Jun 27, 2025
RNA and gDNA Isolation from stabilized Whole Blood (Tempus Blood RNA Tube) using semi-automated Promega Chemistry
- Merz MP1,
- Treue D2,
- Hummel M2,
- Stege A2,
- ZeBanC - Zentrale Biobank Charité2
- 1Berlin Institute of Health (BIH) @ Charité;
- 2Charité Universitätsmedizin
- Merz MP: shared first author; orcid.org/0000-0003-2085-1980;
- Treue D: shared first author
- ZeBanC - Zentrale Biobank Charité: RRID:SCR_023495

Protocol Citation: Merz MP, Treue D, Hummel M, Stege A, ZeBanC - Zentrale Biobank Charité 2025. RNA and gDNA Isolation from stabilized Whole Blood (Tempus Blood RNA Tube) using semi-automated Promega Chemistry. protocols.io https://dx.doi.org/10.17504/protocols.io.5jyl8j1r7g2w/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: January 18, 2023
Last Modified: June 27, 2025
Protocol Integer ID: 75476
Keywords: Tempus Blood Tube, nucleic acid isolation, semi-automated, TEMPUS, Maxwell® RSC simplyRNA Blood, ReliaPrep™ Large Volume HT gDNA Isolation System, gdna isolation from stabilized whole blood, gdna isolation, tempus blood rna tube, rna stability, rna, gdna, stabilized whole blood, liquid handlers in the isolation process, isolation process, use of tempus tube, tempus tube, isolation
Funders Acknowledgements:
Disclaimer
All authors declare no conflicts of interest. There have not been any financial incentives for the publication of this protocol.
While we believe that, a similar worflow can be adapted for different manufacturers liquid-handlers, this protocol in its published form has only been validated on machines and chemicals as described here.
Abstract
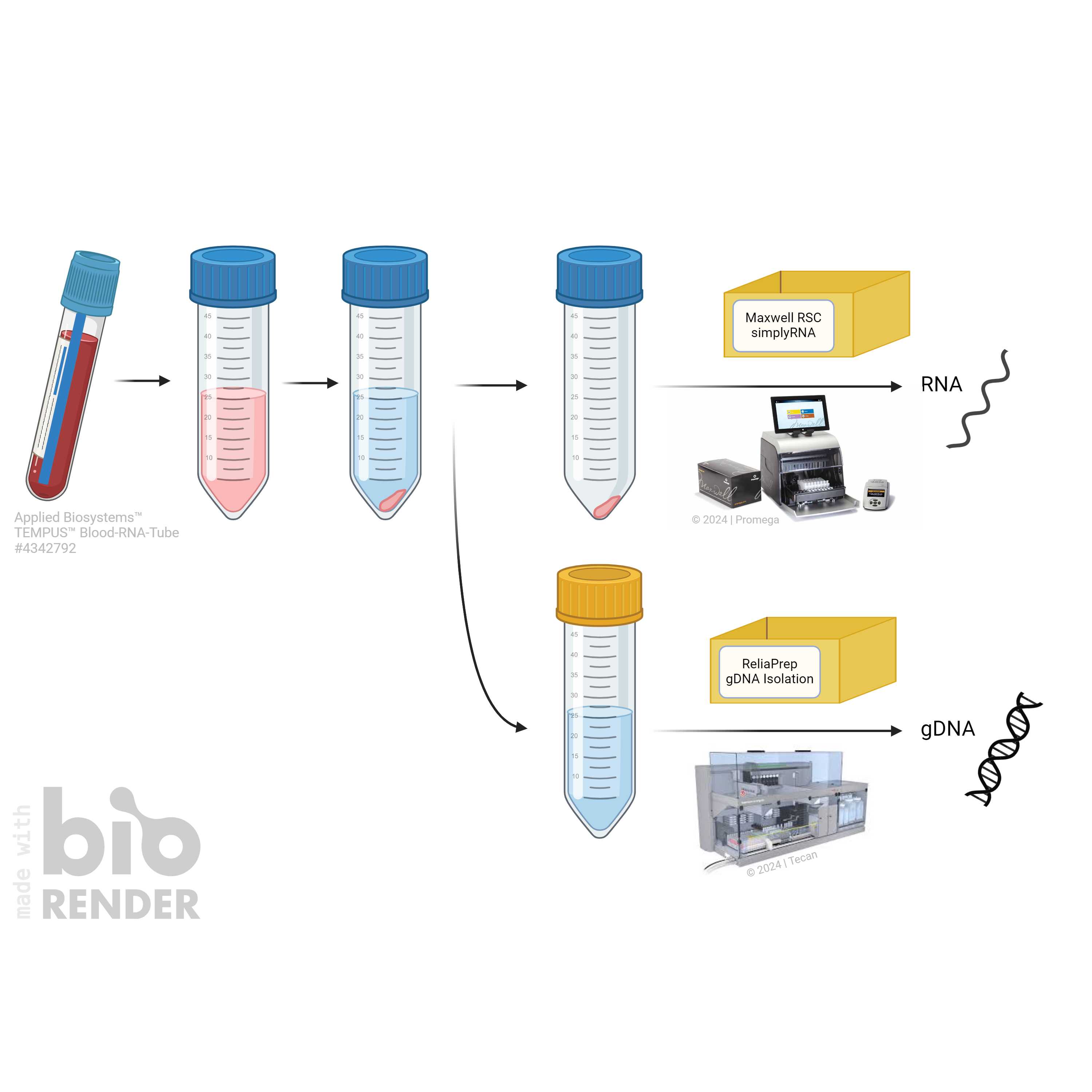
Here we describe a semi-automated way to isolate RNA and gDNA from (the same) TEMPUS Tube. This allows the user to expand the use of TEMPUS Tubes (designed for RNA stability only) as well as describing a high throughput, highly reprudicible, semi-automated way using liquid handlers in the isolation process.
Image Attribution
Merz MP, created with BioRender
Guidelines
The Central Biobank Charite (ZeBanC) works under the best practice laboratory guidelines and ISO:9001 quality standards.
