Jan 16, 2026
QCSOP25-2: Identification of Anti-Rhesus IgA by Direct and Indirect ELISA
- Yasmine IACONE1,
- Vimala Gollamudi1,
- Diogo Magnani1
- 1NHPRR, UMass Chan Medical School, Worcester, MA (USA)
- NHPRR

External link: http://nhpreagents.org
Protocol Citation: Yasmine IACONE, Vimala Gollamudi, Diogo Magnani 2026. QCSOP25-2: Identification of Anti-Rhesus IgA by Direct and Indirect ELISA. protocols.io https://dx.doi.org/10.17504/protocols.io.3byl41m2zlo5/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: June 21, 2025
Last Modified: January 16, 2026
Protocol Integer ID: 220641
Keywords: ELISA, Anti-IgA antibody, Rhesus Macaque, Nonhuman Primate (NHP), Immunoglobulin A (IgA), Mucosal Immunity, Monoclonal antibodies, Polyclonal Antibodies, Monomeric IgA, Dimeric IgA, NHPRR, Nonhuman Primate Reagent Resource, performing antibody identity testing, antibody identity testing for research, indirect elisa this elisa, standardized elisa method, antibody reagent, antibody specificity, antibody, indirect elisa, direct elisa, antigen specificity, immunoassay, nhprr reagent, identification of anti, reliable use of nhprr reagent, polyclonal goat, elisa, nhprr laboratory staff, nonhuman primate reagent resource,
Funders Acknowledgements:
NIH
Grant ID: AI126683
Abstract
This ELISA-based protocol confirms the identity and reactivity of Nonhuman Primate Reagent Resource (NHPRR) polyclonal goat anti-rhesus IgA and monoclonal mouse anti-rhesus IgA antibody reagents. Both Indirect and Direct ELISA formats are used to evaluate antibody binding specificity and lot-to-lot consistency. The procedure involves plate coating, blocking, sample incubation, detection, and data analysis, all performed according to defined acceptance criteria.
Objective:
To provide a standardized ELISA method for verifying the antigen specificity and identity of NHPRR anti-IgA antibodies.
Scope:
Applies to NHPRR laboratory staff performing antibody identity testing for research and quality control purposes.
Background:
Accurate identification of anti-IgA antibodies ensures reliable use of NHPRR reagents in immunoassays. The Indirect ELISA detects antibody binding through a secondary conjugate, while the Direct ELISA uses labeled anti-IgA for direct detection. Together, they confirm antibody specificity and support NHPRR’s quality testing program.
Image Attribution
Created in BioRender. de Vasconcellos Castro, J. (2026) https://BioRender.com/m2svlm2
Materials
Standard ELISA materials and reagents:
| Item | Purpose | Vendor | Catalog Number | |
| TMB Substrate | Substrate for HRP | 1-Step Ultra TMB ELISA Substrate Solution, Thermo Fisher Scientific | 34029 | |
| Corning® 96-well Clear Flat Bottom Polystyrene High Bind Microplate, Nonsterile | High-binding plates for ELISA assay | Corning | 9018 | |
| Gibco™ [1X] DPBS, calcium, magnesium, glucose, pyruvate | Diluent for reagents used for coating the working plates | Gibco™ | 14-287-080 | |
| Gibco™ PBS (10X), pH 7.2 | Used in the preparation of the Washing Buffer | Gibco™ | 70-013-032 | |
| VWR® High-Performance [15 mL] Centrifuge Tubes with Flat or Plug Caps, PP | Used for the preparation/dilution of the test sample and other reagents | VWR or equivalent | 89039-668 | |
| ELISA Stop Solution | Stops the enzymatic reaction | Invitrogen™ | SS04 | |
| Non-fat Dried Milk | Used for the Blocking Buffer solution | LabScientific Biotin-free non-fat dry milk | M0841 | |
| Low-binding plates | Dilution plate | Of your choice | Of your choice | |
| 1-Step™ TMB ELISA Substrate Solutions | Acts as the substrate for the HRP enzyme | Thermo Fisher Scientific | 34029 | |
| TWEEN® 20 | Used in the preparation of the Washing Buffer | Sigma Aldrich | P6585-100ML | |
| Fisherbrand™ Hexagonal Polystyrene Weighing Dishes | Used for weighing dry milk during Blocking Buffer preparation | Fisher Scientific | 02-202-103 | |
| Timer | To keep track of incubation and developing time | of your choice | N/A | |
| Rainin Classic™ Manual Single-Channel Pipettes and Allocated Pipette Tips | Used for the preparation, dilution, and transfer of the test sample and other reagents | Rainin or equivalent | N/A | |
| Pipette Tips SR LTS 200µL F 960A/5 | Used for transferring the test sample and other reagents from the dilution plate to the working plate | Rainin | 17005859 | |
| PPE (Personal Protective Equipment) | For analyst protection: lab coat, nitrile gloves, and safety goggles | of your choice | N/A | |
| Plate Sealers | To cover ELISA plates for incubation | BioLegend | 423601 | |
| Elisa Plate Reader Software | Analyze the OD values | Molecular Devices | Model: SoftMax® Pro | |
| Nalgene™ Rapid-Flow™ Sterile Filter Storage Bottles | Used for the preparation of the Blocking Buffer | Thermo Fisher Scientific | 455-0250 | |
