Oct 31, 2025
Stepwise Protocol for Efficient PBMC Extraction and Immunostaining in Camel Samples
- Cinthia Mariel Olexen1,
- Ignacio Perez1,
- Gisela María Suárez Formigo1
- 1Abu Dhabi Stem Cell Center

Protocol Citation: Cinthia Mariel Olexen, Ignacio Perez, Gisela María Suárez Formigo 2025. Stepwise Protocol for Efficient PBMC Extraction and Immunostaining in Camel Samples . protocols.io https://dx.doi.org/10.17504/protocols.io.n2bvjej8wgk5/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: October 09, 2025
Last Modified: October 31, 2025
Protocol Integer ID: 229387
Keywords: Camel, Blood sample, Flow Cytometry, immunostaining in camel sample, efficient collection of camel blood, camel blood, camel sample, optimal recovery of peripheral blood mononuclear cell, reliable downstream analyses in hematology, camel, blood chemistry, peripheral blood mononuclear cell, immunostaining, immunology, immunological profile, unique hematological characteristic, stepwise protocol for efficient pbmc extraction, efficient pbmc extraction, hematology, detailed guidance on animal handling
Abstract
Camels are increasingly recognized as valuable models for biomedical research and as sources of novel therapeutic agents, yet comprehensive data on their hematological and immunological profiles remain limited. This protocol presents a standardized method for the safe and efficient collection of camel blood, taking into account their unique hematological characteristics and physiological adaptations. It provides detailed guidance on animal handling, sample collection, and centrifugation to ensure optimal recovery of peripheral blood mononuclear cells (PBMCs). The procedure also includes immunostaining of T and B cells, facilitating reliable downstream analyses in hematology, blood chemistry, and immunology, while minimizing stress to the animals and ensuring operator safety.
Attachments

Appendix 1.docx
74KB
Materials
EDTA tubes (purple top)
Gel separator tubes (yellow top)
Sterile needles and syringes or vacutainer system
Alcohol swabs
Sharps disposal container
Sample transport cooler
Benchtop centrifuge (Kubota)
Benchtop microcentrifuge
Label marker
Micropipette 10ul to 1000ul (Eppendorf)
Biosafety Cabinet (BSC)
Thermostatic bath (Branson)
Hemocytometer Neubauer-improved
Inverted microscope (Olympus)
Mr Frosty container Nalgene (at RT)
1.5-mL cryovials
Cytometer (BD)
Reagents:
70% EtOH
100% Bleach
Ficoll paque PLUS (GE Healthcare, 17-1440-03)
Ca+ and Mg+ Free PBS (Invitrogen,10010-049)
Trypan Blue 0.4% solution (PAN-Biotech, P08-34100)
Fetal Bovine Serum (FBS) (Heat Inactivated) (Gibco, 26140079)
DMSO (PAN-Biotech, P60-36750100)
Supplemented RPMI medium (sRPMI): 10% of FBS plus 1% of antibiotics.
Troubleshooting
Problem
PBMC cloud not defined
Solution
To improve recovery of peripheral blood cells from camels, check and adjust the centrifuge g-force. Use a lower speed than typically applied for other species, as camel red blood cells are larger and denser, which can cause some cells to remain in the Ficoll layer.
Safety warnings
This protocol involves handling large animals, which requires thorough knowledge of animal anatomy and adequate assistance to perform the extraction efficiently, with minimal stress to the animal, and ensuring the safety of the operator.
Ethics statement
All procedures involving camels were conducted in accordance with internationally accepted standards for animal welfare. Sample collection was performed with minimal stress to the animals and ensuring the safety of the personnel.
Before start
*This procedure requires adequate assistance and should be conducted by personnel with expertise in handling and restraining large animals to minimize stress and ensure efficiency.
