Sep 30, 2025
Version 2
Synergistic Enhancement of AAV Gene Delivery in 2D Cells and 3D Organoids Using Polybrene and Hydroxychloroquine V.2
Peer-reviewed method
- Hyeon-Jin Na1,2,
- Yongbo Shin2,3,
- Seung-Hyun Kim1,
- Seung pil Jang1,
- Myung Jin Son2,3,
- Yong Min Choi1,2,
- Hyeon Gyeol Jeon2,3,
- Ok-Seon Kwon1,2,
- Kyung-Sook Chung1,2
- 1Center for Gene and Cell Therapy, Korea Research Institute of Bioscience and Biotechnology (KRIBB), Republic of Korea;
- 2Department of Advanced Bioconvergence, Korea University of Science & Technology (UST), Republic of Korea;
- 3Stem Cell Convergence Research Center, Korea Research Institute of Bioscience and Biotechnology, Daejeon, Republic of Korea
- PLOS ONE Lab ProtocolsTech. support email: [email protected]

External link: https://doi.org/10.1371/journal.pone.0336164
Protocol Citation: Hyeon-Jin Na, Yongbo Shin, Seung-Hyun Kim, Seung pil Jang, Myung Jin Son, Yong Min Choi, Hyeon Gyeol Jeon, Ok-Seon Kwon, Kyung-Sook Chung 2025. Synergistic Enhancement of AAV Gene Delivery in 2D Cells and 3D Organoids Using Polybrene and Hydroxychloroquine. protocols.io https://dx.doi.org/10.17504/protocols.io.4r3l29mjjv1y/v2Version created by Hyeon-Jin Na
Manuscript citation:
Na H, Shin Y, Kim S, Jang SP, Son MJ, Choi YM, Jeon HG, Kwon O, Chung K (2025) Synergistic enhancement of AAV gene delivery in 2D cells and 3D organoids using polybrene and hydroxychloroquine. PLOS One 20(11). doi: 10.1371/journal.pone.0336164
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: September 24, 2025
Last Modified: September 30, 2025
Protocol Integer ID: 228092
Keywords: Polybrene, Hydroxychloroquine sulfate (HCQs), Organoids, Virus transduction, Adeno-associated virus(AAV), Gene Therapy, synergistic enhancement of aav gene delivery, aav gene delivery, significant challenges for efficient gene delivery, efficient gene delivery, gene delivery, advancing gene therapy application, treatment with virus, gene therapy application, aav transduction, 3d organoid, liver organoid model, using polybrene, liver organoid system, enabled organoid, aav vector, hydroxychloroquine, current limitations in organoid, evaluating aav vector, 2d cell, viral entry, endosomal processing, polybrene
Funders Acknowledgements:
National Research Council of Science & Technology
Grant ID: GTL24022-000
Korea Research Institute of Bioscience and Biotechnology (KRIBB) Research Initiative Program
Grant ID: KGM5362521
National Research Foundation of Korea (NRF)
Grant ID: RS-2024-00352135
Abstract
Recent advances in three-dimensional (3D) culture platforms have enabled organoids to serve as physiologically relevant models for recapitulating human biology and assessing therapeutic efficacy and toxicity. Despite their promise, their complex architecture presents significant challenges for efficient gene delivery, thereby limiting their broader application in drug discovery and translational research. To overcome this challenge, we developed a sequential treatment strategy that combines polybrene (PB), which facilitates viral entry, and hydroxychloroquine (HCQ), which modulates endosomal processing. By applying PB as a pre-treatment and HCQ as a post-treatment, we achieved an approximate 1.3- to 2-fold increase in adeno-associated virus (AAV) transduction efficiency in both retinal and liver organoid models compared to single-agent treatments, and a 1.7- to 2.5-fold increases compared to treatment with virus alone. Importantly, this combinatorial treatment preserved cellular integrity, as confirmed by minimal TUNEL assay and high overall viability. Our findings demonstrate that sequential administration PB and HCQ significantly improves AAV transduction in 3D retinal and liver organoid systems, offering a robust method to improve gene delivery. This approach not only overcomes current limitations in organoid-based research but also supports the development of more predictive platforms for evaluating AAV vectors and advancing gene therapy application.
