Mar 13, 2025
Sensitive targeted analysis of salivary steroids by liquid chromatography mass spectrometry for studies of infertility
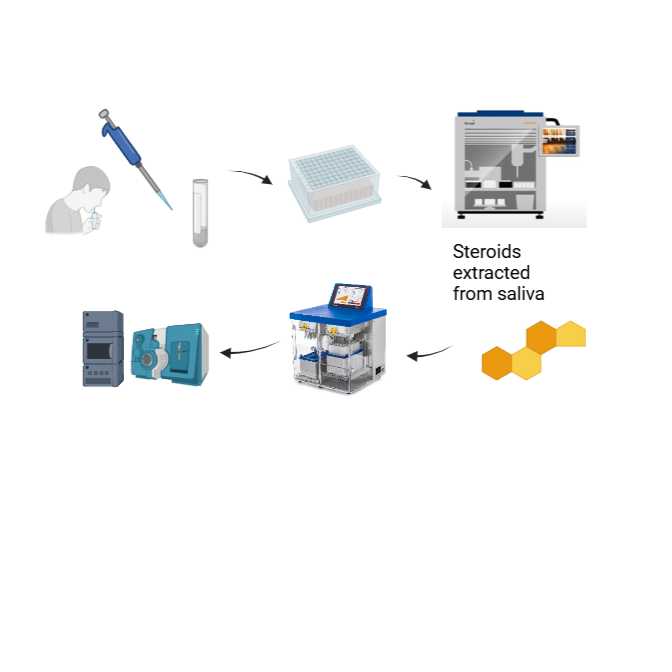
- Joanna Simpson1,
- Scott Denham1,
- Birgit Alsbjerg2,
- Natalie Z M Homer1
- 1Edinburgh Clinical Research Facility, Centre for Cardiovascular Sciences, University of Edinburgh, UK;
- 2Department of Clinical Medicine, The Fertility Clinic, Skive Regional Hospital, Aarhus, 8200, Denmark

Protocol Citation: Joanna Simpson, Scott Denham, Birgit Alsbjerg, Natalie Z M Homer 2025. Sensitive targeted analysis of salivary steroids by liquid chromatography mass spectrometry for studies of infertility . protocols.io https://dx.doi.org/10.17504/protocols.io.14egn9rzpl5d/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: February 11, 2025
Last Modified: March 13, 2025
Protocol Integer ID: 119903
Keywords: steroid profiling, saliva, LC-MS/MS, assisted reproductive technologies, progesterone, IVF, ART, measuring progesterone, assessment of progesterone level, analysis of progesterone, attractive for progesterone measurement, measurement of the salivary steroid profile, progesterone measurement, analysis of salivary steroid, salivary steroid hormone, profiles multiple salivary steroid hormone, progesterone level, low blood progesterone in the luteal phase result, salivary steroid profile, multiple salivary steroid hormone, low blood progesterone, including progesterone, progesterone, free progesterone in women, superior for steroid analysis, steroid analysis, estrone in human saliva sample, liquid chromatography mass spectrometry for study, free progesterone, liquid chromatography mass spectrometry, targeted liquid chromatography mass spectrometry, salivary steroid, liquid chromatography tandem mass spectrometry, targeted liquid chromatography tandem mass spectrometry, human saliva sample, saliva sample, mass spectr
Funders Acknowledgements:
Rosetrees Trust
Grant ID: Seedcorn2023\100344
Abstract
Progesterone plays a key role in implantation and early pregnancy (Mesen et al, 2015) and studies have shown that low blood progesterone in the luteal phase result in lower pregnancy rates and an increased pregnancy loss rate following fresh and frozen embryo transfer (Alpcetin et al, 2025). As such, infertility treatments require assessment of progesterone levels to determine success, typically measured in serum following a blood-draw.
