Dec 19, 2025
Standardized Clinical Protocol for 650 nm and 810 nm Diode Laser Photobiomodulation After Dental Implant Placement
- Mohamed M. Y. Abdelsalam1,2,
- Zein Zuheir Abdussalam Adham1,3,
- Reema Saeed-ur-Rahman4,
- Ahmad A. Zaky1
- 1Department of Medical Applications of LASER, National Institute of Laser Enhanced Sciences (NILES), Cairo University, Giza, Egypt;
- 2Faculty of Dentistry, Cairo University, Giza, Egypt;
- 3Faculty of Oral and Dental Medicine and Surgery, Tripoli University, Tripoli, Libya;
- 4Gandhara University, Peshawar, Pakistan
- Zein Zuheir Abdussalam Adham: Corresponding Author;
- 650nm vs 810nm PBM Implant Study Protocol

External link: http:// https://doi.org/10.5281/zenodo.15956427
Protocol Citation: Mohamed M. Y. Abdelsalam, Zein Zuheir Abdussalam Adham, Reema Saeed-ur-Rahman, Ahmad A. Zaky 2025. Standardized Clinical Protocol for 650 nm and 810 nm Diode Laser Photobiomodulation After Dental Implant Placement. protocols.io https://dx.doi.org/10.17504/protocols.io.4r3l2zm6jl1y/v1
Manuscript citation:
Abdussalam MMY, Adham ZZA, Saeed-ur-Rahman R, Zaky AA. Analgesic outcomes of 650 nm versus 810 nm diode laser photobiomodulation after dental implant placement in a randomized controlled trial. Scientific Reports. 2025 (Accepted). https://doi.org/10.1038/s41598-025-32248-8
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: December 17, 2025
Last Modified: December 19, 2025
Protocol Integer ID: 235243
Keywords: diode laser, 650 nm, 810 nm, dental implant, postoperative pain, laser therapy, randomized controlled trial, oral surgery, OHIP-14, Photobiomodulation therapy, low-level light therapy, analgesia, sham-controlled study, near-infrared laser, red light therapy, oral health-related quality of life, numeric rating scale, pain measurement, implant dentistry, evidence-based dentistry, diode laser, 650 nm, 810 nm, dental implant, postoperative pain, laser therapy, randomized controlled trial, oral surgery, OHIP-14, Photobiomodulation therapy, low-level light therapy, analgesia, sham-controlled study, near-infrared laser, red light therapy, oral health-related quality of life, numeric rating scale, pain measurement, implant dentistry, evidence-based dentistry, diode laser photobiomodulation after dental implant placement, analgesic effects of photobiomodulation, analgesic consumption in active photobiomodulation group, diode laser photobiomodulation, following dental implant placement, dental implant placement, dental implant placement i
Disclaimer
This protocol describes the exact procedures followed in a randomized clinical trial and is intended for research and educational purposes. Clinical application should be performed only by qualified professionals and in accordance with institutional ethical approvals and local regulations.
Abstract
This protocol describes a randomized controlled clinical procedure for evaluating the analgesic effects of photobiomodulation using 650 nm and 810 nm diode lasers following dental implant placement in the posterior maxilla. The procedure standardizes surgical parameters, laser irradiation settings, treatment schedules, and outcome assessments, including postoperative pain, analgesic intake, and oral health–related quality of life.
Expected outcomes include reduced postoperative pain intensity and decreased analgesic consumption in active photobiomodulation groups compared with sham treatment. The protocol is designed to support reproducibility and transparent reporting of laser-assisted pain management following implant surgery.
Developed in accordance with SPIRIT guidelines for interventional protocols, this document underpins the randomized controlled trial “Analgesic outcomes of 650 nm versus 810 nm diode laser photobiomodulation after dental implant placement,” which has been accepted for publication in Scientific Reports. The study adheres to CONSORT recommendations and is registered at ClinicalTrials.gov (Identifier: NCT06988722). A preprint version is available on Research Square (DOI: 10.21203/rs.3.rs-6858207/v1).