Guidelines
- The stock concentration may vary depending on experimental requirements.
- Sterilize 1.5 mL micro-centrifuge tubes prior to use.
- The cell culture process can be optimized based on cell line characteristics, such as trypsin-EDTA incubation time.
- The optimal maturation day for retinal organoids is between day 90 to day 120.
Note
Differentiation day may vary according to the experimental objectives.
- For chemical treatment, add the diluted chemical directly to the media, adjusting for the buffer volume.
- When conducting multiple reactions, it is advisable to prepare a master mix that includes a 10% excess to account for pipetting errors.
Materials
- Laboratory Equipment
- CO2 incubator, clean bench, Vacuum pump
- Pipette, pipette aid, table-top centrifuge, centrifuge, vortex
- Refrigerator (4 °C ), freezer( -20 °C ) and deep-freezer (-80 °C )
- Bright-field microscope, ZEISS confocal microscope (LSM800), Cytation 5
- Real-time PCR, Cryostat microtome
- GraphPad Prism, ZEISS blue software, BioTek Gen5
- Personal Protective Equipment (PPE) at a minimum laboratory coat with fitted sleeves, latex or nitrile gloves and safety glasses
- Materials
| A | |
| 50-mL, 25-mL, 10-mL, 5-mL, 2-mL sterile pipette | |
| 1000-l, 200-l, 10-l pipette tips | |
| 50-mL, 15-mL conical tubes | |
| 1.5-mL sterile micro-centrifuge tubes | |
| 35-mm, 100-mm cell culture dishes | |
| 24-well, 48-well cell culture plates | |
| 96-well v-bottom ultra-low attachment plates |
- Reagents for stock solution preparations
| Reagent | Company | Cat # | |
| DPBS, no calcium no magnesium | ThermoFisher | 14190144 | |
| UltraPure DNase/RNase-Free Distilled Water | Invitrogen | 10977015 | |
| Dimethyl Sulfoxide (DMSO), cell culture reagent | Santa Cruz | sc-358801 |
DPBS, no calcium, no magnesiumThermo FisherCatalog #14190144
UltraPure™ DNase/RNase-Free Distilled WaterThermo Fisher ScientificCatalog #10977015
Dimethyl Sulfoxide (DMSO), cell culture reagent (CAS 67-68-5)Santa CruzCatalog #sc-358801
- Reagent for cell line culture
| Reagent | Company | Cat # | |
| DMEM | ThermoFisher | 11995-065 | |
| Fetal Bovine Serum | ThermoFisher | 14000044 | |
| Penicillin/Streptomycin | ThermoFisher | 15140-122 | |
| Trypsin-EDTA(0.5%), no phenol red | ThermoFisher | 15400054 | |
| PBS pH 7.4(10X) | ThermoFisher | 70011-044 |
DMEMGibco - Thermo Fisher ScientificCatalog #11995065
Penicillin-Streptomycin (10,000 U/mL)Thermo Fisher ScientificCatalog #15140122
Trypsin-EDTA (0.5%) no phenol redThermo Fisher ScientificCatalog #15400054
10X PBS pH 7.4Thermo Fisher ScientificCatalog #70011044
- Reagent for hPSCs and Organoid culture
| Reagent | Company | Cat # | |
| mTeSR1 complete kit | STEMCELL | ST85850 | |
| DMEM/F12 | ThermoFisher | 11320-033 | |
| IMDM | ThermoFisher | 12440053 | |
| Ham F12 | ThermoFisher | 11765054 | |
| dPBS | ThermoFisher | 14190144 | |
| Matrigel | Corning | 354277 | |
| Knockout Serum replacement | ThermoFisher | 10828028 | |
| Fetal Bovine Serum | ThermoFisher | 14000044 | |
| ReLeSR | STEMCELL | ST05872 | |
| Gentle cell dissociation reagent | STEMCELL | ST07174 | |
