Nov 13, 2023
Rapid Staining Technique for Visualizing Arbuscular Mycorrhizal Fungi (AMF) Colonization in Legume Roots
- Manoj-Kumar Arthikala1,
- Kalpana Nanjareddy1
- 1Ciencias Agrogenómicas, Escuela Nacional de Estudios Superiores Unidad-León, Universidad Nacional Autónoma de México (UNAM), C.P. 37689 León, Guanajuato, México.

Protocol Citation: Manoj-Kumar Arthikala, Kalpana Nanjareddy 2023. Rapid Staining Technique for Visualizing Arbuscular Mycorrhizal Fungi (AMF) Colonization in Legume Roots. protocols.io https://dx.doi.org/10.17504/protocols.io.rm7vzx1oxgx1/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: November 09, 2023
Last Modified: November 13, 2023
Protocol Integer ID: 90726
Keywords: Arbuscular Mycorrhizal Fungi, Staining Technique , Legume Roots, Rapid Protocol, rapid staining technique for visualizing arbuscular mycorrhizal fungi, visualizing arbuscular mycorrhizal fungi, arbuscular mycorrhizal fungi, accurate assessment of mycorrhizal colonization, mycorrhizal colonization, mycorrhizal structure, damage to mycorrhizal structure, mycorrhizal association, colonization in legume root, fungi, document fungal structure, fungal, legume root, fungal symbiotic interaction, stained root, better understanding of plant, meticulous staining process, rapid staining technique, diseased plant, trypan blue stain, light microscope, plant
Disclaimer
DISCLAIMER – FOR INFORMATIONAL PURPOSES ONLY; USE AT YOUR OWN RISK
The protocol content here is for informational purposes only and does not constitute legal, medical, clinical, or safety advice, or otherwise; content added to protocols.io is not peer reviewed and may not have undergone a formal approval of any kind. Information presented in this protocol should not substitute for independent professional judgment, advice, diagnosis, or treatment. Any action you take or refrain from taking using or relying upon the information presented here is strictly at your own risk. You agree that neither the Company nor any of the authors, contributors, administrators, or anyone else associated with protocols.io, can be held responsible for your use of the information contained in or linked to this protocol or any of our Sites/Apps and Services.
Abstract
This protocol outlines a rapid staining technique for visualizing Arbuscular Mycorrhizal Fungi (AMF) colonization in legume roots. Careful sample selection is emphasized, avoiding stressed or diseased plants that may compromise mycorrhizal associations. Root sampling involves gentle extraction to prevent damage to mycorrhizal structures. The FAA fixative is recommended for preserving roots before staining. Following fixation, a meticulous staining process involves successive treatments with KOH, HCl, Trypan Blue stain, and an acetic acid:glycerol:distilled water solution. Stained roots, mounted on microscopic slides with 30% glycerol, are examined under a light microscope. The protocol provides detailed steps, from sample collection to microscopic observation, allowing researchers to efficiently visualize and document fungal structures, including extra-radicle hyphae, intra-radicle structures, vesicles, and arbuscules. This technique facilitates accurate assessment of mycorrhizal colonization in legume roots, contributing to a better understanding of plant-fungal symbiotic interactions.
Materials
FAA fixative solution
Formaldehyde 1 ml
Acetic acid 1 ml
Ethanol 18 ml
-------------------------------------
Total 20 ml
5% KOH (100 ml)
5 gm of KOH pellets
dissolved in 50 ml of distilled water and make upto final volume 100 ml.
0.05% Trypan Blue stain: (100 ml)
Lactic Acid 33 ml
Glycero 33 ml
Distilled water 33 ml
Trypan Blue 0.5 g
------------------------------------
Total 100 ml
Safety warnings
Caution: When handling hot KOH solution and HCl, wear appropriate protective gear, including gloves and goggles, to prevent skin and eye contact. Work in a well-ventilated area to minimize inhalation risks, and exercise extreme care to avoid accidental splashes or spills.
