Aug 05, 2024
Sonication-induced fragmentation analysis of α-synuclein fibrils
- Arpine Sokratian1
- 1Duke Univeristy
- West lab protocols

External link: https://doi.org/10.1126/sciadv.adq3539
Protocol Citation: Arpine Sokratian 2024. Sonication-induced fragmentation analysis of α-synuclein fibrils. protocols.io https://dx.doi.org/10.17504/protocols.io.6qpvr3nebvmk/v1
Manuscript citation:
Sokratian A, Zhou Y, Tatli M, Burbidge KJ, Xu E, Viverette E, Donzelli S, Duda AM, Yuan Y, Li H, Strader S, Patel N, Shiell L, Malankhanova T, Chen O, Mazzulli JR, Perera L, Stahlberg H, Borgnia M, Bartesaghi A, Lashuel HA, West AB Mouse α-synuclein fibrils are structurally and functionally distinct from human fibrils associated with Lewy body diseases. Science Advances 10(44). doi: 10.1126/sciadv.adq3539
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: January 23, 2024
Last Modified: August 05, 2024
Protocol Integer ID: 93934
Keywords: ASAPCRN, synuclein fibril, ultrasonic frequencies through sonication, synuclein, bath sonication method, points of sonication, fibril length, distribution of ultrasonic wave, hour of sonication, ultrasonic wave, fibril, sonication, ultrasonic frequency, scattering measurement, induced fragmentation analysis
Funders Acknowledgements:
ASAP
Grant ID: Grant ID: 020527
Abstract
This protocol is designed to quantify the fragmentation pattern of full-length α-synuclein fibrils, induced by ultrasonic frequencies through sonication. To ensure consistency, we have adopted the indirect
water-bath sonication method, which offers a more controlled distribution of ultrasonic waves compared to the cup-horn technique. Our protocol involves multiple time-points of sonication, specifically at 1 minute, 2
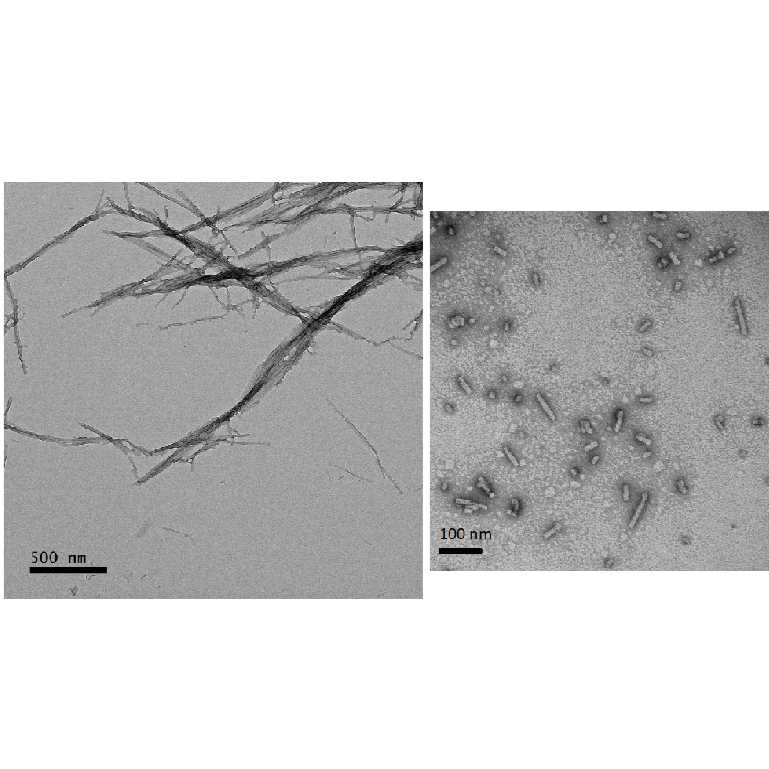
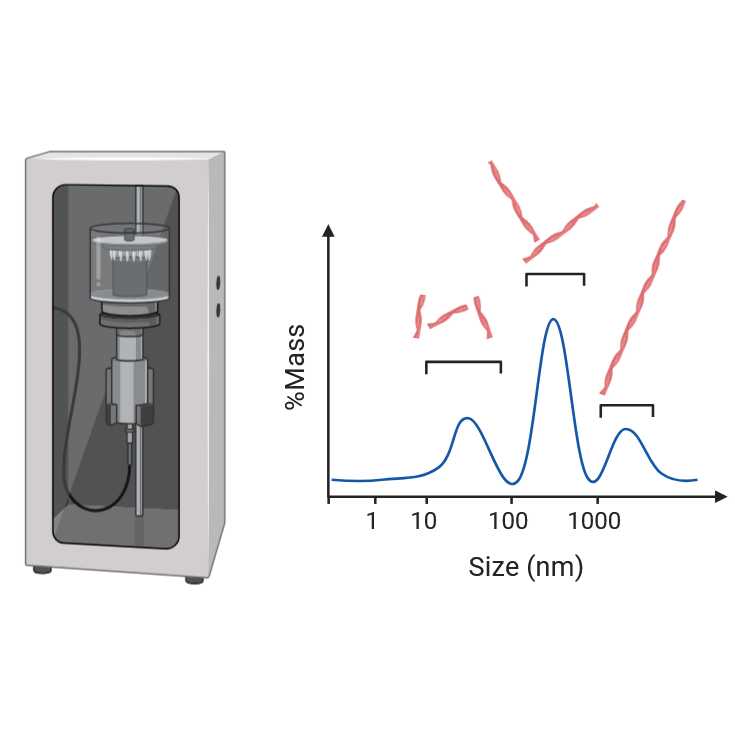
minutes, 10 minutes, 30 minutes, and 1 hour of sonication. We recommend analyzing the sonication-induced fragmentation of α-synuclein fibrils using dynamic light scattering measurements and acquisition electron
micrographs for quantification of fibril length and size distribution
Protocol materials
Thermo Scientific™ Low Protein Binding Collection Tubes (1.5 mL)Catalog #PI90411
Cell Culture Phosphate Buffered Saline (1X)CorningCatalog #MT21040CM
BrandTech™ BRAND™ Thin Wall 0.2 mL PCR TFisher ScientificCatalog #13-882-58
Safety warnings
Hazard Identification and Risk of Exposure to the Hazards:
Inhalation or spread through food or drink that contain fibrils aerosols or fibrils.