Image Attribution
Image created by the authors using BioRender.com, Whimsical.com.
Guidelines
Laser Safety & Environment
Mandatory Eye Protection: All personnel (operator, assistant) and the participant must wear wavelength-specific safety goggles (OD > 5 for 650/810 nm) during all active and sham sessions to maintain safety and blinding.
Controlled Environment: Ensure the treatment room is enclosed, with reflective surfaces covered and a warning sign illuminated outside.
Surgical Standardization
Time Limit: The surgical phase (incision to suture) must be completed within 20–25 minutes. Cases exceeding this duration must be excluded to prevent surgical trauma from confounding pain outcomes.
Implant Consistency: Use only the specified implant dimensions (3.5–4.0 mm x 10 mm) and ensure placement is in healed D3 posterior maxillary bone to standardize insertion torque outcomes.
Blinding & Sham Protocol
Sham Fidelity: For the Sham group, the device must be positioned identically (gentle contact) and the "beep" sound simulator must be synchronized exactly with the 60-second "treatment" to ensure the participant cannot distinguish the group assignment.
Assessor Blinding: The outcome assessor and statistician must remain blinded to group allocation until the final database lock.
Data Collection & Pain Scoring
Pain Definition:
- Mild Pain (1–3): Noticeable but does not interfere with daily activities. 1 = Brief | 2 = Intermittent | 3 = Continuous
- Moderate Pain (4–6): Distracting; interferes with some activities but is manageable. 4 = Brief | 5 = Intermittent | 6 = Continuous
- Severe Pain (7–9): Intense; limits physical activity/functioning and requires analgesics. 7 = Brief | 8 = Intermittent | 9 = Continuous
- Unbearable Pain (10): Completely incapacitating.
Pain Scoring Logic:
Two-Step Scoring System: Participants must be trained to determine their NRS score using the following hierarchical process to ensure the "worst pain" is accurately captured:
Step 1: Identify the Worst Severity Category. Determine the highest intensity level felt since the last recording (Mild, Moderate, or Severe).
Step 2: Assign Score based on Frequency. Within that identified category, select the specific number based on how long that specific intensity lasted:
- Lower Score (1, 4, 7): Brief (transient, lasted a few minutes).
- Middle Score (2, 5, 8): Intermittent (recurring but with pain-free gaps).
- Upper Score (3, 6, 9): Continuous (persistent throughout the interval).
Scoring Example (for Participant Training):
- Scenario: A participant feels "Moderate" pain for most of the day but experiences a single "Brief" episode of "Severe" pain.
- Correct Score: (7). (Logic: The "Severe" category takes precedence as the worst pain. Since it was "Brief," the score is the lowest in the severe range: 7).
- Rescue Medication: Acetaminophen (1000 mg) is permitted only if the score reaches the Moderate range or higher (NRS > 3). Any intake must be recorded immediately.
Materials
Reagents (Drugs & Consumables)
- Local Anesthetic: 2% Lidocaine with 1:100,000 epinephrine.
- Analgesics: Acetaminophen (1000 mg tablets).
- Disinfectants: Chlorhexidine gluconate solution (0.12% or similar standard), Alcohol swabs.
- Surgical Consumables:
- 5-0 Silk sutures.
- Sterile saline solution (for irrigation).
- Sterile gauze and cotton rolls.
- Protective Gear: Sterile gloves, masks, surgical gowns, and drapes.
Equipment (Hardware & Instruments)
- Dental Implants: Two-piece dental implants, ConicalFit connection (3.5–4.0 mm diameter, 10 mm length; Nuvo Implant, Brazil).
- Surgical Kit: Surgical handpiece, physiodispenser, and burs for osteotomy preparation.
