Mar 10, 2026
Synthesis of AOAO-12 Nucleic Acid Stain (equivalent to EvaGreen™)

- Aishwarya Venkatramani1,
- Ishtiaq Ahmad1,
- Ljiljana Fruk1,
- Jennifer Molloy2,3
- 1Department of Chemical Engineering and Biotechnology, University of Cambridge;
- 2Department of Biochemistry, University of Cambridge;
- 3International Centre for Genetic Engineering and Biotechnology
- Reclone.org (The Reagent Collaboration Network)
- Open Bioeconomy Lab

Protocol Citation: Aishwarya Venkatramani, Ishtiaq Ahmad, Ljiljana Fruk, Jennifer Molloy 2026. Synthesis of AOAO-12 Nucleic Acid Stain (equivalent to EvaGreen™). protocols.io https://dx.doi.org/10.17504/protocols.io.n92ld1nk8l5b/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: February 27, 2026
Last Modified: March 10, 2026
Protocol Integer ID: 244159
Keywords: alkylation of an acridine orange base, affinity nucleic acid stain, nucleic acid stain, acridine orange base, final dimeric dye, carboxylic acid, stage chemical synthesis, synthesis
Funders Acknowledgements:
BBSRC
Grant ID: BB/Y007808/1
Abstract
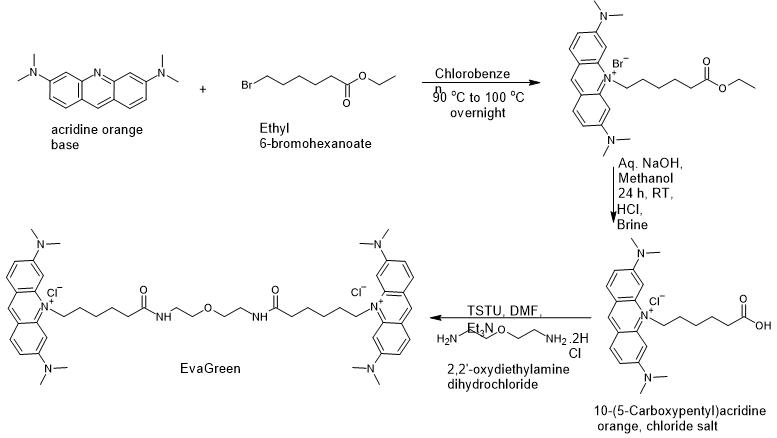
This protocol outlines the two-stage chemical synthesis of AOAO-12/EvaGreen[TM] (Dye No. 19 in US Patent No. US7803943B2, filing date 17 March 2006, expected expiry date 16 March 2026), a high-affinity nucleic acid stain. Originally described in patent literature, this procedure has been adapted for in-house laboratory production. The process involves the N-alkylation of an acridine orange base to create a carboxylic acid intermediate, followed by a TSTU-mediated conjugation with a flexible linker to form the final dimeric dye.
Attachments
Materials
Acridine Orange base | CAS 494-38-2 | Sigma-Aldrich 235474 | 5 g
Ethyl 6-bromohexanoate | CAS 25542-62-5 | Sigma-Aldrich 324728 | 4.20 g
Chlorobenzene | CAS 108-90-7 | Sigma-Aldrich 284513 | 10 mL
Sodium hydroxide (NaOH) | CAS 1310-73-2 | Sigma-Aldrich S8045 | 1.43 g
Methanol | CAS 67-56-1 | Sigma-Aldrich 179337 | ~100 mL
Concentrated hydrochloric acid | CAS 7647-01-0 | Sigma-Aldrich 320331 | ~5 mL
Sodium chloride (for saturated solution) | CAS 7647-14-5 | Sigma-Aldrich S3014 | ~20 g
Ethyl acetate | CAS 141-78-6 | Sigma-Aldrich 270989 | ~260 mL
TSTU (N,N,N′,N′-Tetramethyl-O-(N-succinimidyl)uronium tetrafluoroborate) | CAS 105832-38-0 | Sigma-Aldrich 385530 | 320 mg
Triethylamine | CAS 121-44-8 | Sigma-Aldrich T0886 | 0.25 mL
2,2′-Oxydiethylamine dihydrochloride | CAS 60792-79-2 | Sigma-Aldrich 176095 | 25 mg
DMF (dimethylformamide, anhydrous) | CAS 68-12-2 | Sigma-Aldrich 227056 | 5 mL
Protocol materials
Acridine Orange baseMilliporeSigmaCatalog #235474
Troubleshooting
Safety warnings
Specific Chemical Hazards:
Chlorobenzene is a suspected carcinogen and CNS depressant. All steps involving it must be done in a fume hood with no open flames nearby (flash point 29°C).
DMF (dimethylformamide) is a reproductive toxin (Category 1B), readily absorbed through skin even through many glove types double-glove with nitrile, and change gloves frequently.