Protective gloves, safety glasses and lab coat must always be used when handling anything that possibly could contain α-synuclein fibrils. Food or drink is strictly prohibited in any environment where α-syn fibrils are used.
Routes of Transmission: Prior to assigning containment requirements, it is imperative to
understand the routes of transmission.
Some issues to address:
- What are the exposure routes/risks of most concern:
Inhalation or spread through food or drink that contain fibril aerosols or fibirls accordingly. Fibrils possibly might reach the brain regions through the olfactory epithelium; Risk of accidental needlestick/droplet splash while handling fibrils for in vitro or in vivo work.
- What are the consequences of exposure (potential illness, etc)
Fibrils may be considered as infectious material. Minimum to no hazard is expected from α-syn protein. There is no evidence that transmission of fibrils can lead to development Parkinson’s disease. However, taking into account prion-like properties of α-syn fibrils should therefore be handled cautiously and wisely. Strictly recommended using disposable materials and Personal Protective Equipment (PPE) such as gloves, face mask, etc.
PRECAUTIONS:
Laboratory work where high concentration of fibrils (more than 300 uM) is needed must comply with biosafety level 2 (BSL2) containment as described in the current edition of the CDC/NIH’s
Biosafety in the Microbiological and Biomedical Laboratories: http://www.cdc.gov/od/ohs/biosfty/bmbl5/bmbl5toc.htm
Sharps safety precautions:
The use of sharps (glass pipettes, glass slides and cover slips, scalpels and lancets) should be eliminated, when possible. Appropriate precautions should be taken to avoid percutaneous injuries. These items should be disposed of immediately in a puncture-resistant sharps container. Bending, recapping or clipping of needles is prohibited. As described in CDC’s sharps safety website: https://www.cdc.gov/sharpssafety/index.html
Procedural Methods
and Materials:
- Laboratory work where high concentration of fibrils (more than 300 uM) is needed must comply with biosafety level 2 (BSL2) containment. This means all aerosol generating procedures must be performed within the biosafety cabinet.
- All the fibrils work involves using PPE, aerosol-tight centrifuges, water bath sonicator in a closed cabinet, homogenization of frozen brain samples using probe-tip sonicator under the hood (collection of protein fractions in BSL2 cabinets), chromatography equipment in a closed-door fridge, sealed plates, safe lock microfuge tubes (or tubes wrapped/sealed with parafilm), and use of filtered tips for pipettes. All personnel must strictly adhere to these procedures.