- Surgical Instruments: Sterile scalpel (blade no. 15), periosteal elevator, needle holder, tissue forceps.
- Laser Device (Group C): 810 nm Diode Laser:
- Manufacturer: (Elexxion, Germany).
- Active Medium: Gallium Aluminum Arsenide (GaAlAs).
- Probe: 0.5 cm² spot size.
- Laser Device (Group B): 650 nm Diode Laser
- Manufacturer: (Dept. of Laser Applications in Engineering, NILES, Cairo University, Egypt).
- Active Medium: Indium Gallium Phosphide (InGaP).
- Probe: 0.5 cm² spot size.
- Calibration Equipment: Laser power meter (for verifying output).
- Sham Device: Mobile phone or sound simulator (for Sham Laser Group).
- Safety Equipment: Protective eyewear (safety goggles) specific to 650 nm and 810 nm wavelengths for both operator and participant.
- Timekeeping: Digital timer.
Software & Data Tools
- Statistical Software: IBM SPSS Statistics (Version 25).
- Randomization Tool: GraphPad Online Sequence Generator.
- Data Collection:
- Google Forms (for OHIP-14 administration).
- Printed Case Report Forms (CRFs).
- Pain and Analgesic Intake Log Sheets.
- Numerical Rating Scale (NRS) charts.
Troubleshooting
Problem
Laser device does not emit light or output seems weak.
Solution
This usually indicates a power connection issue or physical damage to the fiber optic transmission system.
Check the power supply and ensure all cable connections to the main unit are secure.
Inspect the fiber optic tip for fractures, or debris, as these block light transmission.
Verify the output using a calibrated power meter before the session. If the output deviates by >10% from the set parameter (100 mW), recalibrate the device or replace the handpiece immediately.
Problem
Participant reports a "heat" or "burning" sensation during Laser PBM application.
Solution
Diode lasers can generate thermal effects if the power is set above 500mW.
Immediately pause irradiation.
Confirm the power is locked at 100 mW.
Problem
Laser device malfunction or power fluctuation during the immediate postoperative session.
Solution
PBM must be applied immediately to be effective according to protocol.
Attempt to resolve connections immediately.
If unresolved within minutes, switch to a backup calibrated device if available.
If no device is available, the session is missed. Document this as a major protocol deviation.
Problem
Excessive bleeding at the surgical site interferes with laser application.
Solution
Blood absorbs 650 nm and 810 nm wavelengths, preventing energy from reaching the target bone/tissue.
Apply sterile gauze pressure for 1–2 minutes to achieve hemostasis.
Briefly suction or wipe the area immediately before placing the probe at the crestal/buccal/palatal points.
Problem
Participant reports unusually high pain (NRS > 7) inconsistent with standard implant recovery.
Solution
This may indicate surgical infection, nerve injury, or lack of primary stability rather than just "lack of PBM efficacy."
Verify the timing and accuracy of the recording (rule out patient confusion).
Perform an immediate clinical examination to rule out infection or implant failure.
If a clinical complication is found, treat appropriately and document it as an Adverse Event.
Problem
Participant fails to record NRS scores at specific time points (e.g., 6h or 12h post-op).
Solution
During the consent process, help the participant set alarms on their phone for the 6, 12, and 24-hour marks.
If a point is missed, instruct the participant to leave it blank (do not guess retrospectively).
Document the missing data point in the CRF. Use the "Last Observation Carried Forward" or specific statistical method for missing data as defined in your analysis plan.
Problem
Participant consumes rescue medication (Acetaminophen) despite reporting low pain (NRS < 4).
Solution
Reinforce instructions during the preoperative briefing.
If it occurs, record the intake in the Analgesic Log.
Flag this participant's subsequent pain scores in the analysis, as the drug will mask the natural pain progression.
Problem
Electronic OHIP-14 form (Google Forms) fails to load or patient has no internet access.
Solution
Always keep a printed paper copy of the OHIP-14 in the patient’s file.