| N2 supplement | ThermoFisher | 17504044 | |
| GlutaMAX | ThermoFisher | 35030061 | |
| Chemically Defined Lipid Concentrate | ThermoFisher | 11905031 | |
| Penicillin/Streptomycin | ThermoFisher | 15140-122 | |
| Cell Recovery solution | Corning | 354253 | |
| Trypsin-EDTA | Thermo | 25200056 | |
| Y-27632 | Tocris | 4423 | |
| Advanced DMEM/F12 | Thermo Fisher | 12634028 | |
| HEPES | Thermo Fisher | 15630080 | |
| B27 Supplement with Vitamin A | Thermo Fisher | 17504044 | |
| N-Acetylcysteine | Sigma-Aldrich | A9165 | |
| A83-01 | Tocris | 2939 | |
| DAPT | Sigma-Aldrich | D5942 | |
| Dexamethasone | Sigma-Aldrich | D4920 | |
| [Leu]-Gastrin I human | Sigma-Aldrich | G9145 | |
| Human HGF | Peprotech | 100-39 | |
| Human EGF | Peprotech | AF-100-15 | |
| Human BMP7 | Peprotech | 120-03 | |
| Human FGF19 | Peprotech | 100-32 |
mTeSR™1STEMCELL Technologies Inc.Catalog #85850
DMEM/F-12Thermo Fisher ScientificCatalog #11320033
IMDMThermo FisherCatalog #12440053
Hams F12Thermo ScientificCatalog #11765054
DPBSThermo Fisher ScientificCatalog #14190144
Corning® Matrigel®CorningCatalog #354277
KnockOut™ Serum ReplacementThermo FisherCatalog #10828028
ReLeSRSTEMCELL Technologies Inc.Catalog #05872
Gentle Cell Dissociation ReagentSTEMCELL Technologies Inc.Catalog #07174
Chemically Defined Lipid ConcentrateThermo FisherCatalog #11905031
Penicillin/StreptomycinInvitrogen - Thermo FisherCatalog #15140-122
Cell Recovery solutionCorningCatalog #354253
Trypsin-EDTA (0.25%), phenol redThermo FisherCatalog #25200056
CHIR 99021TocrisCatalog #4423
Advanced DMEM/F-12Thermo FisherCatalog #12634028
HEPES 1MThermo Fisher ScientificCatalog #15630080
N-Acetyl-L-cysteineMerck MilliporeSigma (Sigma-Aldrich)Catalog #A9165
A 83-01TocrisCatalog #2939
DAPTMerck MilliporeSigma (Sigma-Aldrich)Catalog #D5942
[Leu15]-Gastrin I humanMerck MilliporeSigma (Sigma-Aldrich)Catalog #G9145
Gibco™ Human HGF Recombinant Protein, PeproTech®Fisher ScientificCatalog #100-39-10UG
Epidermal growth factorpeprotechCatalog #AF-100-15
Human BMP-7 Recombinant Protein, PeproTech®Thermo Fisher ScientificCatalog #120-03P-10UG
Human FGF-19, Animal-Free Recombinant Protein, PeproTech®Thermo Fisher ScientificCatalog #AF-100-32-25UG
- Reagent for AAV transduction
| Reagent | Company | Cat # | |
| Hydroxychloroquine (HCQ) | Tocris | 5648 | |
| Polybrene (Hexadimethrine bromide) | Sigma | 107689 |
Hydroxychloroquine sulfateTocrisCatalog #5648
Hexadimethrine bromideMerck MilliporeSigma (Sigma-Aldrich)Catalog #107689
- Preparation of Medium
| Medium | Composition | |
| 10DMEM medium | 10% Fetal Bovine Serum, 1% Penicillin and Streptomycin supplied in DMEM media | |
| mTeSR1 medium | 100ml of mTeSR1 5X supplement supplied in mTeSR1 basal media | |
| gfCDM Neural induction medium | 10% Knockout Serum replacement, 1% Chemically Defined Lipid Concentrate, 450 M monothioglycerol, 1% Penicillin and streptomycin supplied in basal media (IMDM:Ham F12 (1:1)) | |
| Neural retina maturation medium | 10% Fetal Bovine Serum, 1% GlutaMAX, 1% N2, 0.5M Retinoic acid, 100 M Taurine supplied in DMEM/F12 media | |
