Dec 11, 2025
Protocol to conduct measure visual evoked responses (VERs) on decapod crustacean species
- Rodrigo Lorenzo1,
- Luca Pettinau2,
- Henny Heinbert2,
- Guiomar Rotllant1,
- Albin Gräns3,
- Hans van de Vis2
- 1Institut de Ciències del Mar, Spanish National Research Council (CSIC), Barcelona, Spain.;
- 2Wageningen Livestock Research, De Elst 1, 6708 WD, Wageningen, The Netherlands.;
- 3Department of Animal Environment and Health, Swedish University of Agricultural Sciences, Skara, Sweden.

Protocol Citation: Rodrigo Lorenzo, Luca Pettinau, Henny Heinbert, Guiomar Rotllant, Albin Gräns, Hans van de Vis 2025. Protocol to conduct measure visual evoked responses (VERs) on decapod crustacean species. protocols.io https://dx.doi.org/10.17504/protocols.io.5jyl883e9l2w/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: In development
We are still developing and optimizing this protocol
Created: December 10, 2025
Last Modified: December 11, 2025
Protocol Integer ID: 234652
Keywords: Visual evoked response, VER, Lobster, shrimp, crab, unconsciousness, decapod crustacean brain, evoked response, decapod crustacean species this protocol, response to visual stimuli, decapod crustacean species, visual stimuli, crustacean, information on the consciousness state, ver, consciousness state, monitoring
Funders Acknowledgements:
European Union’s Horizon Europe Project (EUPAHW)
Grant ID: 101136346
Disclaimer
The authors acknowledge the anatomical and ontogenetic differences from a vertebrate brain; however, the terms 'brain' and 'brain activity' are used in accordance with the guidelines of Richter et al. (2010) to ensure comparability across invertebrate taxa.
Abstract
This protocol aims to standardize the monitoring of visual evoked responses (VERs) on decapod crustacean brain. This measure provides information on the consciousness state of a crustacean by evidencing the response to visual stimuli.
Guidelines
Visual Evoked Responses (VER) measure the brain’s response to a visual stimulus or light pulse (Baseler et al., 1994). This protocol aims to provide a real-time neurophysiological measure as an indicator of consciousness (Bowman et al., 2019; Hjelmstedt et al., 2022). Although tested and trialed, it may be modified as necessary. A prior anatomical study of the species is essential to ensure correct electrode placement. Crustaceans possess photoreceptors that respond to light stimuli with varying sensitivity (Meyer-Rochow, 2001; Starker Tomsic, 2004); therefore, the timing, intensity, frequency, and duration of light flashes should be optimized.