Administer the paper version during the follow-up visit and manually enter the data into the database later.
Problem
Sham group participant suspects they are not receiving active treatment.
Solution
Ensure the "beep" sound simulator volume matches the actual laser device volume exactly.
The operator and the participant must wear the specific safety goggles even for the Sham group to maintain the psychological environment and blinding.
Safety warnings
- Ocular Hazard: Direct or reflected laser exposure can cause permanent retinal damage. Never activate the laser without verifying that all persons in the room are wearing appropriate protective eyewear.
- Thermal Injury: Although Low-Level Laser Therapy (LLLT/PBM) is non-thermal, a dirty fiber tip or prolonged stationary application at high power can cause tissue heating. Ensure the tip is clean and power does not exceed 100 mW.
- Surgical Risks: This protocol involves invasive oral surgery. Standard risks include hemorrhage, infection, and nerve injury. Immediate clinical intervention takes precedence over protocol data collection if an adverse event occurs.
- Data Validity: Failure to record pain scores at the correct time intervals (specifically the early 2, 6, 12-hour acute phase) significantly degrades the study's power. Emphasize compliance during the "Before Start" phase.
Ethics statement
- Human Subjects: This study was conducted in strict adherence to the Declaration of Helsinki and international ethical standards for research involving human participants.
- Approval: The protocol was reviewed and approved by the Ethics Committee of the National Institute of Laser Enhanced Sciences (NILES), Cairo University (Approval No. NILES-EC-CU 23/7/18(In)).
- Consent: Written informed consent was obtained from all participants prior to enrollment, including consent for the publication of anonymized data.
- Registration: The trial is retrospectively registered at ClinicalTrials.gov (Identifier: NCT06988722).
Before start
- Ethics & Recruitment:
- Obtain written informed consent.
- Assign a unique, anonymized Study ID to the participant.
- Confirm eligibility: Age 20–65, ASA I/II, sufficient bone volume, and absence of exclusion criteria (e.g., smoking >12 cigarettes/day).
- Patient Training (Crucial Step):
- Demonstrate the use of the NRS pain scale and the analgesic log sheet before the surgery begins.
- Verify the participant has a working method to access the electronic OHIP-14 form (e.g., WhatsApp/Email on a smartphone).
- Equipment Calibration:
- Group A (Sham): Test the sound simulator volume against the active device.
- Group B (650 nm): Verify power output is stable at 100 mW.
- Group C (810 nm): Verify power output is stable at 100 mW.
- Surgical Prep:
- Prepare the surgical kit and ensure the specific implant size (ConicalFit, Nuvo) is available.
- Confirm local anesthesia (Lidocaine 2% w/ Epi) and 5-0 silk sutures are ready.
- Schedule Management:
- Map out the full treatment schedule (Days 0, 2, 4, etc.) on a calendar card for the participant to take home.
- Set automated reminders for the participant's data entry points (6h, 12h, 24h).