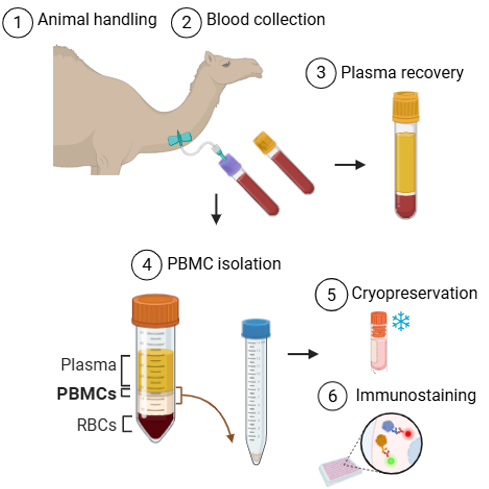
Animal handling and site of extraction
It is necessary to have a correct understanding of the anatomy of the vessels of the cervical region of camels. Two major sites are suitable for jugular venipuncture: low on the neck near the thoracic inlet and high, near the ramus of the mandible (Figure 1, Appendix).
The extraction site selected may depend on restraint facilities, assistance available, desires of the client, and experience of the operator. Both locations are used.
Blood collection
Restrain the camel safely to minimize stress and movement.
NOTE * This procedure requires adequate assistance to perform the extraction efficiently and with minimal stress to the animal.
Clean the venipuncture site with an alcohol swab and allow to dry.
Using sterile technique, insert the needle into the selected vein (commonly jugular vein).
Draw blood into the EDTA tube first (purple cap) for hematology. Fill to the indicated volume without overfilling.
Then collect blood into the gel separator tube (yellow cap) for plasma chemistry.
Remove the needle carefully and apply pressure to the puncture site until bleeding stops.
Post-Collection Handling
Gently invert the EDTA tube 8-10 times to mix anticoagulant with blood. Do not shake tubes vigorously to avoid hemolysis.
Store EDTA tubes with blood samples in an appropriate transport container at 15 - 20°C, until processing step.
For gel separator tube, allow the gel separator tube to clot by leaving it upright at room temperature for 30 minutes. Store tubes in an appropriate transport container.
Centrifuge the gel separator tube as soon as possible (e.g., 10 minutes at 1500 x g) to separate plasma.
Transfer the plasma to new and correctly identified tubes and store samples appropriately: refrigerated or frozen depending on posterior analysis.
Peripheral blood mononuclear cell isolation
PBMC isolation
NOTE * Camel erythrocytes are oval-shaped (elliptical) rather than round, allowing them to flow more easily under high viscosity conditions and to withstand dehydration. In addition, camels tend to have a higher hematocrit and elevated hemoglobin concentrations, enhancing their oxygen-carrying capacity. Due to these differences, the density and viscosity of camel blood are higher than human blood. When performing density gradient separation over Ficoll, this may result in altered layering or separation behavior, requiring adjustments in centrifugation speed or time. The interface between plasma and Ficoll may appear less defined, or cells may sediment differently than expected based on human protocols.
In biosafety cabinet, label 50-mL conical tube per specimen with ID# and gently pour whole blood from EDTA tubes. Dilute 1:1 with PBS
Collect recommended milliliters of Ficoll-Paque PLUS with 10-mL pipet according to diluted blood volume, 1 volume of ficoll: 3 volume diluted blood (1:3) in a new 50 ml conical tube.
Transfer the diluted blood slowly and carefully through the wall of the 50ml tube containing ficoll until the blood volume is completed. Both layers should not mix. The blood layer should be above the ficoll layer.
Centrifuge at 1900rpm (≈500-600g) for 20 minutes, with acceleration and brake set to 0, at RT.
NOTE * Ensure that you check the g-force and rpm for each specific centrifuge, as a lower speed is required due to the unique properties of camel blood.
After the spin completes, carefully bring conical tubes back to biosafety cabinet. The whole blood is now separated into 4 layers: the top layer consists of plasma, light yellow in color; bellow the plasma will be a small, cloudy layer of PBMC; below the clear Ficoll layer, and under the Ficoll are the RBCs.