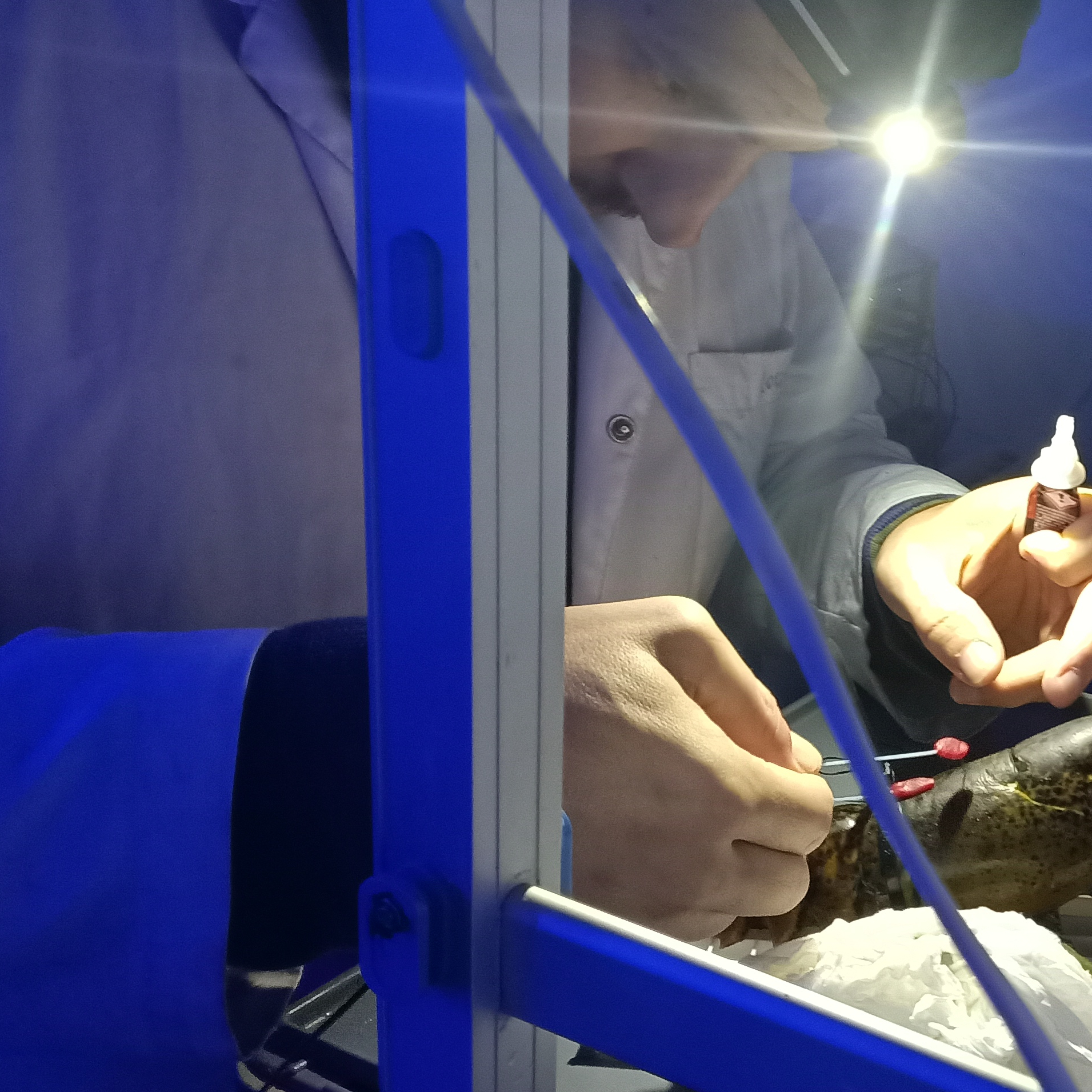
Electrodes should be placed flanking the crustacean brain (see EEG protocol, Lorenzo et al., 2025). On an opaque box, mount a lid that contains both the strobe light and the light detector (Fig. 1).
Place the recording instruments at a distance or on a separate surface from the one holding the animals in their medium (either fresh or seawater). Ensure that the recording device or computer has sufficient memory to prevent measurement interruptions, and verify that a stable electrical current is available to power the equipment.
Materials
-Strobe light
-Light sensor
-Program to modulate light frequency (and possibly intensity)
-Lid (opaque enough to avoid external light stimuli)
-Amplifier (in our case we use Quad and Octal Bio Amp from ADinstruments)
-Data digitalization device (in our case we use PoweLab C)
-Needle electrodes (a minimum of 3: positive, negative and ground)
-Labchart or software compatible with the measuring hardware
-Laptop, computer or device to measure and store data
-Precision driller (we use Proxxon micromot) and small drill bit (needle diameter)
Other materials
-Aquaria or container to hold the animals
-Mesh or platform to secure crustaceans
-Cable ties or other devices to secure objects together
-Dental wax
-Aquarium-safe superglue
Troubleshooting
Before start
1. Load recording program, tailor it for the desired measure (channel digital see Analysis) and pre-save it on a file. Also, pre-load annotation for faster (hot key) activation.
2. Check the light pulses are working on the desired frequency (2 Hz) and duration (50 ms), and the program is able to record them on real time through the scope view (on Labchart).
3. Place electrodes in the crustacean and check the neurological signal.
Experimental setup
Electrodes should be placed flanking the crustacean brain (see EEG protocol, Lorenzo et al., 2025). On an opaque box, mount a lid that contains both the strobe light and the light detector (Fig. 1).
Fig. 1. Schematic representation of the experimental setup.