It is important to develop treatment regimens with high reproductive outcome, that are safe and painless, and minimal time.To ease discomfort and clinic visit burden then non-invasive sampling using saliva is attractive for progesterone measurement. If progesterone levels can be measured and demonstrate a constant level during daytime this could improve patient care during infertility treatment.
Methods for measuring progesterone and sex steroids are commonly immunoassay based, which can be affected by cross-reactivity and imprecision at low levels. However, mass spectrometry based methods are superior for steroid analysis in saliva (Brouillard et al, 2025), enhanced by the ability to multiplex multi-steroid analysis without issues of sensitivity and cross-reactivity (Handelsman, 2013).
In order to assess the feasibility of using saliva to profile free progesterone in women undergoing infertility treatments then a sensitive bioanalytical method that reliably measures salivary steroid hormones is needed. We developed an automated extraction and targeted liquid chromatography tandem mass spectrometry (LC-MS/MS) method that profiles multiple salivary steroid hormones, including progesterone. This method had suitable concentration ranges that allowed us to apply to saliva samples collected over a 12 hour cycle from women during the mid-luteal phase of IVF and FET cycles.
This protocol describes the extraction and targeted liquid chromatography mass spectrometry (LC-MS/MS) analysis of progesterone, 17alpha-hydroxyprogesterone, cortisol, cortisone, aldosterone, testosterone, androstenedione, dehydroepiandrosterone, 17β-estradiol and estrone in human saliva samples from women undergoing infertility treatment. It enables measurement of the salivary steroid profile. This targeted LC-MS/MS method was developed by adapting the method from Gregory et al, 2023, using updated instrumentation set up and improved isotopically labelled internal standards.
Saliva samples (200 μL) were enriched with isotopically labelled internal standards, diluted with water (0.1% formic acid v/v) and extracted alongside a (0.0025 - 400 ng) calibration curve, by automated 96-well supported liquid extraction (SLE), using dichloromethane and isopropanol as an organic solvent, on an Extrahera automated liquid handler by Biotage (Uppsala, Sweden). Extracted steroids were separated on an Acquity I-Class UPLC by Waters (UK) with gradient elution on a Kinetex C18 column (150 x 2.1 mm; 2.6 µm) by Phenomenex (UK) and a mobile phase of methanol and water (with 0.05 mM ammonium fluoride in water and methanol). The run time was 16 minutes, followed by analysis on a QTrap 6500+ mass spectrometer operated in multiple reaction mode in both positive and negative ionisation modes, scanning for 10 steroids and appropriate internal standards.
The amount of steroid in each sample was calculated using linear regression of the peak area ratio of the analytes to the isotopically labelled internal standard as determined by analysis of a calibration curve.
Image Attribution
Guidelines
Ensure all training is up-to-date for operating the necessary laboratory instrumentation and equipment.
Materials
Consumables Table
| A | B | C | D | |
| Item | Supplier | Part no. | Quantity | |
| 1.75 mL glass vials with lids | Scientific Laboratory Supplies Ltd | TUB1200 | 10 | |