Ethics statement
N/A
Before start
When conducting sample selection, it is crucial to choose healthy plants, avoiding those displaying signs of disease or stress, as such conditions can potentially compromise the quality of mycorrhizal associations. Additionally, consider the timing of tissue collection, selecting a stage of plant growth that aligns with your research objectives, as different stages may exhibit varying levels of mycorrhizal colonization. During root sampling, exercise caution to prevent damage to mycorrhizal structures. Gently dig around the plant to expose the root system, employing a shovel or hand trowel to carefully lift the roots from the soil. Following this, ensure thorough root washing by rinsing them gently with water, eliminating excess soil and debris. Use a fine-mesh sieve or a gentle stream of water to prevent any potential damage to the mycorrhizal structures.
Preserving root samples
If you're not proceeding with staining immediately, the FAA fixative is used for preserving root samples prior to mycorrhizal staining. Here's a brief protocol for using FAA fixative before mycorrhizal staining:
Prepare FAA Fixative:
Mix formaldehyde, acetic acid, and ethanol in a 1 : 1 : 18 ratio, respectively, to create the FAA fixative solution.
Store in FAA Fixative:
Submerge the cleaned roots in the FAA fixative solution. Roots can be stored for up to 4 weeks at 4 °C.
Before proceeding with the staining process, thoroughly rinse the sample 5 times with deionized water for one minute each time to eliminate any residual traces of FAA.
Staining: Step 1
Add a freshly prepared 5% (w/v) KOH solution to each tube, ensuring complete coverage of the root sample, and incubate at a temperature of 89±1°C for a duration of one hour.
Note
In the case of field-grown roots, immerse them in a 5% KOH solution and let them soak overnight at 40°C. Observe the KOH turning yellow overnight, signifying the removal of tannins from the tissue into the solution. This step can be performed either before or after the heat treatment process.
Staining: Step 2
Next, thoroughly rinse the root samples with tap water three times. Then add a 5% HCl solution, allowing it to sit for approximately 3 minutes.
Staining: Step 3
Preparation of a 0.05% Trypan Blue stain:
33 ml Lactic Acid
33 ml Glycero
33 ml Distilled water
0.5 g Trypan Blue
Heat the 0.05% Trypan Blue stain to 89 ± 1°C. Remove the HCl solution and replace it with the heated stain. Maintain incubation at 89 ± 1°C for 15 minutes.
Replace the stain with a freshly prepared solution of ´acetic acid:glycerol:distilled water (1:1:1)´ and allow the roots to soak at 40°C for 15 minutes. Repeat this procedure three times consecutively.
Note
If the roots exhibit a deep blue color, it indicates that they were not adequately cleared during the 5% KOH process, signaling the need for additional optimization.
Roots can be preserved in 30% glycerol at 4°C until they are ready for MICROSCOPIC examination.