| Pipet-Lite™ XLS+ Manual 12-Channel Pipette, 20-200 μL | Used for transferring the test sample and other reagents from the dilution plate to the working plate | Rainin | 17013810 | |
| Sterile GRIPTIPS® INTEGRA 1250 μL Pipette Tips | Used for the preparation, dilution, and transfer of the test sample and other reagents | INTEGRA | 6544 (non-filtered) 6545 (filtered) | |
| ELISA Plate Washer | Wash ELISA plates | BioTek | Model #: 405LSRS | |
| ELISA Microplate Reader | Measure the OD values | Molecular Devices | Model: SpectraMax ABS | |
| INTEGRA VIAFLO 12-Channel 50-1250 μL Electronic Pipette | Used for the preparation, dilution, and transfer of the test sample and other reagents | INTEGRA | Model: 12-channel VIAFLO | |
| [Serological] Pipette, 10 mL and 5 mL, Graduated 1/10 mL, Sterile | Used for the preparation/dilution of the test sample and other reagents | Greiner Bio-One | 606180 (5 ml) 607107 (10 mL) | |
| Falcon® 96-well Polystyrene [Dilution] Microplates | Low-binding plates for preparation of the test sample and other reagents | Corning | 353936 | |
| Calculator | Calculate the dilution volumes | of your choice | N/A | |
| Biohazard Waste Bags | Collect used pipette tips and other waste from the experiment | of your choice | N/A | |
| Analytical Balance | For weighing dry milk (part of the Blocking Buffer preparation) | of your choice | N/A | |
| VWR® [50 mL] Disposable Pipetting Reservoirs | For holding the test sample and other reagent solutions before transfer | VWR or equivalent | 89094-684 |
Assay-Specific ELISA reagents:
Reference rhesus monomeric IgA [CC6.29]Nonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00377
Reference rhesus dimeric IgA [CC6.29]Nonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00378
Goat Anti-Rhesus IgA [GARI]Nonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00366
Anti-IgA [NHPMab001]Nonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00423
Anti-IgA [NHPMab001]-HRPNonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00392
Goat Anti-Rhesus IgA [GARI]-HRPNonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00365
Mouse Anti-Goat IgG Fc-HRP (SB115d)Southern BiotechCatalog #6158-05
Peroxidase AffiniPure® F(ab)₂ Fragment Goat Anti-Mouse IgG, Fcγ fragment specificJackson ImmunoResearch Laboratories, Inc.Catalog #115-036-071
| Antibody | Isotype | Species | Vendor | Catalog Number | Purpose | RRID | |
| Reference rhesus monomeric IgA [CC6.29] | IgA | rhesus | NHPRR | CAT-00377 | Coating | AB_3714921 | |
| Reference rhesus dimeric IgA [CC6.29] | IgA | rhesus | NHPRR | CAT-00378 | Coating | AB_3714922 | |
| Goat Anti-Rhesus IgA [GARI] | IgG | goat | NHPRR | CAT-00366 | Primary Antibody | AB_3086850 | |
| Anti-IgA [NHPMab001] | IgG | mouse | NHPRR | CAT-00423 | Primary Antibody | AB_3717991 | |
| Mouse Anti-Goat Fc-HRP (SB115d) | IgG | mouse | Southern Biotech | 6158-05 | Secondary Antibody | AB_2796222 | |
| Peroxidase AffiniPure® F(ab')₂ Fragment Goat Anti-Mouse IgG, Fcγ fragment specific | IgG | goat | Jackson ImmunoResearch | 115-036-071 | Secondary Antibody | AB_2338524 | |
| Goat Anti-Rhesus IgA [GARI]-HRP | IgG | goat | NHPRR | CAT-00365 | Primary Antibody already conjugated | AB_3086849 | |
| Anti-IgA [NHPMab001]-HRP | IgG | mouse | NHPRR | CAT-00392 | Primary Antibody already conjugated | AB_3105907 |
Protocol materials
Mouse Anti-Goat IgG Fc-HRP (SB115d)Southern BiotechCatalog #6158-05
Peroxidase AffiniPure® F(ab)₂ Fragment Goat Anti-Mouse IgG, Fcγ fragment specificJackson ImmunoResearch Laboratories, Inc.Catalog #115-036-071
Reference rhesus dimeric IgA [CC6.29]Nonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00378
Anti-IgA [NHPMab001]Nonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00423
Goat Anti-Rhesus IgA [GARI]-HRPNonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00365
Reference rhesus monomeric IgA [CC6.29]Nonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00377
Goat Anti-Rhesus IgA [GARI]Nonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00366
Anti-IgA [NHPMab001]-HRPNonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00392
Troubleshooting
Problem
Blank Wells Show High Signal
Solution
The issue may be caused by incomplete washing, insufficient blocking, incorrect buffer volume, contamination of the washing buffer, or an inadequate number of wash cycles. To address this, repeat the assay using freshly prepared blocking reagent and ensure reagent reservoirs are clean or replaced. Confirm that all wash steps are performed using the correct buffer and that full plate coverage is achieved during the blocking step. Use new pipette tips for all reagent transfers to prevent cross-contamination.