| 7 mL glass vials with lids | Scientific Laboratory Supplies Ltd | TUB1220 | 5 | |
| Isolute SLE+ 400 96 well plate | Biotage | 820-0400-P01 | 1 | |
| 96-well plate sealing film | VWR | 391-1250 | 1 | |
| Adhesive Plate Seal | Waters | 186006336 | 1 | |
| Kinetex C18 (150 x 2.1 mm; 2.6 um) | Phenomenex | 00F-4462-AN | 1 | |
| Kinetex KrudKatcher, 0.5 um | Phenomenex | AFO-8497 | 1 | |
| Deep well 96 well collection plate | Biotage | 121-5203 | 1 | |
| Deep well (2 mL) 96 well collection plate | Waters | 186002482 | 1 |
Table M1 - Consumables for extraction and liquid chromatography separation of steroids
| A | B | C | |
| Item | Supplier | Article no. | |
| Water (HPLC grade) | Fisher Scientific | C-10449380-X | |
| Acetonitrile (LC-MS grade) | VWR | 83640.320 | |
| Methanol (LC-MS grade) | VWR | 83638.320 | |
| Water (LC-MS grade) | VWR | 83645.320 | |
| Isopropanol (HPLC grade) | VWR | 20880.320 | |
| Dichloromethane (HPLC grade) | Fisher Scientific | C-23373320-X | |
| Cortisol | Sigma-Aldrich/Cerilliant | (C-106 ) 1 mg/mL in methanol (certified) | |
| Cortisone | Sigma-Aldrich/Ceriliiant | (C-130) 100 µg/mL in methanol (Certified) | |
| Aldosterone | Sigma-Aldrich/Cerilliant | A-096) 100 µg/mL in acetonitrile (certified) | |
| Androstenedione | Sigma-Aldrich/Cerilliant | (A-075) 1 mg/mL in acetonitrile (certified) | |
| Testosterone | Sigma-Aldrich/Cerilliant | (T-037) 1 mg/mL in acetonitrile (certified) | |
| Dehydroepiandrosterone | Sigma-Aldrich/Cerilliant | (D-063) 1 mg/mL in methanol (certified) | |
| 17alpha-hydroxyprogesterone | Sigma-Aldrich/Cerilliant | (17OHP) H-085 1 mg/mL in methanol (certified) | |
| Progesterone | Sigma-Aldrich/Cerilliant | (P-069) 1 mg/mL in acetonitrile (certified) | |
| 17beta-estradiol | Sigma-Aldrich/Cerilliant | (E-060) 1 mg/mL in acetonitrile (certified) | |
| Estrone | Sigma-Aldrich/Cerilliant | (E-075) 1 mg/mL in methanol (certified) | |
| 13C3-Cortisol | Sigma-Aldrich/Cerilliant | (C-216) 100 µg/mL in methanol (certified) | |
| 13C3-cortisone | Sigma-Aldrich/Cerilliant | (C-160) 100 µg/mL in methanol (certfied) | |
| 13C3-aldosterone | Sigma-Aldrich/Cerilliant | (A-120) 10 µg/mL in acetonitrile (certified) | |
| 13C3-testosterone | Sigma-Aldrich/Cerilliant | (T-070) 100 ug/mL in acetonitrile (certified) | |
| 13C3-androstenedione | Sigma-Aldrich/Cerilliant | (A-084) 100 ug/mL in acetonitrile (certified) | |
| d5-dehydroepiandrosterone | Sigma-Aldrich/Cerilliant | (D-064) 100 µg/mL in methanol | |
| d9-progesterone | Sigma-Aldrich/Cerilliant | P-070 100 ug/mL in acetonitrile | |
| d8-17hydroxyprogesterone | Sigma-Aldrich/Cerilliant | (H-096) 100 µg/mL in methanol | |
| 13C3-estradiol | Sigma-Aldrich/Cerilliant | (E-073) 100 µg/mL in acetonitrile (certified) | |
| 13C3-estrone | Sigma-Aldrich/Cerilliant | E-108) 100 µg/mL in methanol (certified) | |
| Ammonium Fluoride | Fisher Scientific |
Table M2 - Chemicals and Analytical Standards
Solutions Required
- 0.1% formic acid (aq) (200 mL) Make up to 200 mL with Water (HPLC grade). Mix thoroughly.
- 98:2 Dichloromethane:Isopropanol (1 L) - Add 20 mL Isopropanol (HPLC grade) to 980 mL Dichloromethane (HPLC grade). Mix thoroughly.
- Methanol (HPLC grade): for preparation of calibration standard/internal standard dilutions.
- Water (HPLC grade): for preparation of calibration standards.
- 70:30 Water:Methanol (100 mL) - Add 30 mL methanol (LC-MS grade) to 70 mL water (LC-MS grade). Mix thoroughly.