Light Microscopy
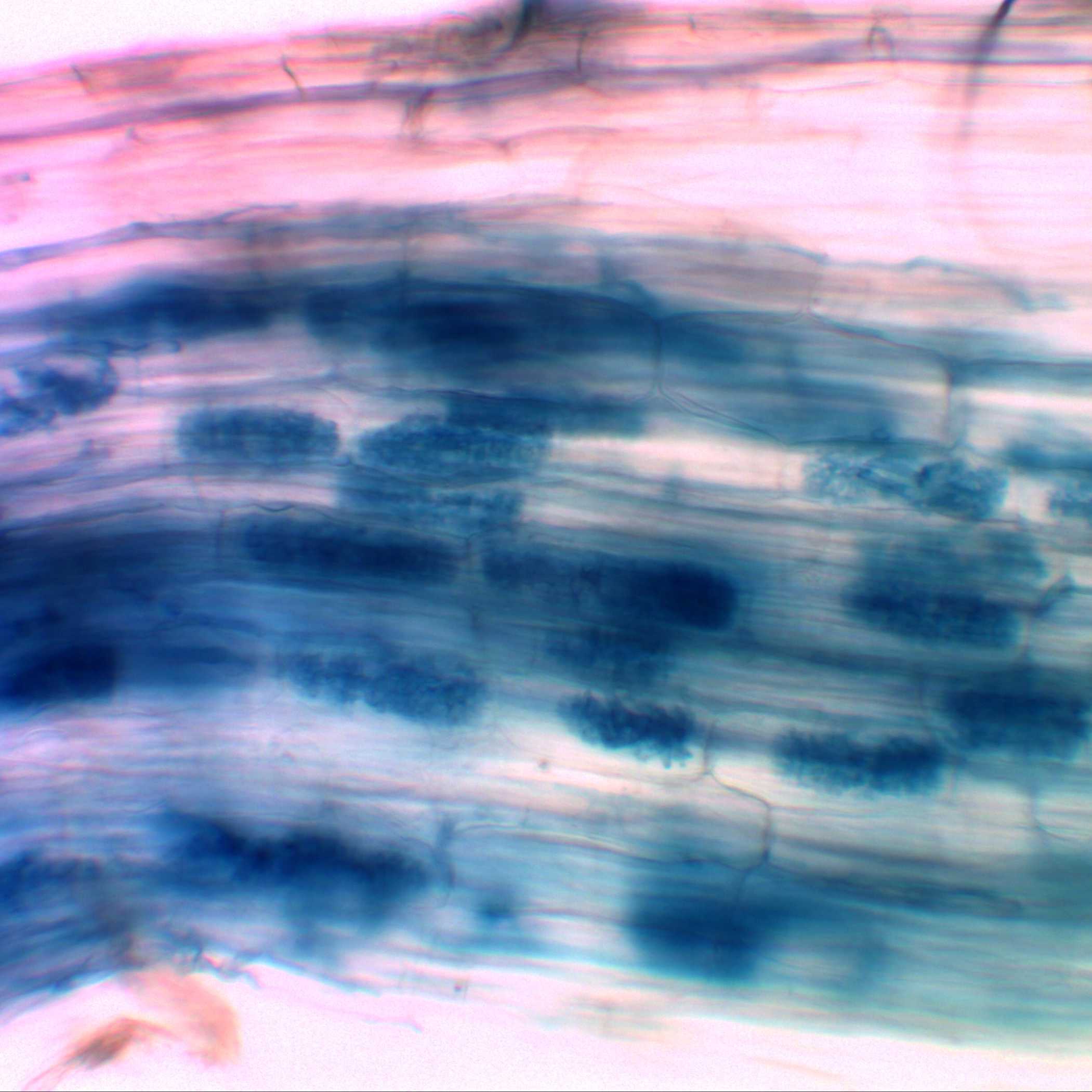
Stained roots measuring 1-5 cm are affixed to a microscopic slide using 30% glycerol. Initially, the roots are examined under a light microscope and documented. Subsequently, the tissues are gently smashed and dispersed, facilitating the observation and documentation of fungal structures, including extra-radicle hyphae (Figure 1), intra-radicle structures (Figure 2), vesicles (Figure 3), and arbuscules (Figure 4).
Results & Observations
Figure 1. Typan Blue-stained common bean roots, highlighting the presence of mycorrhizal extra-radicle hyphae.
Figure 2. Typan Blue-stained common bean roots, highlighting the presence of mycorrhizal intra-radicle hyphae.
Figure 3. Typan Blue-stained common bean roots, highlighting the presence of mycorrhizal vesicles.
Figure 4. Typan Blue-stained common bean roots, highlighting the presence of mycorrhizal mature arbuscules.
Protocol references
Manoj-Kumar Arthikala*, Kalpana Nanjareddy, Lourdes Blanco, Xochitl Alvarado Affantrange, Miguel Lara (2021). Target of
rapamycin, PvTOR, is a key regulator of arbuscule development during mycorrhizal symbiosis in Phaseolus. Scientific Reports 11: 11319.
https://doi.org/10.1038/s41598-021-90288-2
