Mar 19, 2026
Sacral stimulation and multisensor colonic motility recording in minipigs
- Muriel Larauche1,
- Alex Baldwin2,
- Sahar Elyahoodayan2,
- Victor Pikov3,
- Xinghao Huang2,
- Mona Mohamed2,
- Hangbo Zhao2,
- Maral Mousavi2,
- Ellis Meng2,
- Million Mulugeta1
- 1University of California, Los Angeles;
- 2University of Southern California;
- 3Medipace, Inc.
- SPARCTech. support email: [email protected]

External link: https://github.com/CARSSCenter/
Protocol Citation: Muriel Larauche, Alex Baldwin, Sahar Elyahoodayan, Victor Pikov, Xinghao Huang, Mona Mohamed, Hangbo Zhao, Maral Mousavi, Ellis Meng, Million Mulugeta 2026. Sacral stimulation and multisensor colonic motility recording in minipigs. protocols.io https://dx.doi.org/10.17504/protocols.io.36wgq14pxvk5/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: January 06, 2026
Last Modified: March 19, 2026
Protocol Integer ID: 237142
Keywords: colon, manometry, strain, electromyography, sacral stimulation, neuromodulation, bioelectronic medicine, open source, minipig, motility, multisensor colonic motility recording in minipig, multisensor colonic motility recording, monitoring distal colon motility, sacral stimulation of minipig, sacral stimulation, involving sacral stimulation, distal colon motility, colon motility, stimulation at various amplitude, stimulation, s3 sacral root, electrochemical dopamine sensing, sacral root, sensing modality, intraluminal pressure manometry, including electromyography, acute experiment
Funders Acknowledgements:
NIH SPARC
Grant ID: 5U41NS129514
Abstract
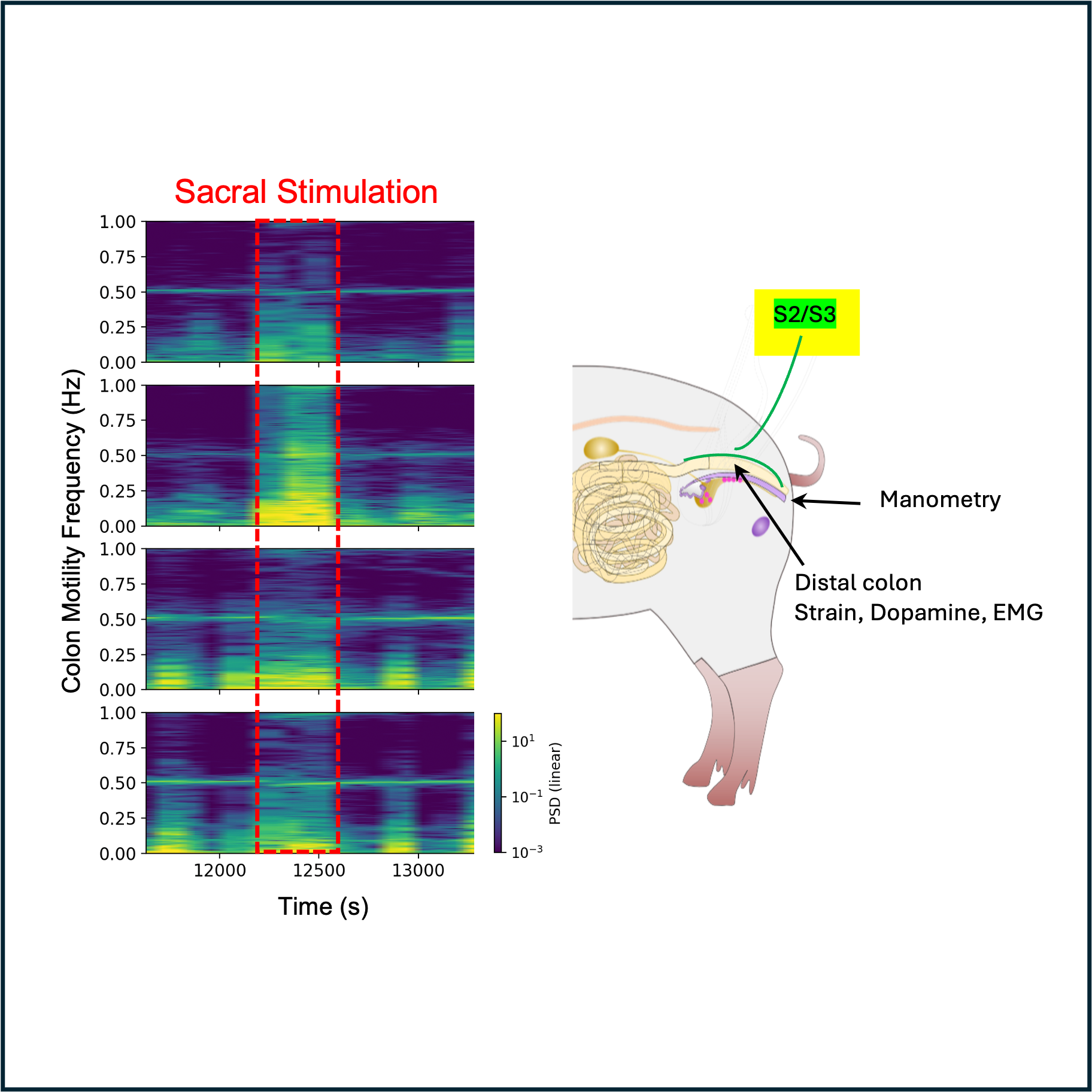
This protocol describes acute experiments involving sacral stimulation of minipigs while monitoring distal colon motility. Stimulation at various amplitudes and frequencies is applied to the S2 or S3 sacral roots using a bipolar cuff. Colon motility is monitored using multiple sensing modalities including electromyography (EMG), colon wall strain sensing, intraluminal pressure manometry, and electrochemical dopamine sensing.