Equipment Table
| A | B | C | |
| Item | Model | Supplier | |
| Liquid Chromatography Pumps | I-Class UPLC | Waters | |
| mass spectrometer | QTrap 6500+ | AB Sciex | |
| Gilson Repetman | Gilson Repetman | Gilson | |
| Deepwell plate thermoshaker | TS-DW | Grant Scientific | |
| Liquid handling robot | Extrahera | Biotage, Sweden | |
| SPE Dry 96 dual evaporator | SPE Dry | Biotage, Sweden |
Table M3 - Equipment required for automated extraction and LC-MS/MS steroid analysis
Troubleshooting
Safety warnings
Ensure risk assessments are up to date and that all local laboratory guidelines are followed for handling chemicals and biological samples.
Ethics statement
Ensure all human samples used in the analysis have been collected following ethical approval.
Before start
Prepare the automated robot for operation.
Prepare the liquid chromatography tandem mass spectrometer (LC-MS/MS) for operation and ensure sufficient mobile phase solutions and needle wash. Prime the solvents. Check that the chromatographic column is correctly installed and is not leaking when the mobile phase is pumping through.
Preparation of human saliva samples for extraction
Remove human saliva samples from the freezer and defrost on ice
Preparation of calibration standard stock solutions
Prepare a mixed stock of 10 steroids (Table M2) - progesterone, 17alpha-hydroxyprogesterone, cortisol, cortisone, aldosterone, testosterone, androstenedione, dehydroepiandrosterone, 17β-estradiol and estrone - by using 100 µg/mL stock solutions. Do this by adding 50 µL x 100 µg/mL P4, 50 µL x 100 µg/mL 17OHP4, 50 µL x 100 µg/mL F, 50 µL x 100 µg/mL E, 50 µL x 100 µg/mL Aldo, 50 µL x 100 µg/mL T, 50 µL x 100 µg/mL A4, 50 µL x 100 µg/mL DHEA, 50 µL x 100 µg/mL E2 and 50 µL x 100 µg/mL E1 + 500 µL methanol to give a 5 µg/mL stock.
Dilute the 5 µg/mL stock Mixed STOCK by 1:10 dilution (100 µL x 5 µg/mL + 900 µL methanol ) to give 500 ng/mL stock
Dilute the 500 ng/mL mixed STOCK by 1:10 dilution (100 µL x 500 ng/mL + 900 µL methanol ) to give 50 ng/mL stock
Dilute the 50 ng/mL mixed STOCK by 1:10 dilution (100 µL x 5 µg/mL + 900 µL methanol ) to give 5 ng/mL stock
Dilute the 5 ng/mL Mixed STOCK by 1:10 dilution (100 µL x 5 µg/mL + 900 µL methanol ) to give 500 pg/mL stock
Dilute the 500 pg/mL Mixed STOCK by 1:10 dilution (100 µL x 5 µg/mL + 900 µL methanol ) to give 50 pg/mL stock
Preparation of internal standard solution
Prepare 100 µg/mL solutions of each isotopically labelled internal standard (Table M2) (d9-progesterone, d8-17a-hydroxyprogesterone, 13C3-cortisol, 13C3-cortisone, 13C3-aldosterone, 13C3-testosterone, 13C3-androstenedione, d5-dehydroepiandrosterone, 13C3-estrone and 3C3-estradiol) in methanol.
Prepare a mixed 5 µg/mL Internal Standard mix stock solution of the isotopically labelled steroids by adding 25 µL x 100 µg/mL d9-progesterone, 25 µL x 100 µg/mL d8-17a-hydroxyprogesterone, 25 µL x 100 µg/mL 13C3-cortisol,25 µL x 100 µg/mL 13C3-cortisone, and250 µL x 100 µg/mL13C3-aldosterone,25 µL x 100 µg/mL 13C3-testosterone 25 µL x 100 µg/mL 13C3-androstenedione,25 µL x 100 µg/mL d5-dehydroepiandrosterone, 25 µL x 100 µg/mL 13C3-estrone, and25 µL x 100 µg/mL 3C3-estradiol to25 µL methanol.