- Use of proper PPE as stated in the section below. Use of available N95 respirators is voluntary (same for the use of available sleeve protectors). Follow safety precautions for sharps (for e.g., to avoid accidental needle sticks) while working with PFFs in the lab and for doing in vivo work.
Personal Protective
Equipment (PPE): Appropriate PPE includes gloves, lab coat and safety glasses,
face mask (voluntary N95 respirator use and sleeve protectors), face / bench
top splash shield for specific procedures as stated above.
Methods to minimize personal exposure: Strictly adhere to sharps safety precautions using needles or any material that can potentially cause wounds. Use disposable supplies where possible. Use the minimal amounts of α-fibrils needed for an experiment. Keep fibrils in closed tubes. 10% of SDS solution in water must be used for decontaminating work areas. Do not use NaOH or Sodium Hypochlorite or ethanol. Do not leave samples containing fibrils unattended at the bench.
Methods to prevent the release of fibrils/protect workers from aerosols, splashes, splatters: protective gloves and clothing always be always be worn when handling frozen vials. High concentration of fibrils(>1mg/mL) always be handled under Biosafety cabinet and containment caps will be used while centrifugation. Centrifuge cups will be opened inside a biosafety cabinet. Face shield or benchtop splash shield will be used when working at the open bench.
Specimen transport and removal of material(s) from the laboratory: Transported in secondary container (plastic/Styrofoam) in a closed box. The closed box is carried in a bag.
Standard microbiological methods: hand washing after removal of gloves and before
leaving the work area, no mouth pipetting, strictly no food or drink in refrigerators where material is stored, no eating in work area.
Cleaning & Disinfection: Work area must be cleaned with 10% SDS in water. Wipes used must be immediately disposed into biohazard waste container. Any piece of equipment or supplies that possibly
have been exposed to fibrils must be wiped with 10% of SDS.
Waste Generation and Disposal Methods: The solutions that contain α-syn fibrils must be
decontaminated with 10% of SDS in water for 30 minutes and be thrown as a biohazard waste in a sealed container/bag (use a minimal volume of fibrils needed for an experiment, do not generate large volumes of fibril-containing liquids). Use small biohazard bags to collect tips and consumables of experiment
performed, appropriately tie neck of bag in single knot and place in into secondary biohazard waste container.
Spill and Accident Response Procedure: Describe all emergency procedures including spill clean-up. Describedisinfectants and environmental decontamination. (ex., Outside of a BSC: If spill is a
respiratory hazard, evacuate 30 minutes to allow aerosols to settle. Place absorbent towels over the spill, apply freshly prepared 10% SDS solution to entire area of spill starting on the outer edges and working inward, pick up sharp items with mechanical device (not hands), place all clean-up materials in a biohazard bag)
Amplification of α-synuclein fibrils
Prepare α-synuclein fibrils according to the protocol attached
Concentration measurements
Measure concentration of fibrils according to the protocol step #5
Dilute fibrils to the concentration 5 mg/mL in PBS
Cell Culture Phosphate Buffered Saline (1X)CorningCatalog #MT21040CM
Use protein-low binding tubes
Thermo Scientific™ Low Protein Binding Collection Tubes (1.5 mL)Catalog #PI90411
Prepare 20 ul aliquots as estimated sonication time-points: three-five technical replicates for each experimental sample, three separate sonication runs. BrandTech™ BRAND™ Thin Wall 0.2 mL PCR TFisher ScientificCatalog #13-882-58
Place first round of replicates into the tube holder for the water-bath sonicator
Equipment
Cuphorn
NAME
Qsonica
BRAND
431C2
SKU
Set the amplitude to 30%, temperature to 10 °C , adjust the water level in the water tank to align the liquid level in the tubes
Set the desired time for the first time-point (for example: 2 minutes)
Collect the tubes after sonication, transfer 2 uL from each sonicated/technical repeat sample into eppendorf tube and dilute 100 times, then measure size range of fibrils using DLS
Collect 30 acquisitions with the acquisition time set for 10 seconds.
See the protocol
Continue collecting the samples from the remaining time-points then repeat with another set of replicates.
Analyze the size distribution of small fibrils (ranging from 10 to 100 nm) at multiple time points