Collect the PBMC layer with a 5-mL pipet on slow, avoiding the Ficoll layer, and transfer to a new conical containing PBS.
Centrifuge PBMC at 1800 rpm for 10 minutes with brake set to 9. Discard the used Ficoll conical tubes in the biohazard trash.
After the centrifugation, discharge the supernatant and resuspend the cell pellet in 10 mL PBS according to the volume of pellet collected and repeat the wash step two times.
After the final wash, resuspend the cells in sRPMI. Transfer 10 uL of the resuspended cells in a 1,5ml vial with 90 ul of Trypan Blue solution for counting.
Separate 2 - 5 x 106 PBMC obtained for immunostaining. (See below)
Place 10 uL of the cell/Trypan Blue mixture in one chamber of a hemacytometer. Trypan Blue stains dead cells blue. Viable cells will remain unstained. Count viable cells and calculate the total number.
Cryopreservation
After counting, wash the cells from step 22 with more supplemented RPMI medium. Centrifuge at 1800 rpm 10 min. Discard supernatant.
Resuspend pellet in cold freezing media (FBS 90% + DMSO 10%) and transfer the cells to 1 cryovial correctly labeled (specimen ID (initials/chip number). Recommended concentration: at least 5 x 106 PBMC/vial
Cap cryovials tightly, and place in a room-temperature Mr. Frosty container. Transfer Mr. Frosty container to -80°C freezer and allow cooling for at least 48 hours before transfer to nitrogen.
Immunostaining
For T and B cell staining procedure, wash the peripheral cells separated from step 22.1 with more sRPMI at 1800 rpm for 10 minutes. Discharge the supernatant.
Resuspend the cell pellet in 200ul of PBS1x and transfer to 96-well plate for staining.
Centrifuge the cells at 300g for 5 minutes. Discard the supernatant by invert the plate in a single motion.
Resuspend the cells in 200 µL of blocking/staining buffer containing 0.5% BSA, 0.01% sodium azide (NaN₃), and 1× PBS. Incubate the cells on ice for 15 minutes, then wash them twice with the same buffer, centrifugating cells 300g for 5 minutes.
Dilute the primary antibodies in blocking/staining buffer at the manufacturer’s recommended concentration and incubate for 20 minutes on ice.
Suggested immunostaining panels:
T Effector/Naïve Panel: CD4+ WC1−. Marker expression: WC1 negative, CD4 high, CD11a medium, CD44 low to high, CD62L positive, CD18 medium, CD11b positive. Where T naïve phenotype is CD4+ CD11alow CD44low, and T effector phenotype is CD4+ CD11ahigh CD44high.
B Cell Panel: GC26A+ MHCII+ CD14−. Marker expression: B cell IgM high, MHCII IgG2a high, CD14 IgG1 negative.
After incubation, complete the plate volume up to 200 µL of blocking/staining buffer, centrifugate, discharge supernatant. No extra washing step is recommended.
Resuspend the cell pellets with the secondary antibodies in adequate dilution with blocking/staining buffer and incubate 20 min on ice, protected from the light.
After incubation, complete the plate volume up to 200 µL of blocking/staining buffer, centrifugate, discharge supernatant and directly resuspend the cells in FACS.
Filter and transfer the samples to FACS tubes and run in the cytometer.
Protocol references
1- Medicine and Surgery of Camelids. 2010. Author(s): Murray E. Fowler DVM,, P. Walter Bravo. DOI:10.1002/9781118785706.
2- World Organization for Animal Health (OIE) Quality Standard and Guidelines for Veterinary Laboratories. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals
3- Hussen, J. (2018). Expression Patterns of Cell Adhesion Molecules on CD4+ T Cells and WC1+ T Cells in the Peripheral Blood of Dromedary Camels. Pakistan Veterinary Journal.