This protocol uses readily available Kits (by PromegaTM) in a semi-automated fashion. While we believe that, a similar worflow can be adapted for different manufacturers liquid-handlers, this protocol in its published form has only been validated on machines and chemicals as described here.
In case of doubt, please feel free to consult attached manufacturers SOPs or reach out to us.
Materials
Maxwell® RSC simplyRNA Blood KitPromegaCatalog #AS1380 ReliaPrep™ Large Volume HT gDNA Isolation SystemPromegaCatalog #A2751 TempusTM Blood RNA Tube Thermo Fisher ScientificCatalog #4342792
Equipment
Falcon® Centrifuge Tubes
NAME
Polypropylene, Sterile, 50 mL
TYPE
Corning®
BRAND
352070
SKU
Equipment
centrifuge tube, 50mL
NAME
50 mL conical bottom tube; polypropylene
TYPE
TPP® centrifuge tubes
BRAND
TPP 91051
SKU
LINK
Equipment
Maxwell RSC instrument
NAME
Automated nucleic acid purification platform
TYPE
Promega
BRAND
AS4500
SKU
Equipment
Freedom EVO-2 HSM2 (custom)
NAME
liquid handling and robotics workstation
TYPE
TECAN (RRID:SCR_016771)
BRAND
30084252
SKU
LINK
Protocol materials
Maxwell® RSC simplyRNA Blood KitPromegaCatalog #AS1380
ReliaPrep™ Large Volume HT gDNA Isolation SystemPromegaCatalog #A2751
TempusTM Blood RNA Tube Thermo Fisher ScientificCatalog #4342792
Troubleshooting
Before start
Materials to Be Supplied by the User:
• vortex mixer
• 50ml tubes (sterile)
• centrifuge with swinging-bucket rotor
• RNase-free, sterile, aerosol-resistant pipette tips
• Maxwell‱ RSC simplyRNA Blood Kit (Cat.# AS1380)
• ReliaPrep‱ Large Volume HT gDNA Isolation System (Cat.#A2751)
Blood draw, tube handling and storage
Draw up to 3 mL of blood directly into a Tempus™ Blood RNA Tube according to your laboratory's or hospital standard. See manufacurers SOP for more information.
Note
Beware of underfilling the Tempus tube (black mark on tube). In our experience,
underfilling is the biggest contributor to a noticeable decrease of the yield and quality of RNA and gDNA.
10m
Immediately after filling the tube, shake the tube vigorously or vortex the contents for 10 sec to ensure that the stabilizing reagent is thoroughly mixed with the sample.
Note
In our experience, inverting the tube several times (comparable to other blood samling tubes) is sufficent for nucleic acid stabilization. However this is not in line with manufacturer instructions.