Problem
All Wells Show Low Signal
Solution
The issue may be due to inactive streptavidin-HRP or TMB substrate, incorrect incubation temperature, or systematic pipetting errors. To address this, verify reagent activity by testing streptavidin-HRP and TMB substrate using a known positive control. Additionally, confirm that all incubation steps were performed at room temperature or in accordance with SOP requirements. Inspect the performance of the plate reader to ensure proper function. If necessary, repeat the assay using a new high-binding ELISA plate and freshly prepared signal amplification reagents from different lots or new aliquots.
Problem
High Variability Between Duplicates
Solution
The issue may be caused by inconsistent pipetting, incomplete mixing of reagents, or non-uniform loading of samples and reagents. To address this, use calibrated pipettes and maintain a consistent pipetting technique; the use of a multichannel pipette is recommended for uniform application. Additionally, ensure that all samples and reagents are thoroughly mixed before use.
Before start
Note on Concentration Units: All antigen and antibody dilutions in this protocol are prepared using molar concentrations (e.g., nM) to ensure accurate comparison across immunoglobulin types with different molecular weights. When applicable, molecular weight values are used to convert between ng/mL and nM to reflect molar equivalency (e.g., IgG and monomeric IgA ~150 kDa, dimeric IgA ~300 kDa, IgM ~950 kDa).
Concentrations are lot-dependent; always make sure to check the lot even if the catalog number is the same.
Example conversion: To convert 1 mg/mL of monomeric IgA (~150 kDa) to molarity (mM), ensure that the units are consistent: mass in mg/mL and molecular weight in kDa. If using µg/mL, your result will be in µM instead of mM.
(1 mg/mL)/(150 kDa) = 0.00667 mM = 6.67 µM
Alternatively, [(1 mg/mL)/(150 kDa)] x 1000 = 6.67 µM
Prepare all the calculations beforehand.
Definitions and Acronyms
Definitions
Analyte / Antigen (recombinant rhesus IgA, monomeric and dimeric)
Recombinant rhesus IgA, present in either monomeric or dimeric form, is coated onto the ELISA plate and serves as the target analyte. Anti-rhesus IgA antibodies in the sample bind specifically to the immobilized IgA. The assay signal generated reflects the extent of antibody binding to the coated antigen.
Test Articles (TAs), TA-pAb, and TA-mAb
Test Articles (TAs): Anti-rhesus IgA antibody reagents that are being evaluated for identity and specificity.
TA-pAb: Goat anti-rhesus IgA polyclonal antibody Test Article.
TA-mAb: Mouse anti-rhesus IgA monoclonal antibody Test Article.
These reagents are tested in parallel on plates coated with monomeric and dimeric IgA to assess binding profiles and confirm identity.
Reference Control (Positive Control) Antibody
A previously qualified anti-rhesus IgA antibody lot which is included on each plate (typically as a serial dilution/titration curve) to confirm that the assay produces the expected binding behavior (signal range and curve shape) and to support run-to-run trending and lot-to-lot comparability of reagents. (Abcam, 2024). In this ELISA, the positive control is an anti-rhesus IgA antibody sample that is known to specifically bind both monomeric and dimeric recombinant rhesus IgA. Its binding curve serves as a benchmark for the expected signal and plate validity.
Note
If the positive control fails to generate the expected signal, the assay may be invalid, and the run should be repeated (BosterBio, n.d.).
Negative Control Antibody
An antibody of the same species, isotype, and format as the Test Articles (TAs), but known not to bind rhesus IgA. The negative control antibody should produce only baseline signal. It is used to verify assay specificity and to help identify non-specific binding or background issues.
Blank
A buffer-only well that is used to establish the background signal and verify the absence of non-specific binding. Unexpected signal indicates contamination or assay error.
Note
If a blank well exhibits an unexpected signal, this may indicate contamination or non-specific binding, and the assay should be considered invalid (Jackson ImmunoResearch, n.d.).
Monoclonal Antibody (mAb)
An antibody preparation derived from a single B-cell clone, recognizing one specific epitope on an antigen and therefore having a single binding specificity. In this SOP, the monoclonal antibody Test Article (TA-mAb) is a mouse anti-rhesus IgA antibody evaluated for its binding to recombinant monomeric and dimeric rhesus IgA.
Polyclonal Antibody (pAb)
An antibody preparation containing a mixture of immunoglobulin molecules produced by multiple B-cell clones, recognizing multiple epitopes on the same antigen. In this SOP, the polyclonal antibody Test Article (TA-pAb) is a goat anti-rhesus IgA antibody evaluated for its binding profile to recombinant monomeric and dimeric rhesus IgA.