Place the recording instruments at a distance or on a separate surface from the one holding the animals in their medium (either fresh or seawater). Ensure that the recording device or computer has sufficient memory to prevent measurement interruptions, and verify that a stable electrical current is available to power the equipment.
Before start
Load recording program, tailor it for the desired measure (channel digital see Analysis) and pre-save it on a file. Also, pre-load annotation for faster (hot key) activation.
Check the light pulses are working on the desired frequency (2 Hz) and duration (50 ms), and the program is able to record them on real time through the scope view (on Labchart).
Place the electrodes in the crustacean and check the neurological signal.
Procedure
Start the recording on the digital device (laptop) and allow some seconds for the system to have a baseline. If there is no recording or the recording is out of scale, re-check the electrodes placement.
Start the flashing device (either manually or with a program) at the desired frequency.
Check the scope view is correctly averaging the pages recorded (Fig. 2).
Fig. 2. Light (yellow) and dark (grey) phases recorded in Labchart and their average on the scope view.
Software
LabChart
NAME
Windows XP
OS
ADInstruments Australia
DEVELOPER
Perform a quality control procedure to verify the absence of light-induced electrical artefacts.
First, record 2 minutes of VERs under standard flashing-light conditions.
Then, occlude the light source with an dark cover to prevent any light from reaching the animal.
Reactivate the light-flash protocol while the source remains fully covered, and continue recording the electrophysiological signal for 2 minutes.
If the characteristic VER waveform is absent under these conditions, the response can be attributed to genuine neural activity rather than to stimulus-related electrical artefacts.
After a stable baseline and quality control is measured, apply any procedures that may affect the visual evoked response, making comment markers on the file for traceability.
Turn the strobe light back on and continue to measure through the new or novel condition imposed to the animal.
Once the whole experimental timeline is finished, stop the recording and save the file immediately.
Gently remove the implanted electrodes from the crustacean to avoid any damage and untie the animal by cutting their bindings.
Place the animals back into the maintenance tanks, preferably in isolation to follow up with the recovery.
Analysis and results
On the Data pad (for Labchart), configure the columns to extract the following information maximum amplitude on:
0-200 ms (light phase).
200-400 ms (dark phase).
Establish a time selection of the data (1 min preferably), depending on the experimental protocol followed, and add multiple data to Data pad.
Export data and make a quotient between the amplitude of the light and the amplitude of the dark phase (AMPVER).
Protocol references
Baseler, H. A., Sutter, E. E., Klein, S. A., Carney, T. (1994). The topography of visual evoked response properties across the visual field. Electroencephalography and clinical Neurophysiology, 90(1), 65-81.
Bowman, J., Hjelmstedt, P., Gräns, A. (2019). Non-invasive recording of brain function in rainbow trout: Evaluations of the effects of MS-222 anaesthesia induction. Aquaculture Research, 50(11), 3420-3428.
Hjelmstedt, P., Sundell, E., Brijs, J., Berg, C., Sandblom, E., Lines, J., ... Gräns, A. (2022). Assessing the effectiveness of percussive and electrical stunning in rainbow trout: Does an epileptic-like seizure imply brain failure?. Aquaculture, 552, 738012.
Rodrigo Lorenzo, Luca Pettinau , Hans van de Vis , Henny Heinbert , Albin Gräns , Guiomar Rotllant 2025. Protocol to conduct an EEG on decapod crustacean species. protocols.io https://dx.doi.org/10.17504/protocols.io.eq2ly4mdqk9/v1
Meyer-Rochow, V. B. (2001). The crustacean eye: dark/light adaptation, polarization sensitivity, flicker fusion frequency, and photoreceptor damage. Zoological science, 18(9), 1175-1197.
Richter, S., Loesel, R., Purschke, G., Schmidt-Rhaesa, A., Scholtz, G., Stach, T., ... Harzsch, S. (2010). Invertebrate neurophylogeny: suggested terms and definitions for a neuroanatomical glossary. Frontiers in Zoology, 7(1), 29.
Sztarker, J., Tomsic, D. (2004). Binocular visual integration in the crustacean nervous system. Journal of Comparative Physiology A, 190(11), 951-962.
Acknowledgements
European Union’s Horizon Europe Project (EUPAHW)
Project ID: 101136346