Filled Tempus tubes should be shipped at room temperature (18-25°C) within a maximum of 5 days. Please consult manufacturer instructions for details.
Filled Tempus can be frozen and stored at -20°C to -80°C until nucleic acid isolation.
Note
At ZeBanC, we freeze Tempus tubes at -20°C overnight and subsequently transfer them to -80°C the next day for long-term storage. This has merely practical reasons for our intenal workflow.
We have already isolated good quality RNA & gDNA from tubes stored (@ -80°C) for up to 6 years.
RNA isolation from Tempus tubes
Solution Preparation
This can be done in advance. Handle in fume hood!
Prepare (1-Thioglycerol) Homogenisation buffer (we do it for a batch of 32 samples)
12 mL Homogenisation Solution (Promega #Z305H) 240 µL 1-Thioglycerol (Promega #A208B)
Stable at 2-10°C for up to 30 days.
Prepare Rehydrate DNase-I solution (28 samples/tube) according to the manual.
Dissolve lypholysed DNase-I in 275 µL nuclease-free-water and add 5 µL Blue-Dye
Should be stored at -20°C until use. Avoid repeated thaw-freeze cycles.
15m
Thaw (32) Tempus tubes at room temperature (20-22°C). This will take approximately 30-60min.
Note
Do NOT exceed 25°C. Thawing in a water bath did not improve handling and/or yield for us.
Alternatively, samples can be thawed at 4°C over night. We have found no difference in quality or yield between when thawing at 4°C overnight (compared to 60 min @RT) and find handling quite convenient.
We handle 32 samples at a time, since this is the maximum our (semi-)automatic liquid handlers are equipped for.
1h
Label a clean 50 mL (Falcon) tube with the sample identification.
Pour the entire volume of a Tempus tube (approx. 9 mL) into a fresh 50 mL (Falcon) tube
Use 1X PBS (phosphate-buffered saline) to a total volume of 12 mL.
Note
If the initial blood sample was less than 3 mL, make up the difference by adding more PBS.
Vortex for 30-45 sec at highest power.
Make surethat the tube is securely capped. Frothing of the sample after vortexing is normal.
Centrifuge samples 4000 x g, 4°C, 01:00:00 , (+medium brake)
1h
Carefully pour the supernatant from each tube into a new 50 mL tube (keep new tube for later gDNA isolation). Do so in one movement, in order to not dislodge the RNA pellet.
Note
The pellet will be at the bottom wall of the tube and might appear as pink-ish or white-ish smudge. Sometimes the pellet can be barely visible.
Use a paper towel to take off drops from the rim of the RNA 50 mL tube.
Starting the next step with a minimal amount of supernatant is essential, therefore try to drain as much buffer as possible without over-drying or disturbing the pellet.
Note
We keep the supernatant at 4°C if gDNA isolation proceeds within 24h, or store it at -20°C for several weeks, if the gDNA isolation cannot be done the following day.
Pellet can be air-dried (lid off), if you feel uncompfortable using paper towels.
Simplyfied workflow; lid colors of tubes are only exemplary. Figure © ZeBanC, created using biorender.com
Add 300 µL Homogenisation (+THG) buffer (at RT) to every tube and resuspend pellet by vortexing (15sec).
Add 200 µL Lysis Buffer (Promega #MC501C) and 25 µL Proteinase K (PK) Solution (Promega #MC500C) per sample, vortex very shortly (approx 1 sec) and incubate at RT for 10min.
10m
Transfer complete lysate into the first well of the Maxwell Cartridge (Promega # AS138A). Load the rest of the cartridge according to the Maxwell‱ RSC simplyRNA Blood Kit (#AS1380) instructions.
Add 10 µL DNase-I solution to well number 4 (shown in yellow).
Place a 0.5 ml tube (part of Promega Kit #AS1380) in the corresponding slot on the Maxwell® RSC tray (see figure, position 2). Label this tube according to your samples. Add 65 µL nuclease-free water into the 0,5 ml tube.
Cartridge loading scheme. Figure © Promega
Load an execute the protocol for "simplyRNA Blood" on the Maxwell® RSC instrument.
Running time is approx. 45min.