Monomeric IgA
A single IgA immunoglobulin unit composed of two heavy and two light chains (one “Y-shaped” IgA molecule). In this SOP, recombinant monomeric rhesus IgA is used as a coated antigen on ELISA plates to assess antibody binding to the monomeric form of IgA.
Dimeric IgA
A form of IgA composed of two IgA monomers covalently linked, typically via a joining (J) chain, representing the main configuration of secretory IgA at mucosal surfaces. In this SOP, recombinant dimeric rhesus IgA is used as a coated antigen on ELISA plates to assess antibody binding to the dimeric form of IgA.
Direct ELISA
A plate-based immunoassay format in which the antigen is immobilized on the plate and detected using a single enzyme-labeled (e.g. HRP-conjugated) primary antibody that binds directly to the antigen. Signal is generated when the enzyme acts on a chromogenic substrate (e.g. TMB), and the measured absorbance is proportional to the amount of antigen–antibody complex formed. Direct ELISA uses fewer steps and reagents than indirect ELISA but offers less signal amplification because only one labeled antibody binds per antigen site. (ELISA Guide; Part 1: Introduction to ELISA, Formats and Signal Amplification, 2023; NCBI, 2020)
Indirect ELISA
A plate-based immunoassay format in which the antigen is immobilized on the plate, followed by incubation with an unlabeled primary antibody specific for the antigen, and then a labeled secondary antibody (e.g. HRP-conjugated anti-species antibody) that recognizes the primary antibody. The enzyme on the secondary antibody converts a chromogenic substrate (e.g. TMB) to a colored product measured by absorbance. Indirect ELISA provides signal amplification because multiple secondary antibodies can bind each primary antibody, increasing assay sensitivity compared with direct ELISA. (ELISA Guide; Part 1: Introduction to ELISA, Formats and Signal Amplification, 2023; NCBI, 2020)
Dilution Plate
A low-binding microplate used to prepare antibody dilutions and perform vertical serial dilutions of TAs, controls, and HRP-conjugated antibodies before transfer to the working plate. The dilution plate mirrors the layout of the working plate but is not read in the plate reader.
Working Plate
The high-binding ELISA plate on which antigen coating, blocking, antibody incubation, washing, substrate addition, and OD reading at 450 nm are performed. The working plate contains the bound analyte and is the plate that generates the analytical signal.
Blocking Buffer
In this SOP, the blocking buffer is 5% non-fat dry milk in PBS with 0.05% Tween-20. It is applied after the coating step to occupy unbound sites on the plate surface and is also used as a diluent for antibodies. Blocking buffer minimizes non-specific binding and reduces background signal. (Excedr, 2023)
Washing Buffer
PBS containing 0.05% Tween-20 (1× DPBS or PBS base, as prepared under “Wash Buffer Preparation”). It is used between assay steps to remove unbound reagents and reduce non-specific background.
Detection Reagent (HRP-conjugated antibody)
For the indirect ELISA, the detection reagents are HRP-conjugated secondary antibodies (mouse anti-goat HRP and goat anti-mouse HRP) that bind the species-specific primary anti-IgA antibodies. For the direct ELISA, the detection reagents are HRP-conjugated anti-rhesus IgA antibodies themselves. In both formats, the detection reagent binds to the antibody–antigen complex and enables signal generation via HRP-mediated TMB conversion. (ELISA Guide; Part 1: Introduction to ELISA, Formats and Signal Amplification, 2023; NCBI, 2020)
TMB Substrate and Stop Solution
TMB (3,3′,5,5′-Tetramethylbenzidine) is a chromogenic substrate that is oxidized by HRP, producing a blue color proportional to bound HRP. The Stop solution (acid) halts the HRP reaction and converts the color to yellow, allowing stable measurement at 450 nm.
Optical Density at 450 nm (OD₄₅₀)
The absorbance measured at 450 nm using an ELISA plate reader after TMB development and stopping. OD₄₅₀ values reflect the amount of HRP-mediated color change and are used to construct binding curves for each antibody.
Acronyms:
CV – Coefficient of Variation.
DPBS – Dulbecco’s Phosphate-Buffered Saline.
EC₅₀ – Half-maximal effective concentration.
ELISA – Enzyme-Linked Immunosorbent Assay.
HRP – Horseradish Peroxidase.
IgA – Immunoglobulin A.
kDa – KiloDalton (molecular mass unit).
LoD – Limit of Detection.
NHP – Nonhuman Primate.
NHPRR – Nonhuman Primate Reagent Resource.
OD – Optical Density (absorbance).
O/N – Overnight (incubation at 4 °C unless otherwise stated).
PBS – Phosphate-Buffered Saline.
RT – Room Temperature.
TA – Test Article.
TA-pAb – Test Article polyclonal anti-rhesus IgA antibody (goat).
TA-mAb – Test Article monoclonal anti-rhesus IgA antibody (mouse).
TMB – 3,3′,5,5′-Tetramethylbenzidine.