Acridine Orange base is a potential mutagen and sensitizer. Avoid inhalation of the powder and use inside the hood.
Protocol Hazards:
Step 5 (pouring hot mixture into EtOAc): This is a bump/splatter risk. Pour slowly down the side of the vessel. EtOAc is also highly flammable so ensure no ignition sources nearby.
Step 9 (rotovap): Set the water bath to ≤42°C as shown in your protocol image as higher temperatures risk bumping and loss of product. Ensure the vacuum is released slowly to avoid bumping the flask.
Step 9 (HCl acidification followed by saturated NaCl): Confirm the solution is fully cooled before adding NaCl. The "salting out" crash can be quite sudden and use a vessel with sufficient headspace.
Step 14 and repeats (EtOAc precipitation cycles): EtOAc additions to DMF solutions can generate heat and fumes. Work in the fume hood throughout.
Step 17 (centrifugation): Centrifuge tubes must be chemically compatible with DMF use polypropylene, not polystyrene else you would risk melting the tube and contaminating your sample
Overall Reaction Scheme
Key Chemical Components
| A | B | C | D | E | |
| Reagent | CAS | Sigma-Aldrich/Merck Catalogue Number | Required Amount | Unit | |
| Acridine Orange base | 494-38-2 | 235474 | 5 | g | |
| Ethyl 6-bromohexanoate | 25542-62-5 | 324728 | 4.2 | g | |
| Chlorobenzene | 108-90-7 | 284513 | 10 | mL | |
| Sodium hydroxide (NaOH) | 1310-73-2 | S8045 | 1.43 | g | |
| Methanol (MeOH) | 67-56-1 | 179337 | 100 | mL | |
| Concentrated hydrochloric acid (HCl) | 7647-01-0 | 320331 | 5 | mL | |
| Sodium chloride (NaCl) | 7647-14-5 | S3014 | 20 | g | |
| Ethyl acetate (EtOAc) | 141-78-6 | 270989 | 260 | mL | |
| TSTU | 105832-38-0 | 385530 | 320 | mg | |
| Triethylamine | 121-44-8 | T0886 | 0.25 | mL | |
| 2,2′-Oxydiethylamine dihydrochloride | 60792-79-2 | 176095 | 25 | mg | |
| Dimethylformamide (DMF) | 68-12-2 | 227056 | 5 | mL |
Table: key chemical components required
Part 1: Preparation of 10-(5-Carboxypentyl)acridine orange
1d
Combine 5 g of acridine orange (Acridine Orange baseMilliporeSigmaCatalog #235474 ) with one equivalent of ethyl 6-bromohexanate in 10 mL of chlorobenzene.
Safety information
Chlorobenzene is a suspected carcinogen and central nervous system depressant.
All steps involving chlorobenzene must be performed in a fume hood with no open flames nearby (flash point is only 29°C).
Note
In this protocol, “one equivalent” means adding the exact same number of molecules of the alkylating agent as there are molecules of the starting material.
Note: Because Acridine Orange base is often supplied with variable dye content, it is best practice to verify the "Dye Content" percentage on your specific bottle's Certificate of Analysis (CoA) and adjust this calculation if it differs from the standard 75%.
Starting Material: You are using 5 g of Acridine Orange base and Sigma-Aldrich Cat # 235474 dye content is 75%, i.e. 3.75 g .
Since Acridine Orange's molar mass is 265.35 g/mol, this equates to 0.0141 moles (14.1 mmol).
To provide one equivalent, you must also add 0.0141 moles of ethyl 6-bromohexanoate.
Multiplying 0.0141 moles by ethyl 6-bromohexanoate’s molar mass (223.11 g/mol) gives you the required weight of 3.15 g .
Stir the mixture Overnight at a temperature between90 °C and100 °C .
Expected result
Figure: Reaction setup on a stir plate, showing the transition from a clear to a deep reddish-orange solution as the reaction progresses after addition of acridine orange
Figure: the chemical reaction where the nitrogen in the acridine ring is alkylated by the hexanoic acid chain.
Pour the heated reaction mixture into approximately 200 mL of EtOAc (ethyl acetate).
Safety information
This step has a bump/splatter risk. Pour slowly down the side of the vessel.
EtOAc is also highly flammable so ensure there are no ignition sources nearby.
Precipate Overnight then collect the orange precipitate through filtration and dry it under a vacuum.
Expected result
Figure: image of expected orange precipitate once filtered.
Suspend 5 g (~11.9 mmol) of this crude product in ~100 mL of methanol and add 3 equivalents of NaOH (Sodium Hydroxide) i.e. 1.43 g (35.7 mmol), dissolved in 30 mL of water.
Stir the suspension at Room temperature for 24:00:00 .
1d
Remove the methanol via evaporation, using a rotavap with water bath set to 42 °C
Safety information
Setting temperatures higher than 42 °C risk bumping and loss of product.
Ensure the vacuum is released slowly to avoid bumping the flask.