| Virus medium | 10% Fetal Bovine Serum, 1% GlutaMAX, 1% N2, 1% Penicillin and Streptomycin supplied in DMEM/F12 media | |
| Liver organoid differentiation medium(DM) | 1% Penicillin/Streptomycin (P/S), 1% GlutaMAX, 1% HEPES, 1X N2 Supplement, 1X B27 Supplement with Vitamin A, N-Acetylcysteine 1 mM, A83-01 0.5 μM, DAPT 10 μM, Dexamethasone 3 μM, [Leu]-Gastrin I human 10 μM, Human HGF 25 ng/ml, Human EGF 50 ng/ml, Human BMP7 25 ng/ml, Human FGF19 100 ng/ml supplied in Advanced DMEM/F12 media | |
| Basal medium | 1% Penicillin/Streptomycin (P/S) supplied in Advanced DMEM/F12 media |
- Cell lines
| Cell line | Description | Company, Cat # | |
| HEK293FT | fast-growing, highly transfectable clonal isolate derived from human embryonal kidney cells | ThermoFisher, R70007 | |
| 661W | immortalized cone photoreceptor cell line derived from the retinal tumor of a mouse | kindly provided by Prof. Muayyad Al-Ubaidi, University of Houston | |
| H9 | Human embryonic stem cell (hESC) | WI Cell, WA09 |
- Material for analysis preparation
| Reagent | Company | Cat # | |
| 4% Paraformaldehyde | Biosesang | PC2031-050-00 | |
| Tritone X-100 | Sigma-Aldrich | X100 | |
| Tween-20 | Sigma-Aldrich | P7949 | |
| Sucrose | LPS Solution | SUC05 | |
| Tissue-Tek O.C.T. Compound | SAKURA | 4583 | |
| Aqueous Mounting Medium | Vector Laboratories, Inc | H-5501 | |
| ImmEdge Pen | Vector Laboratories, Inc | H-4000 | |
| In Situ Cell Death Detection Kit, Fluorescein | Roche | 11684795910 | |
| Cell Counting Kit-8 | DOJINDO Laboratories | CK04 |
Troubleshooting
Safety warnings
Proper Personal Protective Equipment (PPE) needed; a minimum laboratory coat with fitted sleeves, latex or nitrile gloves and safety glasses
Formulation of chemical
Polybrene
Prepare autoclaved tertiary distilled water.
Dissolve the polybrene powder in tertiary distilled water to make a final concentration of 5 µL ~10 µL .
Filter polybrene solution with 0.22 m filter.
Aliquot polybrene stock into 1.5 mL sterile micro-centrifuge tubes.
Hydroxychloroquine
Prepare autoclaved tertiary distilled water.
Dissolve the HCQ tablet in tertiary distilled water to make a final concentration of 10 millimolar (mM) .
Filter HCQ solution with 0.22 m filter.
Aliquot HCQ stock into 1.5 mL sterile micro-centrifuge tubes.
Preparation of cell line
Sub-culture of normal cell line
Aspirate culture media and wash with 1X PBS.
Add 1 mL of 0.05% Trypsin-EDTA in the culture plate and incubate culture plate in 5% CO2, 37 °C culture incubator for approximately 00:02:00 .
2m
Add 1 mL of 10DMEM medium and detach the cancer cell by tapping the plate with hands.
Transfer the detached cells to 15 mL conical tube and centrifuge it under 1000 rpm, 00:03:00 .
3m
Aspirate supernatant and add 1 mL of 10DMEM medium to resuspend cells.
Seed a ratio of 1/5 of the cells into a new 100-mm cell culture dish with 10DMEM medium.
Seeding of cell lines
Repeat steps in Sub-culture of cell lines [ ].
Dilute 100 µL to 1/10 by adding 900 µL of fresh media, making a final volume of 1 mL .
Mix 20 µL of 1/10 diluted cell resuspension with 20 µL of Trypan Blue in a 1.5 mL sterile micro-centrifuge tube.