Prepare a 5 ng/mL Working Internal Standard solution by taking 10 µL x 5 µg/mL Int Std Mix + 1990 µL methanol.
Set up of supported liquid extraction of steroids from calibration standards and samples
Label a 2 mL deep well 96-well collection plate (Table M1). Label a Supported Liquid Extraction SLE400 plate with batch details. Label a 2 mL deep well 96-well collection plate (Waters).
Design and prepare batch of standards and saliva samples in Microsoft Excel template, following a column-wise plate map design as below (Table S1).
Table S1 - Plate Map - Column-wise plate layout for automated Supported Liquid Extraction on an Extrahera liquid handling robot (Biotage, Sweden)
Preparation of calibration standard curve and samples
Prepare calibration standards directly into the 96-well deep well plate using the following table for volumes of each stock concentration, into a final volume of 200 µL water.
| A | B | C | D | |
| Standard name | Amount (ng) | STD Mix Vol (uL) | Vol water (uL) | |
| 0 STD | 0 | 0 | 200 | |
| 0.00250 STD | 0.00250 | 5 uL x 500 pg/mL | 195 | |
| 0.00500 STD | 0.00500 | 10 uL x 500 pg/mL | 190 | |
| 0.01000 STD | 0.0100 | 20 uL x 500 pg/mL | 180 | |
| 0.0250 STD | 0.0250 | 5 uL x 5 ng/mL | 195 | |
| 0.0500 STD | 0.0500 | 10 uL x 5 ng/mL | 190 | |
| 0.100 STD | 0.100 | 20 uL x 5 ng/mL | 180 | |
| 0.250 STD | 0.250 | 5 uL x 50 ng/mL | 195 | |
| 0.500 STD | 0.500 | 10 uL x 50 ng/mL | 190 | |
| 1.00 STD | 1.00 | 20 uL x 50 ng/mL | 180 | |
| 2.50 STD | 2.50 | 5 uL x 500 ng/mL | 195 | |
| 5.00 STD | 5.00 | 10 uL x 500 ng/mL | 190 | |
| 10.0 STD | 10.0 | 20 uL x 500 ng/mL | 180 | |
| 25.0 STD | 25.0 | 5 uL x 5 ug/mL | 195 | |
| 50.0 STD | 50.0 | 10 uL x 5 ug/mL | 190 | |
| 100 STD | 100.0 | 20 uL x 5 ug/mL | 180 | |
| 200 STD | 250 | 5 uL x 50 ug/mL | 195 | |
| 400 STD | 400.0 | 8 uL x 50 ug/mL | 192 |
Table 2 - Calibration standard preparation table
Aliquot 200 µL saliva sample into the correct well according to the plate map design.
Supported liquid extraction of steroids from calibration standards and saliva samples
Using a multi-step pipette enrich the plate containing calibration standards with WIS by adding 20 µL x 5 ng/mL Working Internal Standard into each calibration standard, including 0 std and each sample (human saliva), except for the double blank and solvent blank.
Using the Extrahera liquid handling robot, set up with the batch labelled SLE400 extraction plate and the deep well extraction plate, containing the calibration standards and samples. Programme Extrahera to aliquot200 µL 0.1% formic acid in water (v/v) into each well of the 96-well deep well plate containing the samples and standards.
Programme the Extrahera to transfer 400 µL of liquid from each well (containing sample and the diluent, into a 400 µL volume Supported Liquid Extraction plate (SLE400), pre-placed into the deck on the Extrahera, with a deep well Waters 2 mL deep well collection plate below, pre-labelled with the batch details and date of extraction.
Allow the diluted sample to adsorb onto the SLE extraction bed for 00:05:00 before eluting with 600 µL x 98:2 (v/v) dichloromethane/isopropanol and repeating twice more, each time collecting the eluent into the collection plate
Dry down the eluent collected into the 2 mL collection plate using the SPE Dry down for 96-well plates under nitrogen.