Part 2: Preparation of AOAO-12/EvaGreen™ (Dye No. 19)
To a suspension of 147 mg (0.35 mmol) of Intermediate (1) (10-(5-carboxypentyl)acridine orange chloride salt) in 5 mL of DMF, add 0.15 mL Et3N (Triethylamine) (1.05 mmol) and 320 mg TSTU (1.05 mmol)
Safety information
DMF (dimethylformamide) is a reproductive toxin (Category 1B), readily absorbed through the skin even through many glove types. Double-glove with nitrile gloves, and change gloves frequently.
Expected result
Figure: final step in production of AOAO-12/EvaGreen, Dye No. 19 in US Patent No. US7803943B2
Stir this mixture at Room temperature for 00:15:00 .
Add Et3N (0.1 mL ) and 2,2′-oxybis(ethylamine) dihydrochloride (25 mg ) to the mixture.
Expected result
Figure: mixture set up and ready for stirring overnight
Stir the resulting mixture at Room temperature Overnight .
Add 20 mL of EtOAc to precipitate the crude product.
Safety information
EtOAc additions to DMF solutions can generate heat and fumes.
Work in the fume hood throughout.
Re-dissolve the crude product in 5 mL (approximately until the product dissolves) DMF and precipitate it using 20 mL EtOAc.
Repeat the re-dissolving and precipitation cycle i.e. and repeat 3-4 times until the supernatant remains clear and the solid reaches a consistent dark yellowish-green color.
Separate the resulting solid (approx 250 mg ) using centrifugation at 4000 rpm, Room temperature, 00:10:00
Safety information
Centrifuge tubes must be chemically compatible with DMF!
Use polypropylene, not polystyrene else you risk melting the tube and contaminating your sample.
Expected result
Figure: AOAO-12 dissolved in DMF then precipitated by EtOAc, prior to centrifugation
Part 3: Storage of centrifuged product (solid pellet)
Dissolve the dried solid product AOAO-12 in DMSO at 70 mg/mL (e.g. 0.35 g in 5 mL DMSO) to prepare a concentrated stock solution.
Aliquot into amber tubes and store at -20 °C , desiccated and protected from light. Avoid repeated freeze-thaw cycles.
For working stocks, dilute the 70 mg/mL DMSO stock of AOAO-12 with water.
From our results, a concentration of 17.5 mg/mL AOAO-12 (approximately 4-fold dilution) is functionally equivalent to a 2× commercial EvaGreen‱ working solution, as determined by fluorescence intensity matching against a fixed dsDNA concentration under identical instrument settings.
Note
The molar concentration of commercial EvaGreen‱ from Biotium is proprietary.
Equivalence should be confirmed empirically for each new synthesis batch of AOAO-12 by comparing fluorescence signal in the presence of a known dsDNA standard against commercial EvaGreen‱ on your specific fluorometer.
Part 4: Validation and QC
Quality control of each AOAO-12 synthesis batch was performed by UV-visible absorption spectroscopy, confirming a characteristic peak at ~500 nm consistent with the bis-acridine chromophore and spectral agreement with commercial EvaGreen‱ across a serial dilution series. Batches showing residual absorption from the uncoupled mono-acridine intermediate or poor linearity across dilutions should be subjected to additional DMF/EtOAc precipitation cycles prior to use.
UV-visible absorption spectra of purified in-house EvaGreen‱ compared with commercial EvaGreen‱ (after several washes with EtOAc and DMF)
Absorption spectra (350–650 nm) of purified in-house EvaGreen‱ across a serial dilution series (X/2 to X/32, where X = 70 mg/mL stock in DMSO) alongside the mid-synthesis intermediate (X/2, X/4) and commercial EvaGreen‱ at 200×, 20×, and 2× dilutions.
UV-visible absorption spectra of in-house EvaGreen‱ prior to purification.
Absorption spectra (350–650 nm) of crude in-house EvaGreen‱ at the stock concentration (70 mg/mL, "X") and half-stock concentration (X/2, 35 mg/mL) compared with the uncoupled mono-acridine intermediate, the coupling reaction wash, and commercial EvaGreen‱ at 200× and 20× dilutions. The uncoupled intermediate and coupling wash show a shifted absorption profile relative to the final product, confirming the presence of unreacted species that are removed during subsequent purification. These spectra are taken when dissolved in DMF.
Example Usage in Protocols
Protocol references
Mao, F. and Leung, W.-Y. (2010). Methods of using dyes in association with nucleic acid staining or detection and associated technology. US Patent No. US7803943B2. Biotium Inc. Filed: 15 June 2009. Granted: 28 September 2010.
Mao, F. and Leung, W.-Y. (2010). Methods of using dyes in association with nucleic acid staining or detection and associated technology. US Patent Application No. US20100323453A1 (granted as US8232050B2). Biotium Inc. Filed: 11 August 2010. Published: 23 December 2010. Granted: 31 July 2012.