Clean a hematocytometer and load 10 µL of the sample in each side.
Count the cells and seed appropriate number of cells into a new 48-well plate.
Sub-culture of human Embryonic Stem Cells
Human embryonic stem cells (hESCs; H9) were maintained under feeder-free conditions using mTeSR1 medium on Matrigel-coated culture plates at 37 °C with 5% CO2
The culture medium was refreshed on a daily basis.
For routine passaging, colonies were dissociated into small clumps with ReLeSR and reseeded at a split ratio of 1:10–1:20 approximately once per week.
Preparation of organoids
Retinal organoids
Perform retinal organoids differentiation according to previously validated protocols.
Citation
LINK
Prepare retinal organoids and transfer retinal organoids to a new 96 well U-bottom or flat-bottom low attachment plate.
Change medium into fresh pre-warmed virus medium up to 200 µL .
Liver organoids
Perform liver organoids differentiation according to previously validated protocols.
Citation
LINK
Citation
LINK
Prepare liver organoid differentiation medium (DM) and basal medium, and store them at 4 °C until use.
Dilute Matrigel in cold DPBS at a 1:100 ratio and coat a flat-bottom 24-well plate for 01:00:00 in a 37 °C incubator. (Keep the Matrigel On ice at 4 °C .)
1h
Harvest the organoid embedded in Matrigel dome using a 1000 μL pipette tip, transfer it into microcentrifuge tubes, and briefly spin down using a mini centrifuge to pellet the organoids.
Remove the supernatant, resuspend the organoid pellet in basal medium, and briefly spin down again using a mini centrifuge.
After removing the supernatant, add cold Cell Recovery Solution (60 μL per dome) and incubate the cultures in Cell Recovery solution at 4 °C for approximately 00:10:00 .
10m
Briefly centrifuge the cultures to separate the organoids from the Cell Recovery solution. Remove the supernatant and wash the cultures with cold DPBS twice.
Remove the supernatant and resuspend the organoids in DM (100 μL per well of a flat-bottom 24-well plate).