Participant Enrollment & Randomization
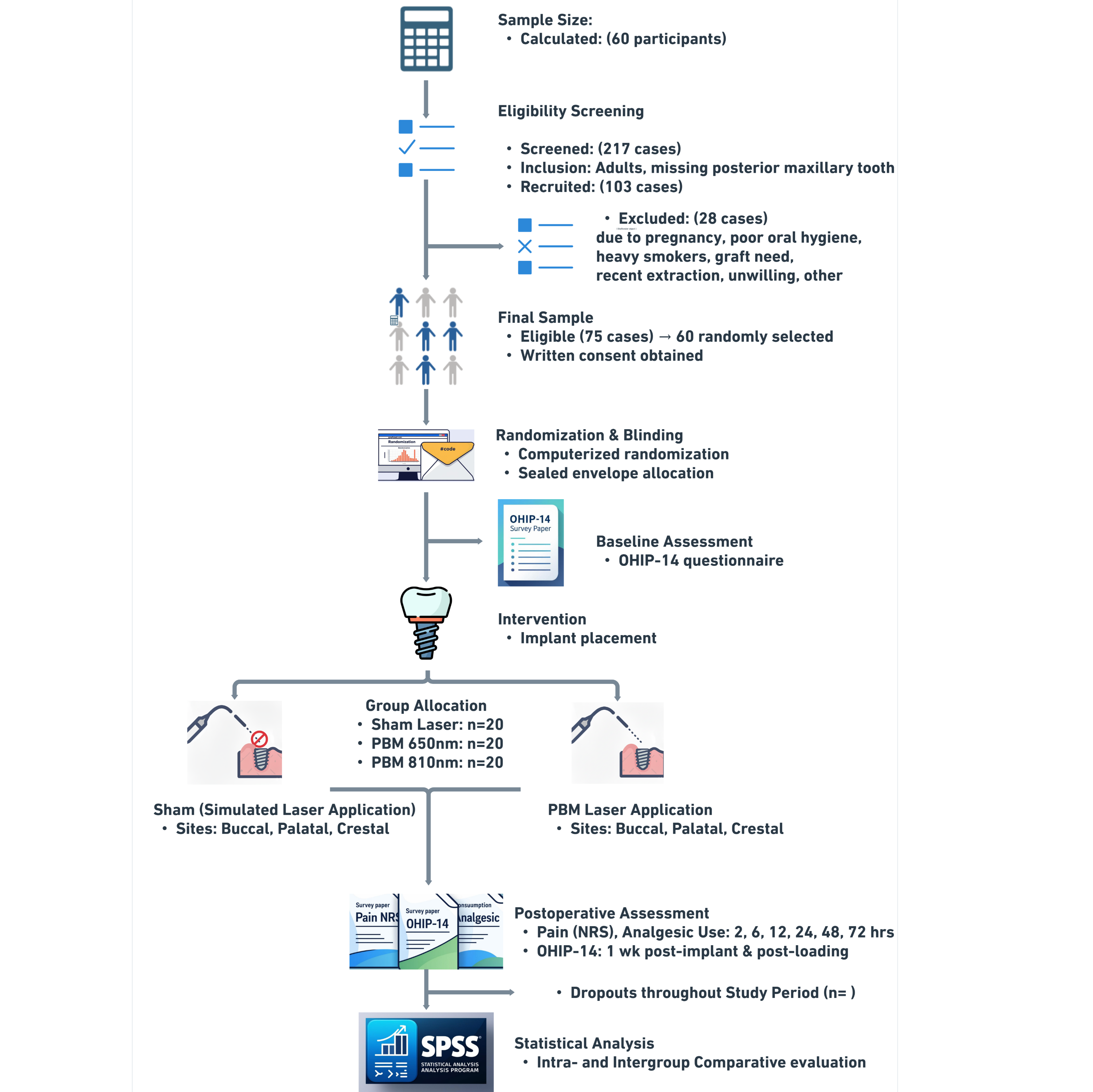
Screening: Screen patients requiring single posterior maxillary implants. Verify eligibility (Age 20–65, ASA I/II, healed site, D3 bone quality).
Consent: Obtain written informed consent.
Anonymization: assign a unique, anonymized Study ID to each participant.
Randomization: Use the computer-generated sequence to assign the participant (1:1:1) to Group A (810 nm), Group B (650 nm), or Group C (Sham). Stratify randomization by sex using a fixed block size of six.
Allocation: Prepare sequentially numbered, opaque, sealed envelopes containing group assignments by an independent research assistant. Keep the allocation concealed in an opaque envelope until the moment of laser application.
Training and baseline setting
Pain NRS record Training (Crucial): Train the participant on the Two-Step NRS Logic:
Step 1: Identify severity category (Mild 1–3, Moderate 4–6, Severe 7–9).