Principle
Analyte/Antigen: recombinant rhesus IgA, monomeric and dimeric forms (two plates processed in parallel).
Test Articles (TA):
TA-pAb: goat anti-rhesus IgA (polyclonal)
TA-mAb: mouse anti-rhesus IgA (monoclonal)
Controls:
Negative Control Ab: same species/format as TA, known non-binder to rhesus IgA
Reference Positive Control Ab: previously qualified anti-IgA (optional but recommended for plate validity)
Identity is confirmed by a specific binding signature to both monomeric and dimeric IgA with acceptable curve fit and specificity, and by matching expected relative response with defined limits.
See Acceptance Criteria Section below.
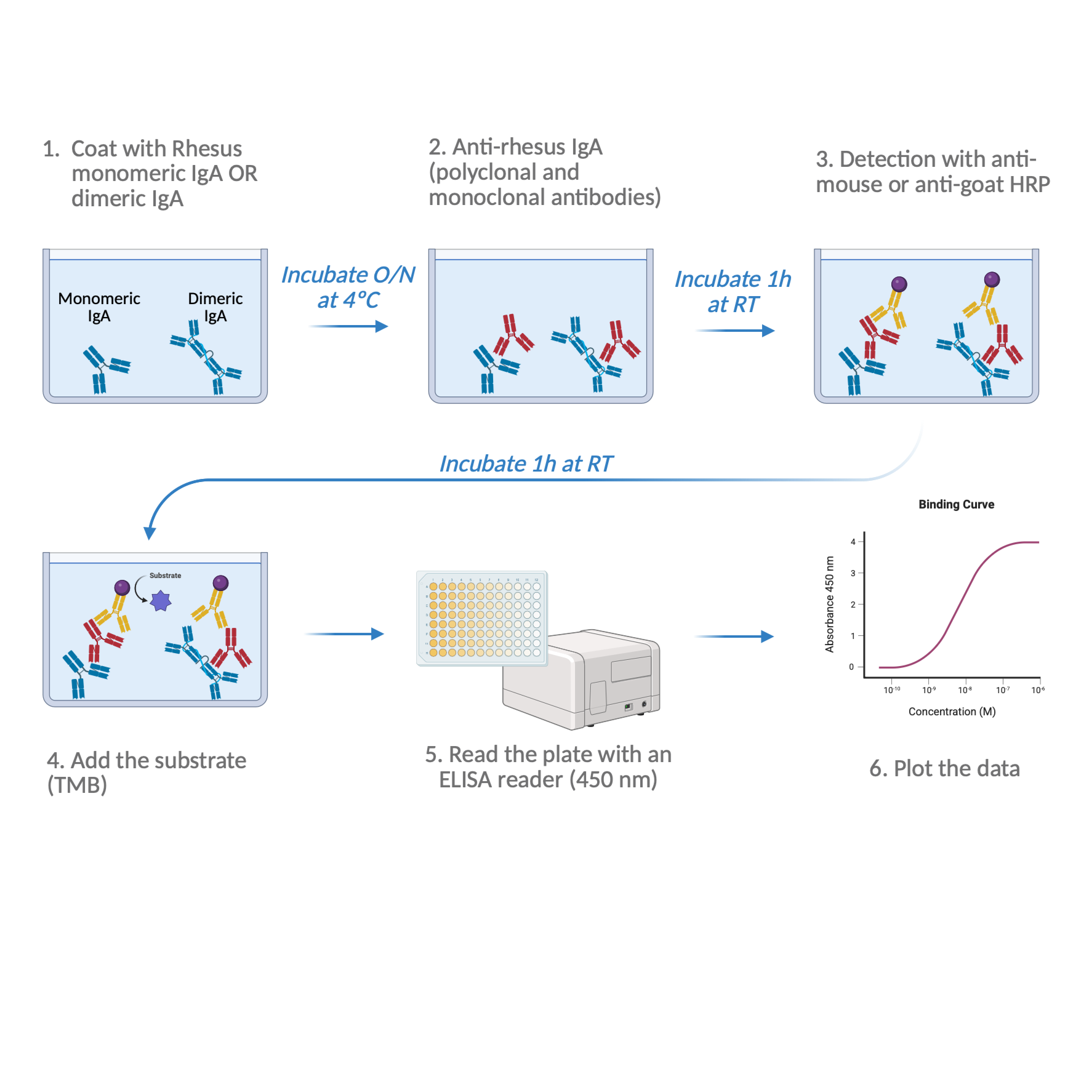
Figure 1: Workflow for Identification of Anti-Rhesus IgA by ELISA
Publication citation: Created in BioRender. de Vasconcellos Castro, J. (2026) https://BioRender.com/m2svlm2
Concentration & Dilution Notes
Please read the guidelines on concentration units under Guidelines & Warnings.