Resuspend in 100 µL x 70:30 water/methanol, seal the plate with a zone-free plate seal and shake on ThermoShaker for 00:05:00 at 300 rpm
5m
Place the plate in the autosampler for LC-MS/MS or store at-20 °C until ready for analysis.
Steroid measurement by LC-MS/MS
Set up an acquisition batch in Analyst software using the electronic excel file of the calibration standards and sample list. Set to inject 20 µL per sample and use a method of chromatographic separation as described in steps 22 and 23 and mass spectrometer settings as outlined in steps 24 and 25.
Set up the liquid chromatography system and fit with a Phenomenex Krud Katcher and a Phenomenex 150 x 2.1 mm; 2.6 µm Kinetex C18 liquid chromatography column, using mobile phase A - water with 0.05 mM ammonium fluoride and mobile phase B - methanol with 0.05 mM ammonium fluoride at 0.3 mL/min and 50 °C diverting to the mass spectrometer at 0.2 mins and returning to waste at 15.9 mins
Set up chromatographic gradient as below (Table S3) with a run time of00:16:00 per sample
| A | B | C | D | |
| Time (min) | Flow (mL/min) | A (%) | B (%) | |
| Initial | 0.3 | 50 | 50 | |
| 4 | 0.3 | 50 | 50 | |
| 9 | 0.3 | 25 | 75 | |
| 10 | 0.3 | 0 | 100 | |
| 12 | 0.3 | 0 | 100 | |
| 12.1 | 0.3 | 50 | 50 | |
| 16.00 | 0.3 | 50 | 50 |
Table S3 - Chromatographic gradient details. A - water w/ 0.05 mM ammonium fluoride; B - methanol w/ 0.05 mM ammonium fluoride. 50oC. Kinetex C18 (150 x 2.1 mm; 2.6 µm)
Set up the mass spectrometer for Multiple Reaction Monitoring (MRM) method in positive mode, with electrospray ionisation as below, with divert of LC flow into the mass spectrometer set at 1 minute and 15.9 minutes.
| A | B | |
| Instrument | Sciex QTrap 6500+ | |
| Source, Ionisation Mode | IonDrive Turbo V Source, ESI | |
| Scan Mode, Polarity | MRM, Positive and Negative | |
| Resolution (Q1/Q3) | unit/unit | |
| Mass range | Low mass | |
| Pause Time | 5.007 ms | |
| Acquisition time | 16.0 min | |
| Delay time | 0 sec | |
| Curtain Gas (CUR) (N2) | 30 units | |
| Collision Gas (CAD) (N2) | Medium | |
| IonSpray Voltage (IS) (Positive) | 5500 V / -4500 V | |
| Temperature (TEM) | 600 °C | |
| Ion Source Gas 1 (GS1) (Air) | 40 units | |
| Ion Source Gas 2 (GS2) (Air) | 60 units | |
| Entrance Potential (EP) (Positive) | 10 V | |
| Probe position (x – axis) | 5 | |
| Probe position (y – axis) | 2 |
Table S4 - Mass Spectrometry source settings for positive and negative ion electrospray ionsiation on QTrap 6500+ mass spectrometer
Set up the mass spectrometer to monitor for the following multiple reaction monitoring (MRM) transitions for each steroid and each isotopically labelled steroid in positive and negative mode (Table S5).