Materials
Animals
Yucatan minipigs, 25-36 kg, male castrated, S&S Farms, Ramona, CA
Surgical Preparation and Device Placement
Midazolam
Ketamine
Meloxicam
Isoflourane
Lactated ringers
Heparin
Pentobarbitol
Heating pad
Animal respirator
Electrocautery
Scalpel
Freer elevator
Kerrison Rongeur
Cotton gauze
Heating pad
Sutures
Saline
Hook clips
Stimulation
Med-Ally bipolar cuff electrodes (2 contacts at 4mm spacing, 2 or 3 mm ID, 8 mm length)
Agilent U1733C Handheld LCR Meter
Anker PowerCore Slim 10000 battery pack
Strain sensor
PCap04-EVA-Kit V2.0 evaluation board, ScioSense
PCap04 Evaluation Software V2.1, ScioSense
Dopamine sensor
Sensit BT potentiostat, PalmSens
PStrace electrochemical software, PalmSens
EMG
EMG Electrodes, Med-Ally LLC
Needle electrodes
NI 9239 4-channel differential amplifier, National Instruments
NI CDAq 9184, National Instruments
Data acquisition software (custom)
Manometry
Mikro-Cath diagnostic pressure catheters, #825-0101, Millar, Houston, TX
PEC-10D pressure cables, #850-5090, Millar, Houston, TX
PCU-2000 pressure transducer, Millar, Houston, TX
Micro 1401 data acquisition system, CED, Cambridge, UK
Spike2 software, CED, Cambridge, UK
Troubleshooting
Animal preparation and anesthesia
Six-to-seven month old (25-36 kg) male Yucatan minipigs (S&S farms, Ramona, CA), castrated at 7 days of age, were used for these experiments. Animals were group housed in pens (either bedding or grate floor depending on housing availabilities - 2 pigs/pen, 42 sq. ft.) in an environmentally controlled room (lights on/off 6 AM/6 PM, 61-81 ºF) under specific-pathogen-free (SPF) conditions. All pigs were offered ad libitum access to food (5p94 Prolab mini pig diet, PMI nutrition) and filtered tap water.
Before surgery, pigs were fasted for at least 12 hours with free access to water.