Use the dilution formula:
C₁V₁= C₂V₂
Where:
- C₁ = stock concentration
- V₁ = volume of stock needed
- C₂ = desired final concentration
- V₂ = final total volume
Example: To prepare 1 mL of antigen at 1 µM from a 1 mM stock:
- C₁ = 1 mM = 1000 µM
- C₂ = 1 µM
- V₂ = 1 mL = 1000 µL
- V₁=(C₂ × V₂)/ C₁= 1 μM × 1000 μL/1000 μM = 1 μL
So: Add 1 µL of stock + 999 µL of DPBS to reach 1 µM in 1 mL total volume.
Wash Buffer Preparation (1x DPBS + 0.05% Tween-20)
Add 18 L of deionized water in a container.
Add 2 L of 10x DPBS and mix gently.
Add 50 mL of Tween-20 and mix gently.
Note
Scale up or down according to the need.
Day 1 - Plate Coating (~30 min preparation + O/N incubation)
1d
Prepare 10 nanomolar (nM) (C2) of IgA in 1x DPBS.
Two working plates in parallel:
- Plate 1: Monomeric IgA
- Plate 2: Dimeric IgA
Plate Layout (See Figure 2):
- Columns 1-9: Coating with IgA (as per plate type);
- A10-A11: Coat only (IgA);
- B10-F11: 1x DPBS only controls;
- G10-G11: Blank (1x DPBS)
Figure 2: Coating Plate Layout
Publication citation: Created in BioRender. de Vasconcellos Castro, J. (2026) https://BioRender.com/rh7lu1d
Calculation Volumes (in Table 1)
V2 = 74 wells/plate x 100 µL = 7400 µL; prepare 7800 µL/plate to allow excess.
Reference rhesus monomeric IgA [CC6.29]Nonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00377
Reference rhesus dimeric IgA [CC6.29]Nonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00378
| Antibody | Initial Concentration (C1) | Final Concentration (C2) | Final Volume (V2) | Initial Volume (V1) | |
| Reference rhesus monomeric IgA | 2.69 mg/mL, 146.6 kDa, 18.3 µM | 10 nM | 7800 µL | 4.3 µL | |
| Reference rhesus dimeric IgA | 0.56 mg/mL, 308.7 kDa, 1.8 µM | 10 nM | 7800 µL | 43.3 µL |
Table 1: Calculation for the coating solution.
Note
For accuracy, remove the equivalent DPBS volume and add the same volume of stock.
Add 100 µL /well.
Seal the plate/s and incubate Overnight at 4 °C .
Day 2 - Blocking (~15 min preparation + 1 h incubation)
1h 15m
Wash the working plates twice with wash buffer.
2m
Prepare the Blocking buffer with 5 % volume milk in wash buffer:
For 150 mL : add 7.5 g milk to 150 mL PBS + 0.05 % volume Tween-20 .
9m
Add 200 µL /well.
3m
Seal the plate/s.
1m
Incubate 01:00:00 at Room temperature
Note
Potential STOP point: store up to 1-2 days at 4 °C in blocking buffer
1h
OPTION A - Indirect ELISA: Primary antibody preparation and incubation (~30 min preparation + 1 hour incubation)
1h 30m
Prepare 40 nanomolar (nM) (C₂) working stocks of TA-pAb, TA-mAb, and Negative Control Ab in blocking buffer.
Plate Layout (Figure 3):
- Columns 1-3: TA-pAb (goat anti-rhesus IgA, pAb);
- Columns 4-6: TA-mAb (mouse anti-rhesus IgA, mAb);
- Columns 7-9: Negative control Ab (non-binder);
- Row A10-11: Blocking buffer (coated wells only);
- Row B10-11: TA-pAb controls;
- Row C10-11: TA-mAb controls;
- Row D10-11: Negative control Ab controls;
- Row E10-F11: Reserved for secondary-only controls;
- Row G10-11: Blank.
Figure 3: Primary Antibodies Plate Layout
Publication citation: Created in BioRender. de Vasconcellos Castro, J. (2026) https://BioRender.com/kvj3gee
Volume Calculations V2 (in Table 2):
5 wells/plate/each antibody (3 wells for the triplicate + 2 wells for the control)
2 plates total
200 µL of primary antibody solution/well
V₂ = 5 x 2 x 200 = 2000 µL - Make it in excess
V₂ = 2500 µL
Goat Anti-Rhesus IgA [GARI]Nonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00366
Anti-IgA [NHPMab001]Nonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00423
| Antibody | Initial Concentration (C1) | Final Concentration (C2) | Final Volume (V2) | Initial Volume (V1) | |
| Goat anti-rhesus IgA [GARI] | 6.64 mg/mL, 150 kDa, 44 µM | 40 nM | 2500 µL | 2.272 µL | |
| Mouse anti-rhesus IgA [NHPMab001] | 3.78 mg/mL, 150 kDa, 25 µM | 40 nM | 2500 µL | 4 µL |
Table 2: Calculation for the working stock solution.
Note
For accuracy, remove the equivalent blocking buffer volume and add the same volume of stock.
In a dilution plate, dispense 200 µL of working stock to row A of designated wells (Figure 3). Control wells will also receive the working stock.