Add 400 µL of fresh DM to each well of the Matrigel-coated 24-well plate ( ), and seed the 12-15 organoids per well by adding 100 µL of organoid suspension (as prepared in ). (Additionally, prepare three wells of organoids for cell counting, to be used in below.)
To anchor the organoids (but not fully attached) to the bottom of the Matrigel-coated plate, incubate the organoids for 04:00:00 -06:00:00 in a 37 °C , 5% CO2 incubator.
10h
Treatment of chemicals
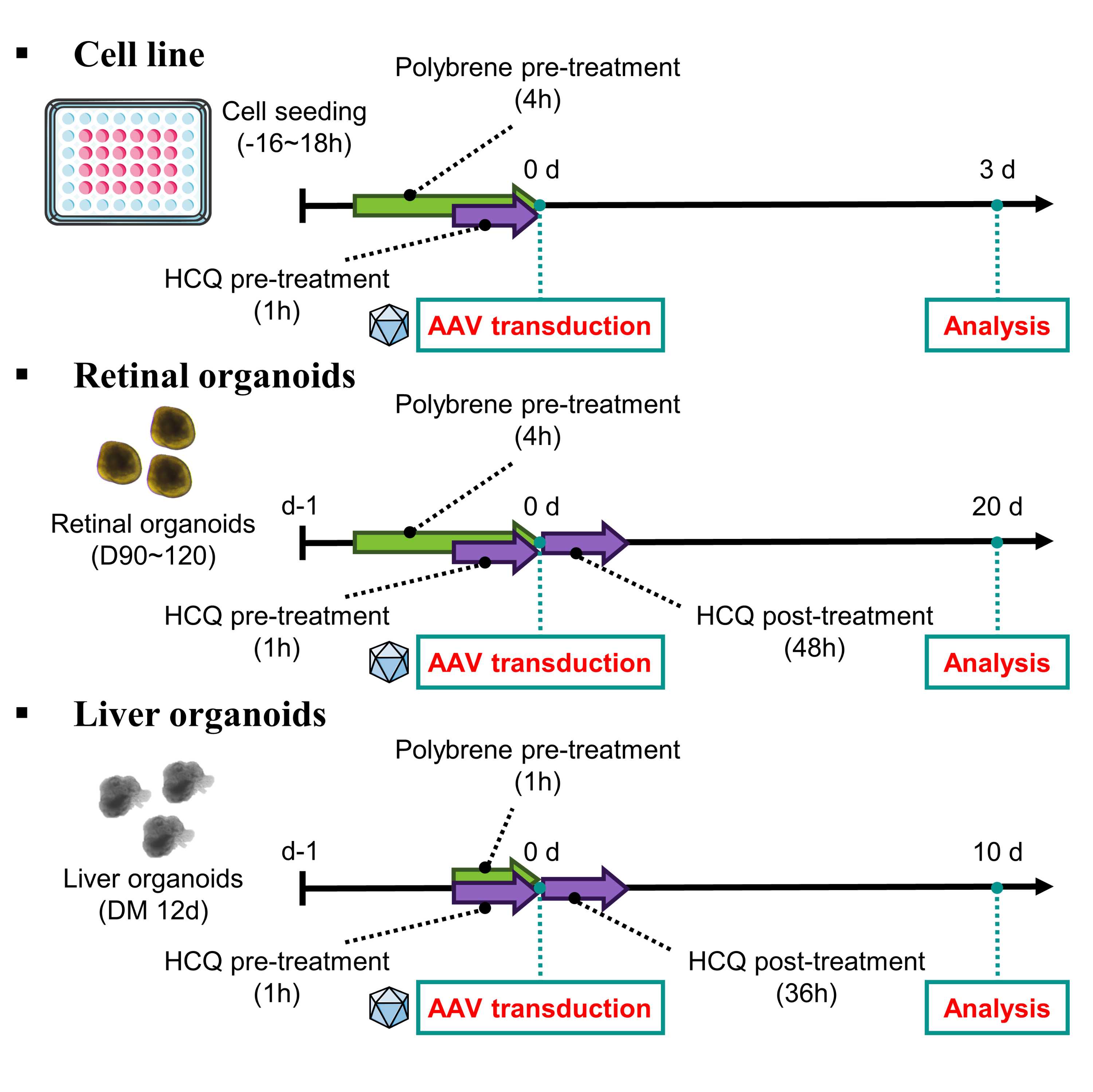
Treatment of polybrene
16:00:00 to 18:00:00 after seeding an appropriate number of cells per well, treat with the polybrene at a concentration of 5 µL ~10 µL .
In the case of organoids, on the day organoids are transferred, polybrene can be treated at a concentration of of 5 µL ~15 µL .
About 01:00:00 to04:00:00 after, AAV can be transduced in either cells or organoids.
Note
Cell viability under these treatment conditions was assessed and found to remain above 80% (see Analysis for cell viability section).
Treatment of HCQ
16:00:00 to 18:00:00 after seeding an appropriate number of cells per well, treat with the HCQ at a concentration of 15 μM .
In the case of organoids, on the day organoids are transferred, HCQ at a concentration of 10 μM , 15 μM , 20 μM .
01:00:00 after, AAV can be transduced in either cells or organoids.
In the sequential treatment protocol for organoids, HCQ is applied for 36:00:00 to 48:00:00 post-viral transduction.
Note
Cell viability under these treatment conditions was assessed and found to remain above 80% (see Analysis for cell viability section).
Virus transduction
Transduction of AAV to cell line and retinal organoids
Observe the morphology and condition of cells.
Dilute AAV in 1X PBS at the desired MOI. (Ex. 2×104vg/cell or 1×1010vg/organoid)
Note
AAV stock needs to be diluted with the appropriate dilution factor. The Volume of transduced AAV should be over 1 µL to avoid pipetting errors.
Transduce the diluted AAV into the cells.
Incubate in a 37 °C , 5% CO2 incubator.
In the case of retinal organoids, Incubate for 15 days in a 37 °C , 5% CO2 incubator. Change the medium every 2 days.