To initiate anesthesia, premedicate animal with midazolam (1 mg/kg), ketamine (15 mg/kg), and meloxicam (0.3 mg/kg) injected intramuscularly.
Position animal on a heating pad (32°C). Intubate animal and connect to a respirator for ventillation, with breathing rate maintained between 13-16 breaths per minutes. Maintain general anesthesia using 1-3% inhaled isoflourane. Administer maintenance fluid (lactated ringers) at 10 ml/kg/h. Place a femoral artery line flushed with sterile saline containing 0.5% heparin.
At the completion of the experiment, euthanize animal with an intravenous injection of pentobarbital (100 mg/kg).
Spinal electrode placement
Place the animal prone, with clear access to the back.
Perform a laminectomy to access the S1-S4 sacral spinal roots, as described below and in this reference.
Before surgery, palpate the lumbar spinous processes along the midline and identified the vertebral levels of interest (T12-L1 or S1-S4).
Perform a midline incision using a scalpel/electrocautery and dissect the subcutaneous tissue and fat to gain access to the lumbosacral fascia. Cut the fascia along the midline to expose the supraspinous ligaments spanning between spinous processes.
Open the supraspinous ligaments over a few millimiters. Then using a Freer elevator, gently detatch the supraspinous ligaments from spinous processes (subperiosteal dissection). Extend the dissection to the interspinous ligaments up to the facet joints of the spinous processes along the area of interest.
After locating the lamina, open the lamina using a surgical saw or a Kerrison rongeur to extract bone in a piece-wise fashion over the whole vertebral segment of interest in order to gain access to the ligamentum flavum, periosteum, epidural fat, and dura sac. Remove the spinous processes corresponding to the region of interest, allowing access to the meninges. Gently remove the epidural fat, taking care not to damage the dura sac and allowing identification of the spinal roots.
While performing laminectomy, perform hemostatsis with bipolar electrosurgery. Do not use monopolar electrosurgery due to proximity of neural structures. Place bone wax along sites of bleeding from exposed bone and absorbable hemostatic dressing to obtain hemostatis near soft tissues. Use cotton gauze to wick fluid and blood when necessary.
Place one or more cuff electrodes around the target sacral roots. Cuffs may be placed at the S2 or S3 levels, unilaterally or bilaterally. After placement, use the thread attached to each cuff to close and tighten. A second suture may be placed around the lead for stability.
Cuff electrodes placed on the left S3 and right S2 sacral roots.
To verify electrode integrity and contact with the nerve, measure impedance using an LCR meter (Agilent U1733C). Impedance magnitude at 1 kHz should be less than 10 kOhms to indicate good contact, but should not be short circuited (<100 Ohms).
To further confirm placement, apply stimulation using the OpenNerve board. A "tail twitch" or "anal wink" reflex will likely be observed if the cuff is placed on an S2 or S3 root.
Close the incision, ensuring that electrode leads are left accessible outside the body and are properly labeled.
Sensor Placement
After closing the laminectomy, flip the animal so that it lays supine.
Perform a midline abdominal incision to gain access to the peritoneum. Identify the distal / descending colon and externalize.
Place one or more strain sensors on the exterior wall of the externalized distal colon. Strain sensors may be placed either circumferentially or longitudinally. Connect the leads of each strain sensor to the PCap04 evaluation board.
For proper operation of the strain sensor, the sensor must be pre-strained during attachment to the colon wall. To achieve pre-strain, first suture the proximal end of the strain sensor to the colon wall using the suture holes, being careful not to pierce the inner colon. Then, stretch the sensor to approximately 1.2x its resting length (20% pre-strain). With the sensor held at this strain, suture the distal end to the colon wall.
Two strain sensors placed on the distal colon. One is placed longitudinally, and the other is placed circumferentially.
Two different electrodes were used during the studies described here: EMG electrodes from Med-Ally LLC, and needle electrodes. This step places one or more pairs of EMG electrodes within the colon wall and connects them to the NI9239 differential amplifier.
