chromium protocol
For Step 2.5. GEM Incubation:
a. Incubate in a thermal cycler with the following protocol (Lid temperature at 105℃).
| Temperature | Time | |
| 72 ℃ | 00:05:00 | |
| 98 ℃ | 00:00:30 | |
| 98 ℃ | 00:00:10 | |
| 59 ℃ | 00:00:30 | |
| 72 ℃ | 00:01:00; Go to step 3, repeat 7X (Total 8 cycles) | |
| 15 ℃ | Hold |
b. Store at 15°C for up to 18 h or at −20°C for up to 1 week, or proceed to the next step.
For Step 4.1 Sample Index PCR
c. Add 2.5 μl of customized i7 TruSeq primer (25 μM) containing an 8 bp custom barcode to each 10X library. Record assignment. Pipette mix and centrifuge briefly.
d. Incubate in a thermal cycler with the following protocol (Lid temperature at 105℃).
| Temperature | Time | |
| 98 ℃ | 00:00:45 | |
| 98 ℃ | 00:00:20 | |
| 67 ℃ | 00:00:30 | |
| 72 ℃ | 00:00:20; Go to step 2, repeat 4X (Total 5 cycles) | |
| 72 ℃ | 00:01:00 | |
| 4 ℃ | Hold |
Sequencing
| Sequencing Read | Cycles | |
| Read 1N | 51 bp | |
| i7 index (I1) | 8 bp | |
| i5 index (I2) | 16 bp | |
| Read 2N | 78 bp |
Citations
Step 18
Nikita Joshi, Alexander Misharin. Single-nucleus isolation from frozen human lung tissue for single-nucleus RNA-seq
https://protocols.io/view/single-nucleus-isolation-from-frozen-human-lung-ti-zu8f6zwStep 27
Corces MR, Trevino AE, Hamilton EG, Greenside PG, Sinnott-Armstrong NA, Vesuna S, Satpathy AT, Rubin AJ, Montine KS, Wu B, Kathiria A, Cho SW, Mumbach MR, Carter AC, Kasowski M, Orloff LA, Risca VI, Kundaje A, Khavari PA, Montine TJ, Greenleaf WJ, Chang HY. An improved ATAC-seq protocol reduces background and enables interrogation of frozen tissues.
https://doi.org/10.1038/nmeth.4396Step 8
Corces MR, Trevino AE, Hamilton EG, Greenside PG, Sinnott-Armstrong NA, Vesuna S, Satpathy AT, Rubin AJ, Montine KS, Wu B, Kathiria A, Cho SW, Mumbach MR, Carter AC, Kasowski M, Orloff LA, Risca VI, Kundaje A, Khavari PA, Montine TJ, Greenleaf WJ, Chang HY. An improved ATAC-seq protocol reduces background and enables interrogation of frozen tissues.
https://doi.org/10.1038/nmeth.4396