Transduction of AAV to liver organoids
To calculate the treatment concentration of AAVs, transfer organoids from three wells (n=3) as prepared in into microcentrifuge tubes, and briefly spin down using a mini centrifuge.
Remove the supernatant, resuspend the organoid pellet in basal medium, and spread the organoid suspension onto the cell culture dish.
To dissociate the organoids into single cells for counting, mechanically chop the organoids using a surgical blade.
Transfer the cells into microcentrifuge tube and incubate them with Trypsin-EDTA for 00:10:00 in a 37 °C incubator.
Note
Every00:05:00 , pipette up and down using a 200 µL pipette tip to fully dissociate the cells into a single-cell suspension.
10m
Count the number of cells using a standard hematocytometer or automated cell counter. (10–12 organoids per well is approximately equivalent to 1×105 cells per well.)
Change the medium to 250 µL of DM supplemented with 10 μM Y-27632 and the appropriate concentration of AAVs. (Ex, Use 1×101 to 1×103 vg/cell for evaluating AAV cell entry, and 1×106 vg/cell for transgene expression analysis.)
Incubate the organoids on a bi-directional rocker in a 37 °C , 5% CO2 incubator.
1d
For AAV entry analysis, transfer the AAV-transduced organoids ( ) from the matrigel-coated plate into microcentrifuge tubes and wash organoids with DPBS three times.
The organoid pellet can be used for next generation sequencing (NGS) analysis or stored at -80 °C for later analysis.
For further incubation of the AAV-transduced organoids ( ), Resuspend organoids with Matrigel (30 µL per dome) and add the Matrigel droplet to the center of each well in a new flat-bottom 24-well plate.
Incubate the cells for 5–10 minutes in a 37 °C incubator until the Matrigel is solidified.
Overlay the droplet with DM (500 µL per well for a 24-well plate) and incubate for 3–10 days in
a 37 °C , 5% CO2 incubator. Change the medium every 2 days.
Analysis for cell viability
1h
Sample preparation for cell viability assay
Count and seed the cells to multi-well plates.
Note
Make sure to place a well of only media to measure the background.
Add the reagent at a final volume corresponding to 1/10 of the culture medium.
(In our case, this was 20 µL per well in a 48-well plate containing 200 µL of medium.)
Incubate culture plate in 37 °C , 5% CO2 incubator for approximately 01:00:00 .
1h
Transfer 150 µL of medium containing the reagent into each well of a 96-well plate.
Measure the absorbance at 450 nm with a microplate reader.
Enter the absorbance reading from each well in the equation below to calculate the cell survival rate.
Survival rate (%) = (Asample - Ablank)/(Anegative control - Ablank)×100
Analysis for protein expression with Flow cytometry
Sample preparation for Flow cytometry
Observe the morphology and condition of 2D cells.
Aspirate culture media and wash with 1X PBS.
Add 100 µL of 0.05% Trypsin-EDTA in the culture plate and incubate culture plate in 37 °C , 5% CO2 incubator for approximately 00:02:00 .
2m
Add 100 µL of 2% FBS in 1X PBS and detach the cells by tapping the plate with hands.
Pipette and transfer the detached cells to a 1.5 mL sterile micro-centrifuge tube.
Add 300 µL of 2% FBS in 1X PBS and strain using a 5 mL polystyrene round-bottom tube with a cell strainer cap.
Flow cytometry
Run the flow cytometry software.
Load the samples one by one.
Analyze the data.
Analysis for protein expression with immunofluorescence chemistry
4h
Sample preparation for immunofluorescence staining
Collect retinal organoid samples using a 2 mL pipette aid and transfer them to 1.5 mL sterile micro-centrifuge tube.
Note
In the case of liver organoids, follow the steps ( )
Wash the samples twice with 1X PBS.
Remove the 1X PBS and add 1 mL of 4% paraformaldehyde (PFA), incubating for 01:00:00 at Room temperature .
1h
Remove the 4% PFA and wash the samples 3 times with 1X PBS.
Incubate the samples in 15% sucrose in 1X PBS for 01:00:00 at Room temperature .