Equipment
Maxwell RSC instrument
NAME
Automated nucleic acid purification platform
TYPE
Promega
BRAND
AS4500
SKU
Note
Our final RNA volume is approx 40uL and concentrations rank between 20-500 ng/uL, with an average of 130 ng/uL. In our experience, inital blood draw, underfilling and improper mixing of blood with the Tempus stabilizer have the biggest impact on nucleic acid yield
gDNA Isolation from supernatant
5h
On a TECAN Evo Freedom Instrument, run the following protocol on 8–10 ml of the supernatants from step 6.5. This protocol is fully automated (steps 7.2-7.13) and normally does not require hands-on interventions.
Other automated systems (i.e. Hamilton Microlab STAR, PerkinElmer JANUS etc) should be able to run the same or a similar protocol.
on Tecan, load the ReliaPrep HT Large Volumne gDNA Isolation System protocol. Picture © ZeBanC
ReliaPrep™ Large Volume HT gDNA Isolation System. Picture © Promega
ReliaPrep™ Large Volume HT gDNA Isolation SystemPromegaCatalog #A2751
Equipment
Freedom EVO-2 HSM2 (custom)
NAME
liquid handling and robotics workstation
TYPE
TECAN (RRID:SCR_016771)
BRAND
30084252
SKU
LINK
Note
Thaw supernatants (if frozen) at RT or at 4°C over night.
Load the 50 mL (Falcon) tubes with the supernatant into the instrument.
Load the instrument with bulk reagents (see table).
Usual bulk load (all in mL) for one run of 32 samples
In a first step the actual volume of the sample is measured in order to calculate the volumes of subsequent reagents.
Working principle of the ReliaPrep™ Large Volume HT gDNA Isolation System. Figure © Promega
Proteinase K Working Solution (0.02volumes; 60-90µL, based on the starting sample volume) is added to each tube and shaken at 500rpm, 25°C for 1 min.
Alkaline Protease (0.125volumes; 375-600µL) is added to each sample and shaken at 500rpm, 25°C for 1 min.
1volume (3-5mL) of Lysis Buffer is added to each sample.
After Lysis Buffer is added, the samples are incubated at 65°C for 30 minutes while shaking at 500rpm, followed by 10 minutes of shaking at 500rpm at 25°C.
Next, 1.2volumes (3600 - 5600µL) of Binding Buffer is added to each sample and shaken at 500rpm, 25°C for 3 min.
ReliaPrep™ Resin is thoroughly mixed and 0.1volumes (300-500µL) of resin is added to each sample. Binding of gDNA to the resin happens during incubation at 25°C for 20 minutes at 550rpm followed by magnetic capture for 20 minutes to collect the resin.
Waste from the lysis and binding is removed (by vacuum pump), then 5-7ml of Wash Buffer is added to each tube. The magnet then releases the resin into the Wash Buffer.
Samples are shaken at 500rpm, 25°C, for 2 minutes. Afterwards they are tip-mixed to thoroughly disperse the resin. Following this, the instrument mixes by shaking at 500rpm for 2 more minutes.
Waste from the first wash is removed from each tube, and 4-9ml of Wash Buffer is added (based on the original sample volume) while shaking at 500rpm for 4 more minutes.
Next, the resin is mangnetically captured for 3 minutes, the waste removed and the beads are resuspended in 4-6mL of 50% Ethanol for a last wash step. Then the instrument then shakes at 500rpm for 4 minutes and subsequently captures the resin magnetically for 3 minutes.
All waste is removed and 525uL low-TE Buffer is added to each tube. Samples are shaken at 500rpm for 3 minutes, then at 400rpm for 15 minutes at 80°C. Magnetic capture is performed for 4 minutes to deplete sample of the resin, and the eluates are transferred to 2D-barcoded tubes (SBS rack).
Note
SBS racks (for barcoded tubes) have the same layout as a 96-well PCR plate; which could also be used.
The instrument is finished.
Nucleic acid concetration is determined by either NanoDrop or Quantus measurement.
Note
Usual volume of gDNA is approx. 450-400ul and concentrations rank between 50-150ng/uL, with an average of 100ng/uL.
Protocol references
This SOP, as used in the Central Biobank Charité, was developed with the help of and using individual Kits manufactured by Promega (RRID:SCR_006724).
We are aware, that other methods (for gDNA isolation form Tempus tubes) have also been successfully established and do NOT claim novelty of this approach.
DOI: 10.1186/s13104-018-3671-4
Acknowledgements
The authors would like to thank the dedicated technical staff of the Central Biobank Charite (ZeBanC) for their shared experience and delicate insights into this protocol. The authors would also like to thank Promega (TM) Germany for their help in the establishment and troubleshooting of this protocol.