Step 2: Assign specific score based on frequency (Brief = lower score, Intermittent = middle score, Continuous = upper score).
Example: "Continuous Moderate Pain" = Score 6.
Baseline Assessment: Administer the baseline OHIP-14 questionnaire via the electronic link (Google Forms).
Implant Surgery Procedure
Confirm participant identity using the assigned study identification code and verify group allocation envelope remains sealed prior to surgery.
Preparation: Administer local anesthesia (2% lidocaine with 1:100,000 epinephrine).
Incision: Start the timer. Raise a minimally invasive, full-thickness H-shaped flap to preserve papillae.
Osteotomy: Prepare the implant bed according to the manufacturer’s drill sequence for Nuvo ConicalFit implants.
Placement: Insert the implant (single two-piece 3.5–4.0 mm diameter, 10 mm length) subcrestally.
Critical Check: Ensure insertion torque is ≥ 35 Ncm. If stability is not achieved, exclude the patient.
Closure: Suture with interrupted 5-0 silk sutures. Stop the timer.
Time Check: Confirm the procedure duration was 20–25 minutes. Document the exact time and designate this time point as time zero for all postoperative pain, analgesic intake, and photobiomodulation scheduling.
Photobiomodulation Intervention (650 nm, 810 nm, Sham)
Preparation: Operator, assistant, and participant must put on safety goggles (specific to the assigned wavelength, or opaque/dark for Sham blinding).
Device Setup: Group A (810 nm): Set to 100 mW, CW. Group B (650 nm): Set to 100 mW, CW. Group C (Sham): Prepare sound simulator.
Application: Place the 0.5 cm² probe in gentle contact with the tissue.
Irradiation Sequence: Treat three specific points for 60 seconds each:
Point 1: Buccal aspect of the implant.
Point 2: Palatal aspect of the implant.
Point 3: Crestal aspect (directly over the suture line).
Total Dose: Ensure total time is 180 seconds (3 minutes) and total energy is 18 J (6 J per point).
Blinding Maintenance: For the Sham group, hold the probe in the exact same positions for 60 seconds per point while playing the laser emission sound.
All PBM sessions are performed by a single trained investigator to ensure consistent probe placement, power settings, and session timing.
Record the following for each PBM session:
- Participant ID
- Session number and date
- Group allocation
- Device and wavelength used
- Time of session
- Operator initials
- Any deviations from standard procedure
Postoperative Care & Data Collection
Instruction: Instruct the patient to record NRS scores and analgesic intake at 2, 6, 12, 24, 48, and 72 hours. Remind them to take Acetaminophen (1000 mg) only if NRS > 3 (Moderate/Severe).
Follow-Up Sessions: Repeat the laser irradiation protocol (Steps 12–17) on Day 2 (48h), Day 4, Day 7, Day 9, and Day 11; for a total of 6 sessions over 2 weeks (3 sessions/week).
Suture Removal: Remove sutures on Day 7.
Final Assessment: Administer the post-implant OHIP-14 at 1 week and post-prosthesis OHIP-14 1 week after the final restoration visit.
Data Management & Statistical Analysis
Digitization: Transcribe NRS scores, analgesic logs, and OHIP-14 responses into the master database.
Verification: Check datasets for completeness. Address missing entries by consulting source documents. Document any "Last Observation Carried Forward" (LOCF) applications for missing pain scores.
Pain Analysis: Calculate daily maximum pain (0–24h, 24–48h, 48–72h). Analyze acute dynamics (2, 6, 12, 24h). Compare groups using ANOVA, or Linear Mixed Model analysis (or Kruskal–Wallis for non-parametric data).
Analgesic Analysis: Calculate the frequency of rescue medication use (Yes/No) per time point and compare using Chi-square tests.
Quality of Life Analysis: Compare mean OHIP-14 scores using paired t-tests (within groups) and ANOVA (between groups).