| A | B | C | D | D | E | F | G | |
| Q1 Mass (Da) | Q3 Mass (Da) | Scan time (msec) | Polarity | Steroid Name | DP (V) | CE (V) | CXP (V) | |
| 363.1 | 121.2 | 10 | + | Cortisol 1 | 66 | 31 | 12 | |
| 363.1 | 91.0 | 10 | + | Cortisol 2 | 76 | 83 | 10 | |
| 361.1 | 163.1 | 10 | + | Cortisone 1 | 81 | 31 | 26 | |
| 361.1 | 77.1 | 10 | + | Cortisone 2 | 81 | 107 | 10 | |
| 289.1 | 97.0 | 10 | + | Testosterone 1 | 101 | 29 | 12 | |
| 289.1 | 109.2 | 10 | + | Testosterone 2 | 101 | 31 | 6 | |
| 287.1 | 97.0 | 10 | + | Androstenedione 1 | 61 | 27 | 14 | |
| 287.1 | 78.9 | 10 | + | Androstenedione 2 | 61 | 67 | 10 | |
| 271.1 | 235.1 | 10 | + | Dehydroepiandrosterone 1 | 106 | 17 | 12 | |
| 271.1 | 188.1 | 10 | + | Dehydroepiandrosterone 2 | 106 | 17 | 12 | |
| 315.0 | 97.1 | 10 | + | Progesterone 1 | 96 | 23 | 10 | |
| 315.0 | 109.1 | 10 | + | Progesterone 2 | 96 | 27 | 10 | |
| 333.1 | 109.1 | 10 | + | 17a-hydroxyprogesterone 1 | 66 | 31 | 12 | |
| 333.1 | 96.9 | 10 | + | 17a-hydroxyprogesterone 2 | 66 | 29 | 12 | |
| 359.1 | 188.9 | 10 | - | Aldosterone 1 | -70 | -24 | -21 | |
| 359.1 | 331 | 10 | - | Aldosterone 2 | -70 | -22 | -35 | |
| 269.1 | 144.9 | 10 | - | Estrone 1 | -150 | -48 | -15 | |
| 269.1 | 142.9 | 10 | - | Estrone 2 | -150 | -70 | -15 | |
| 271.0 | 144.9 | 10 | - | Estradiol 1 | -110 | -52 | -21 | |
| 271.0 | 182.9 | 10 | - | Estradiol 2 | -110 | -52 | -19 | |
| 292.1 | 100.0 | 10 | + | 13C3-Testosterone | 96 | 29 | 12 | |
| 290.2 | 100.1 | 10 | + | 13C3-Androstenedione | 31 | 27 | 12 | |
| 294.1 | 258.2 | 10 | + | d5-dehydroepiandrosterone | 21 | 13 | 28 | |
| 324.1 | 100.0 | 10 | + | d9-progesterone | 151 | 31 | 15 | |
| 339.2 | 96.9 | 10 | + | d8-17hydroxyprogesterone | 66 | 29 | 12 | |
| 367.2 | 121.1 | 10 | + | 13C3-cortisol | 80 | 29 | 16 | |
| 364.2 | 166.0 | 10 | + | 13C3-cortisone | 81 | 31 | 26 | |
| 362.0 | 192.0 | 10 | - | 13C3-aldosterone | -70 | -24 | -12 | |
| 272.1 | 147.8 | 10 | - | 13C3-estrone | -110 | -52 | -21 | |
| 274.0 | 147.9 | 10 | - | 13C3-estradiol | -110 | -48 | -29 |
Table S5 - Multiple reaction monitoring (MRM) settings for each steroid, including quantitative (1) and qualitative (2) ions for each steroid. DP - declustering potential, CE - collision energy, CXP - collision exit potential
Check the retention times of the steroids are as expected, as shown in the chromatogram in Table S6:
Expected result
Retention times; aldosterone at 2.6 mins, cortisol at 3.5 mins, cortisone at 2.9 mins, estrone at 7.2 mins, 17beta-estradiol at 7.0 mins, androstenedione at 6.9 mins, testosterone at 7.6 mins, dehydroepiandrosterone at 7.9 mins, 17alpha-hydroxyprogesterone at 8.0 mins and progesterone at 9 mins
Inject a mid-level standard. Check the chromatography and each steroid retention time is consistent with expected times and peak area response is as expected. Once satisfied then set the batch of samples to analyse, injecting 20 µL per sample.