1h
Remove the 15% sucrose and add 30% sucrose in 1X PBS, incubating Overnight at 4 °C .
1h
Embed the samples in OCT compound and freeze them in a deep-freezer Overnight at -80 °C .
1h
Section the samples at a thickness of 10 µm using a cryotome and mount three sections per silane-coated slide.
Note
The slides should be stored -20 °C unless staining is performed immediately.
Immunofluorescence staining of slides
Wash the slide with tertiary distilled water to remove remaining OCT compound around the samples.
Gently remove excess moisture around the samples using Kimtech wipes.
Note
The samples must not be dried.
Create a barrier around the samples using a barrier pen.
Add blocking and permeabilization buffer consisting of 5% FBS with 0.1% Triton X-100 in 1X PBS and incubate for at least 00:30:00 at Room temperature in a humid chamber.
30m
Dilute the primary antibody with the appropriate dilution factor in 1% FBS in 1X PBS.
Aspirate the blocking and permeabilization buffer and apply the diluted primary antibody solution, incubating the slides in the humid chamber Overnight at 4 °C .
30m
Wash the slides in 0.1% PBST containing 1% FBS five times, for 00:10:00 each.
10m
Dilute the secondary antibody and Hoechst with the appropriate dilution factor in the same solution used for the primary antibody.
Gently remove excess moisture around samples and apply the diluted secondary antibody, incubating the slides in the humid chamber for 02:00:00 at Room temperature .
2h
Wash the slides in 0.1% PBST containing 1% FBS five times, for 00:10:00 each.
10m
Gently remove excess moisture around the samples.
Apply 1~2 drops of mounting medium on each sample and cover with a cover glass.
Let the slides dry in a dark place Overnight at Room temperature .
10m
TUNEL assay of slides
Follow the manual of In Situ Cell Death Detection Kit, Fluorescein from Roche.
Imaging slides
Run the imaging software.
Load the slides upside-down, ensuring cover glass faces microscope lenses.
Capture images at 10X to 40X magnification.
Protocol references
- Mun SJ, Ryu JS, Lee MO, Son YS, Oh SJ, Cho HS, et al. Generation of expandable human pluripotent stem cell-derived hepatocyte-like liver organoids. J Hepatol. 2019;71(5):970-85.
- Mun SJ, Hong Y-H, Shin Y, Lee J, Cho H-S, Kim D-S, et al. Efficient and reproducible generation of human induced pluripotent stem cell-derived expandable liver organoids for disease modeling. Scientific Reports. 2023;13(1):22935.
- Moon HR, Mun SJ, Kim TH, Kim H, Kang D, Kim S, et al. Guidelines for Manufacturing and Application of Organoids: Liver. Int J Stem Cells. 2024;17(2):120-9.
- Na HJ, Kwon JE, Kim SH, Ahn J, Kwon OS, Chung KS. Human Pluripotent Stem Cell-Derived Retinal Organoids: A Viable Platform for Investigating the Efficacy of Adeno-Associated Virus Gene Therapy. Int J Stem Cells. 2024;17(2):204-11.
Citations
Step 6.1
Na HJ, Kwon JE, Kim SH, Ahn J, Kwon OS, Chung KS. Human Pluripotent Stem Cell-Derived Retinal Organoids: A Viable Platform for Investigating the Efficacy of Adeno-Associated Virus Gene Therapy.
https://doi.org/10.15283/ijsc23071Step 7.1
Mun SJ, Ryu JS, Lee MO, Son YS, Oh SJ, Cho HS, Son MY, Kim DS, Kim SJ, Yoo HJ, Lee HJ, Kim J, Jung CR, Chung KS, Son MJ. Generation of expandable human pluripotent stem cell-derived hepatocyte-like liver organoids.
https://doi.org/10.1016/j.jhep.2019.06.030Step 7.1
Mun SJ, Hong YH, Shin Y, Lee J, Cho HS, Kim DS, Chung KS, Son MJ. Efficient and reproducible generation of human induced pluripotent stem cell-derived expandable liver organoids for disease modeling.
https://doi.org/10.1038/s41598-023-50250-w