Aug 02, 2024
Version 1
Protocol for the production and storage of Diplodia sapinea pycnidiospores V.1
- Anne Geertje Oostlander1,
- Laura Brodde2,
- Miriam von Bargen1,
- Rasmus Enderle3,
- Marco Leiterholt1,
- Dagmar Trautmann3,
- Malin Elfstrand4,
- Jan Stenlid4,
- André leißner1
- 1Institute of Genetics, Technische Universität Braunschweig, Braunschweig, Germany;
- 2Swedish University of Agricultural Sciences, Uppsala, Sweden;
- 3Julius Kühn Institute (JKI)—Federal Research Centre for Cultivated Plants, Braunschweig, Germany;
- 4Department of Forest Mycology and Plant Pathology, Swedish University of Agricultural Sciences, Uppsala, Sweden

Protocol Citation: Anne Geertje Oostlander, Laura Brodde, Miriam von Bargen, Rasmus Enderle, Marco Leiterholt, Dagmar Trautmann, Malin Elfstrand, Jan Stenlid, André leißner 2024. Protocol for the production and storage of Diplodia sapinea pycnidiospores. protocols.io https://dx.doi.org/10.17504/protocols.io.kqdg3271pv25/v1
Manuscript citation:
Oostlander AG, Brodde L, Bargen M von, Leiterholt M, Trautmann D, Enderle R et al. A reliable and simple method for the production of viable pycnidiospores of the Pine pathogen Diplodia sapinea and a spore-based infection assay on Scots Pine. Plant Dis 2023; 107(11):3370–7.
doi.org/10.1094/PDIS-01-23-0107-RE
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: July 25, 2024
Last Modified: August 02, 2024
Protocol Integer ID: 104068
Keywords: Diplodia sapinea, Diplodia tip blight, in vitro sporulation, pycnidiospores, Sphaeropsis sapinea, viable pycnidiospores of diplodia sapinea, storage of diplodia sapinea, diplodia sapinea, viable pycnidiospore
Abstract
An efficient, standardized protocol for the production and storage of highly viable pycnidiospores of Diplodia sapinea.
Image Attribution
Oostlander AG, Brodde L, Bargen M von, Leiterholt M, Trautmann D, Enderle R et al. A reliable and simple method for the production of viable pycnidiospores of the Pine pathogen Diplodia sapinea and a spore-based infection assay on Scots Pine. Plant Dis 2023; 107(11):3370–7.
Materials
Material, media and solutions:
• Petri dishes (5.5 cm)
• D. sapinea (e.g. ex-type strain CBS 138184)
• Light shelf with cold white daylight (Osram Lumilux 18W/865), intensity from 5000 to 6500 lux
• Cleanbench
• Centrifuge
Minimal medium (VMM):
as described in Vogel, 1964
20 ml Vogel’s solution
20 g sucrose
15 g agar
add 1 l water autoclave
Trace element solution:
50 g citric acid
50 g zinc sulfate (J.T. Baker)
10 g ammonium iron(II) sulfate
2,5 g copper sulfate
0,5 g manganese sulfate
0,5 g boric acid
0,5 g sodium molybdate
add 1 l water
Chloroform (1 ml) is added as a preservative, store at room temperature.
Vogel’s solution:
125 g sodium citrate
250 g potassium dihydrogen phosphate
100 g ammonium nitrate
10 g magnesium sulfate
5 g calcium chloride
5 ml trace element solution
2,5 ml biotin solution
add 1 l water
Chloroform (2 ml) is added as a preservative, store at room temperature.
Biotin solution:
0,1 g biotin
add 1 l water
Store at -20°C
0,01 % (v/v) Tween:
0,1 ml Tween
add 1 l water
aliquod and autoclave
40% (v/v) glycerol
200 ml glycerol
add 1 l water
aliquod and autoclave
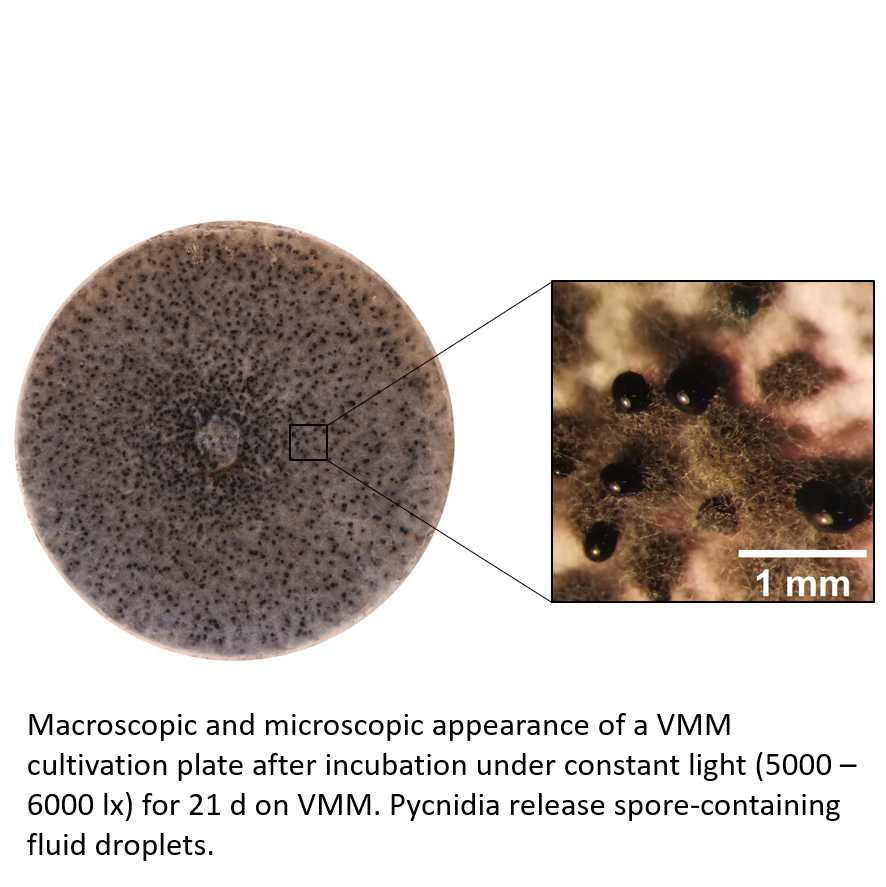
Inoculate a 5.5 cm Petri dish containing 10 to 15 ml of solid VMM with D. sapinea with an agar plug with mycelia from a preculture on VMM.
Incubate the plate for 21 d at 26 to 29°C with constant light (cold daylight at an intensity of 5000 to 650 lux). Pycnidia will form on the surface of the culture and open to release a droplet of liquid containing spores.
Harvest the spores from the plate by adding approximately 2 ml of 0.01% (v/v) Tween to the surface and rinse the surface of the colony several times by pipetting. Repeat the procedure for a higher yield. Typically, between 1x106 and 2x106 spores can be harvested per plate. Spores can be separated from the liquid by centrifugation for 10 s at 5000 rpm and then resuspended in your desired volume.
For storage, mix 500 µl of the spore suspension and 500 µl 40% glycerol in a 2 ml screw top tube or cryovial. Freeze the glycerol stock tube at -80°C.
Protocol references
Vogel HJ. A convenient growth medium for Neurospora crassa (Medium N). Microbial genetics bulletin 1956; (13):42–3.