Med-Ally EMG electrodes: create a pocket by injecting saline within the colon wall. Insert the suture end of the electrode into the pocket and draw it through. Pull the electrode site into the pocket until the trumpet at the end is flush with the colon wall. Tie the suture off at the other end to hold in place. If more stability is necessary, tie another suture loop around the proximal side of the trumpet. Place two electrodes approximately 1 cm apart longitudinally down the side of the colon wall.
Creating a pocket in the colon wall using a saline injection.
A single Med-Ally EMG electrode inserted into a pocket in the colon wall via the attached suture.
Needle EMG electrodes: place the needle electrodes approximately 2-3 mm into the colon wall, so that the active electrode part is in the colon wall. Place two needle electrodes 1 cm apart longitudinally.
Four needle electrodes placed in the colon wall for EMG measurement. A strain sensor is placed nearby.
Place the dopamine sensor in the colon wall. First, create a pocket by injecting saline. Then insert the sensor into the pocket with the electrodes oriented downwards towards the middle of the colon. Connect the leads to the potentiostat.
Dopamine sensor placed in a pocket on the wall of the distal colon.
Place manometry probes. Insert four MicroCath manometry probes at 3 cm spacing into the anus and advance them along until they are directly under the sensors, approximately 15 cm into the colon. were inserted in the distal colon through the anus with sensors at 10, 13, 16 and 19 cm proximal to the anal verge.
Stimulation and recording
Wait 30 minutes after handling the colon to apply stimulation, so that colon motility returns to baseline.
Record 10 minutes of data from the EMG, strain, and manometry sensors without stimulation to capture the baseline. Record the baseline measurement from the dopamine sensor.
EMG measurement is aquired using the NI9239 differential amplifier in the NI CDAq 9184 chassis, connected to a wireless router broadcasting to a Windows computer running custom acquisition software.
Strain measurements are acquired using the PCap04 EVA Kit 2.0 connected to a computer running PCap Evaluation Software V2.1.
Manometry is recorded by connecting the MicroCath catheters to PEC-10D pressure cables, then to PCU-2000 pressure transducers, then to a Micro 1401 data acquision system attached to a laptop running Spike2 software.
Dopamine measurement is accomplished by connecting the custom dopamine sensors to a Sensit BT potentiostat, which broadcasts data to a computer running PStrace software.
Apply stimulation using the OpenNerve board. To apply stimulation, the OpenNerve board was attached to a power bank and controlled via Bluetooth Low Energy connection using a custom Windows software program. Stimulation consists of square biphasic pulses with frequencies between 5 and 30 Hz, pulse widths from 200 us to 1 ms, and amplitudes from 0.5 to 5 mA. Stimulation was applied in cycles of 30s on / 90s off, to better match natural colon contraction and relaxation.
The OpenNerve stimulation board used in the study.
Protocol references
Unger, M. D., Maus, T. P., Puffer, R. C., Newman, L. K., Currier, B. L., Beutler, A. S. Laminotomy for Lumbar Dorsal Root Ganglion Access and Injection in Swine. J. Vis. Exp. (128), e56434, doi:10.3791/56434 (2017).
Huang, Xinghao, et al. "Stretchable Encapsulation for Implantable Strain Sensors." ACS Applied Materials & Interfaces (2025).
Pikov, V., PhD, Hitti, R., Baldwin, A., PhD, Elyahoodayan, S., PhD, & Meng, E., PhD. (2025). OpenNerve Implantable Pulse Generator (Version 1.0) [Computer software]. https://github.com/CARSSCenter/OpenNerve-Implantable-Pulse-Generator
Zhao, H., PhD, Li, X., PhD, & Huang, X. (2025). Mechanical biosensors for closed loop bioelectronic medicine (Version 1.0) [Computer software]. https://github.com/CARSSCenter/Mechanical-Sensors
Mousavi, M., PhD, Ali Mohamed Abdelmonem, M., PhD, Amirghassemi, F., & Soleimani, A. (2025). Sensors for chronic measurement of acetylcholine, catecholamines, and pH in vivo (Version 1.0) [Computer software].https://github.com/CARSSCenter/Chemical-Sensors