Note
- The dilution plate allows for preparing the dilutions ahead of time, while the working plate is for blocking. Also, in case of mistakes, you can toss the dilution plate without having to repeat the whole experiment.
- The dilution plate is an exact same copy of the working plate.
Add 150 µL of blocking buffer to the remaining wells.
Perform 1:4 vertical serial dilutions(50 µL transfers, mix 5x, discard the very last 50 µL ). Ensure equal volumes visually.
See Figure 4 for reference.
Figure 4. Schematic representation of 1:4 vertical serial dilution.
Publication Citation: Created in BioRender. de Vasconcellos Castro, J. (2026) https://BioRender.com/vc899id
Wash the working plate twice.
Transfer 100 µL /well from the dilution plate to the working plate.
Seal the plate and incubate for 01:00:00 at Room temperature .
1h
OPTION B - Direct ELISA: Primary antibody preparation and incubation (~30 min preparation + 1 hour incubation)
1h 30m
Use the same coated and blocked plates as in Section 6.
Prepare HRP-conjugated TA-pAb and TA-mAb at 40 nanomolar (nM) working stocks in blocking buffer.
Note
The layout of the working plate is identical to that in Section 7.
Volume Calculations V2 (in Table 2):
5 wells/plate/each antibody (3 wells for the triplicate + 2 wells for the control)
2 plates total
200 µL of primary antibody solution/well
V₂ = 5 x 2 x 200 = 2000 µL - Make it in excess
V₂ = 2500 µL
Goat Anti-Rhesus IgA [GARI]-HRPNonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00365
Anti-IgA [NHPMab001]-HRPNonhuman Primate Reagent Resource (NHPRR)Catalog #CAT-00392
| Antibody | Initial Concentration (C1) | Final Concentration (C2) | Final Volume (V2) | Initial Volume (V1) | |
| HRP Goat anti-rhesus IgA [GARI] | 0.5 mg/mL, 150 kDa, 3.3 µM | 40 nM | 2500 µL | 30 µL | |
| HRPMouse anti-rhesus IgA [NHPMab001] | 0.5 mg/mL, 150 kDa, 3.3 µM | 40 nM | 2500 µL | 30 µL |
Table 2: Calculation for the working stock solution.
Note
For accuracy, remove the equivalent blocking buffer volume and add the same volume of stock.
Set up the dilution plate as in sections 7.1, 7.2, and 7.3.
Wash the working plate twice.
Transfer 100 µL /well from the dilution plate to the working plate.
Seal the plate and incubate for 01:00:00 at Room temperature .
Note
For Direct ELISA, skip the secondary antibody preparation and incubation in Section 9, and go directly to Detection & Reading (Section 10).
Secondary antibody preparation and incubation (~20 min preparation + 1 hour incubation)
1h 20m
Prepare each HRP secondary antibody at 1 nanomolar (nM) in blocking buffer:
- mouse anti-goat HRP (detects TA-pAb)
- goat anti-mouse HRP (detects TA-mAb)
- Assign secondary for the Negative control Ab species as applicable.
Plate layout (Figure 5):
- Columns 1-3: HRP anti-TA-pAb, mouse anti-goat HRP;
- Columns 4-6: HRP anti-TA-mAb, goat anti-mouse HRP;
- Columns 7-9: HRP anti-negative control Ab, depending on the control antibody of your choice;
- Row A10-11: Blocking buffer (coat only);
- Row B10-11: Blocking buffer (TA-pAb controls);
- Row C10-11: Blocking Buffer (TA-mAb controls);
- Row D10-11: Blocking buffer (Negative control Ab controls);
- Row E10-F11: HRP secondary antibodies controls;
- RowG10-11: Blocking buffer, blank.
Figure 5: Secondary Antibody Plate Layout
Publication Citation: Created in BioRender. de Vasconcellos Castro, J. (2026) https://BioRender.com/n5v2prn
Volume calculations V2 (in Table 3):
26 well/plate will receive the secondary antibody
2 plates total
100 µL of coating solution/well
V₂ = 26 x 2 x 100 = 5200 µL - Make it in excess
V₂ = 5600 µL
Mouse Anti-Goat IgG Fc-HRP (SB115d)Southern BiotechCatalog #6158-05
Peroxidase AffiniPure® F(ab)₂ Fragment Goat Anti-Mouse IgG, Fcγ fragment specificJackson ImmunoResearch Laboratories, Inc.Catalog #115-036-071
| Antibody | Initial Concentration (C1) | Final Concentration (C2) | Final Volume (V2) | Initial Volume (V1) | |
| Mouse anti-goat IgG Fc-HRP (SB115d) | 0.75 mg/mL, 150 kDa 5 µM | 1 µM | 5600 µL | 1.12 µL | |
| HRP F(ab)2 Fragment Goat Anti-Mouse IgG | 0.6 mg/mL, 100 kDa (it’s a F(ab')2 ) = 6 µM | 1 µM | 5600 µL | 0.933 µL |
Table 3: Calculation for the secondary antibody solution.