Reporting: Report data as Mean ± SD (or Median/IQR) with effect sizes (Cohen’s d/f). Frequencies and percentages for categorical outcomes. Significance is set at p < 0.05.
Document any deviations from the planned measurement schedule, missing data, or participant non-compliance.
Maintain logs for audit and reproducibility purposes.
Maintain a secure master dataset for record-keeping and reproducibility.
Ensure that all statistical code and procedures are documented and reproducible.
Protocol references
- Farrar, J. T., Young, J. P. Jr., LaMoreaux, L., Werth, J. L., & Poole, M. R. Clinical importance of changes in chronic pain intensity measured on an 11-point numerical pain rating scale. Pain 94, 149–158 (2001).
- Genc Sen, O., & Kaya, M. Effect of root canal disinfection with a diode laser on postoperative pain after endodontic retreatment. Photobiomodul Photomed Laser Surg 37, 85–90 (2019).
- Safdari, R., et al. The impacts of low-level laser therapy: a complementary treatment in the management of side effects after implant surgery. J. Lasers Med. Sci. 9, 207–211 (2018).
- Koparal, M., et al. Effects of low-level laser therapy following surgical extraction of the lower third molar with objective measurement of swelling using a three-dimensional system. Exp Ther Med 15, 3820–3826 (2018).
- Bourgault, P., et al. Multicomponent interdisciplinary group intervention for self-management of fibromyalgia: a mixed methods randomized controlled trial. PLoS One 10, e0126324 (2015).
- Camolesi, G. C. V., et al. Photobiomodulation in dental implant stability and post-surgical healing and inflammation: a randomized double-blind study. Clin. Oral Implant. Res. 34, 137–147 (2023).
- Othman, M. A., Zaky, A. A., Eltayeb, E. A., & Khalil, N. M. A radiographic and histological study to compare red (650 nm) versus near infrared (810 nm) diode lasers photobiomodulation for alveolar socket preservation. Sci. Rep. 14, 6871 (2024).
- Rodriguez Salazar, D. Y., et al. A systematic review of trends in photobiomodulation in dentistry between 2018 and 2022: advances and investigative agenda. F1000Res 12, 1415 (2023).
- Farazi, N., et al. Photobiomodulation combination therapy as a new insight in neurological disorders: a comprehensive systematic review. BMC Neurol 24, 101 (2024).
- AlQutub, A. W. Pain experience after dental implant placement compared to tooth extraction. Int J Dent 2021, 4134932 (2021).
- Al-Shammari, K. F., et al. Reasons for tooth extraction in Kuwait. Med Princ Pract 15, 417–422 (2006).
- Deporter, D., et al. CBCT data relevant in treatment planning for immediate maxillary molar implant placement. J Adv Periodontol Implant Dent 13, 49–55 (2021).
- Testori, T., et al. How to avoid intraoperative and postoperative complications in maxillary sinus elevation. Periodontol 2000 92, 299–328 (2023).
- Al-Khabbaz, A. K., et al. Assessment of pain associated with the surgical placement of dental implants. J. Periodontol 78, 239–246 (2007).
- Jensen, M. P., Turner, J. A., & Romano, J. M. What is the maximum number of levels needed in pain intensity measurement? Pain 58, 387–392 (1994).
- Khouly, I., et al. Post-operative pain management in dental implant surgery: a systematic review and meta-analysis of randomized clinical trials. Clin. Oral Investig. 25, 2511–2536 (2021).
Acknowledgements
We sincerely thank all participants for their contribution to this clinical trial.
We are grateful to the staff of the Department of Medical Applications of LASER, National Institute of Laser Enhanced Sciences (NILES), Cairo University, for their support in clinical operations and data collection.
We also acknowledge the technical assistance provided by the laser calibration team in the Dept. of Laser Applications in Engineering, National Institute of LASER Enhanced Sciences (NILES), Cairo University, Egypt.