Method specific data evaluation of LC-MS/MS data
Use the data analysis parameters to assess the peak area of the chromatograms for each steroid in the Steroid analytes and their assigned internal standards (Table S6)
| A | B | C | D | |
| Steroid Name | Abbreviation | Retention Time (min) | Internal Standard | |
| Progesterone | P4 | 9.0 | d9-P4 | |
| Cortisol | F | 3.5 | 13C3F | |
| Cortisone | E | 2.9 | 13C3E | |
| Androstenedione | A4 | 6.9 | 13C3A4 | |
| Testosterone | T | 7.6 | 13C3T | |
| Dehydroepiandorsterone | DHEA | 7.9 | d5-DHEA | |
| Aldosterone | Aldo | 2.6 | 13C3-Aldo | |
| Estrone | E1 | 7.2 | 13C3-E1 | |
| 17beta-estradiol | E2 | 7.0 | 13C3-E2 | |
| 17alpha-hydroxyprogesterone | 17OHP4 | 8.0 | d817OHP4 | |
| Internal Standards | ||||
| 13C3-cortisol | 13C3F | 3.5 | Int Std | |
| 13C3-cortisone | 13C3E | 2.8 | Int Std | |
| 13C3-Androstenedione | 13C3A4 | 6.9 | Int Std | |
| 13C3-Testosterone | 13C3T | 7.6 | Int Std | |
| d9-progesterone | d9P4 | 8.9 | Int Std | |
| d8-17hydroxyprogesterone | d817OHP4 | 7.9 | Int Std |
Table S6 - Method specific summary of retention time and specific internal standard of the steroids
Data Evaluation of steroid profiling LC-MS/MS data
Use MultiQuant software and Microsoft Excel to evaluate the LC-MS/MS steroid profiling data, by defining calibration standard levels, ensuring accuracy of the calibration standards and linear regression > 0.99. Use the Table above, to calculate the concentration of steroids in each sample, as detailed in the 'MultiQuant and Excel' protocol below. Remember to account for the volume of sample extracted and express as ng/mL.
Protocol

CREATED BY
Natalie ZM Homer
Protocol references
Arik Alpcetin, S. I., Ince, O., Akcay, B., Cevher Akdulum, M. F., Demirdag, E., Erdem, A., & Erdem, M. (2025). Comparison of Individualized Rescue Luteal Phase Support Strategies with Vaginal and Combined Vaginal & Subcutaneous Progesterone Administration in Artificial Frozen-Thawed Blastocyst Embryo Transfer Cycles Based on Serum Progesterone levels. Frontiers in endocrinology, 15, 1503008. https://doi.org/10.3389/fendo.2024.1503008
Brouillard, A., Davignon, L. M., Cernik, R., Giguère, C. É., Findlay, H., Juster, R. P., Lupien, S. J., & Marin, M. F. (2025). Comparing immunoassay and mass spectrometry techniques for salivary sex hormone analysis. Psychoneuroendocrinology, 174, 107379. https://doi.org/10.1016/j.psyneuen.2025.107379
Gregory, S., Denham, S. G., Lee, P., Simpson, J. P., & Homer, N. Z. M. (2023). Using LC-MS/MS to Determine Salivary Steroid Reference Intervals in a European Older Adult Population. Metabolites, 13(2), 265. https://doi.org/10.3390/metabo13020265
Handelsman, D. J., & Wartofsky, L. (2013). Requirement for mass spectrometry sex steroid assays in the Journal of Clinical Endocrinology and Metabolism. The Journal of clinical endocrinology and metabolism, 98(10), 3971–3973. https://doi.org/10.1210/jc.2013-3375
Mesen, T. B., & Young, S. L. (2015). Progesterone and the luteal phase: a requisite to reproduction. Obstetrics and gynecology clinics of North America, 42(1), 135–151. https://doi.org/10.1016/j.ogc.2014.10.003