Note
Depending on the Control Antibody of your choice, you will need to adjust the volume of secondary antibody (V₂).
Note
For accuracy, remove the equivalent blocking buffer volume and add the same volume of stock.
Wash working plates twice in wash buffer.
Add 100 µL /well.
Seal the plate and incubate for 01:00:00 at Room temperature .
1h
Detection & Reading (~15 min)
15m
Wash the working plates twice with wash buffer.
Add 100 µL /well of TMB substrate.
Incubate for 5-10 min.
Note
Monitor the color development as this can vary depending on the antibodies.
Stop the reaction with 100 µL /well ELISA Stop solution.
Read the plates at 450 nm.
Data Extraction and Organization
Export OD450 readings from the plate reader as a ".csv" or ".xlsx" file.
Option A - Excel
- Organize the data with columns for concentration (nM) and raws for the TAs (raw OD450 values);
- Average the triplicates and calculate %CV;
Note
%CV = Coefficient of Variation, measures the variability among replicates. Lower values indicate precision between replicates. For ELISA assays ≤15% is generally acceptable.
Calculate it as follows:
%CV = (Standard Deviation, SD / Mean) x 100
- Save the table and import into GraphPad Prism or other analysis software;
- Go to Section 12.
Option B - GraphPad Prism
- Open GraphPad Prism and create a new XY data table (X = Concentration, Y = OD450 readings);
- Under X, enter the antibody concentrations and label the column "Concentration (nM)";
- Under Y, paste the corresponding raw OD450 values in three replicate subcolumns for each sample;
- Prism will automatically generate a graph, under the "Graphs" tab;
- Adjust the graph layout as desired (color, font, size, legends, etc...);
- Since concentrations are expressed in linear values, set the X-axis to logaritmic scale:
Double-click the X-axis -> under Scale, select "Log 10".
Now you are ready to analyze the samples.
Data Analysis & Interpretation
In Prism, choose Analyze -> Nonlinear regression (curve fit)
Select "Dose-response - sigmoidal, 4PL, X is the concentration"
Under Settings -> Constraints, leave unconstrained unless you have historical limits already set.
Outputs per curve:
- R2
- EC50
- Top (maximum OD)
- Bottom (baseline OD)
- Hill slope
Acceptance Criteria
The acceptance criteria will be the following:
| Parameter | Acceptance Criterion | Rationale | |
| Goodness of fit (R2) | ≥0.98 | Indicates strong fit of the 4PL model to the experimental data | |
| Top (maximum OD) | within 1.0 and 4.0 OD units (depends on the LoD of the reader) | Ensures the signal is within the reader's linear range and avoids saturation | |
| Bottom (baseline) | ≤0.10 OD | Confirms minimal backgroud and proper washing/blocking | |
| Hill Slope | Between 0.8 and 2.0 | Reflects a good dose-response curve steepness. Values outside may indicate hook effect or non specific binding | |
| EC50 (half-maximal concentration) | within 0.5 to 2 x of an established range or a reference lot if available | Ensures the antibody affinity is consistent with its identity | |
| Replicate precision (%CV) | ≤15 % | Confirms intra-assay repeatability and accurate pipetting | |
| Specificity check (Negative control Ab) | ≤0.10 OD | Verifies that binding is specific to IgA and not background or nonspecific interactions | |
| Hook effect check | No drop in OD at highest concentrations | Ensures no antigen excess. You can exclude the point and refit the curve |
Summary of acceptance criteria
The test articles (antibodies) identity is confirmed when the 4PL regression meets the above criteria on both monomeric and dimeric IgA plates.
Protocol references
Abcam. (2024). Required controls for ELISA. Abcam.com. https://www.abcam.com/en-us/technical-resources/guides/elisa-guide/controls-for-elisa
BosterBio. (n.d.). How to design positive and negative controls for ELISA. https://www.bosterbio.com/how-to-design-positive-negative-controls-ihc-western-blot-elisa
de Vasconcellos Castro, J. (2026) https://BioRender.com/m2svlm2
de Vasconcellos Castro, J. (2026) https://BioRender.com/rh7lu1d
de Vasconcellos Castro, J. (2026) https://BioRender.com/kvj3gee
de Vasconcellos Castro, J. (2026) https://BioRender.com/vc899id
de Vasconcellos Castro, J. (2026) https://BioRender.com/n5v2prn
ELISA Guide; Part 1: Introduction to ELISA, Formats and Signal Amplification. (2023, November 30). https://www.jacksonimmuno.com/secondary-antibody-resource/immuno-techniques/elisa-guide-part-1/
Jackson ImmunoResearch. (n.d.). ELISA Guide Part 3: ELISA Optimization.https://www.jacksonimmuno.com/secondary-antibody-resource/immuno-techniques/elisa-guide-part-3-elisa-optimization/
Acknowledgements
This protocol and reagents were developed by the Nonhuman Primate Reagent Resource (NHPRR; RRID:SCR_012986; nhpreagents.org)
