Jun 08, 2025
Pressure measurement in intact mouse fundus
- Maryrose Sullivan1,
- Vivian Cristofaro1
- 1VA Boston Healthcare System
- SPARCTech. support email: [email protected]

Protocol Citation: Maryrose Sullivan, Vivian Cristofaro 2025. Pressure measurement in intact mouse fundus . protocols.io https://dx.doi.org/10.17504/protocols.io.3byl4q2n8vo5/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: June 26, 2023
Last Modified: June 08, 2025
Protocol Integer ID: 84017
Keywords: gastric fundus, mouse, pressure, motor function of the fundus, excitatory enteric neurotransmission, enteric neurotransmission, region of the stomach, measuring intralumenal pressure, intact fundus, isolated intact fundus, intralumenal pressure, gastric storage, coordinated gastric storage, fundus, stomach, intact mouse
Funders Acknowledgements:
Department of Veterans Affairs
Grant ID: BX001790
Abstract
Each region of the stomach has distinct functional characteristics to ensure coordinated gastric storage and emptying. Inhibitory and excitatory enteric neurotransmission plays an important role in the adaptive relaxation of the fundus and the emptying of its contents. This protocol describes a procedure to examine the functional and mechanical properties of this specific region of the stomach by measuring intralumenal pressure in the isolated intact fundus. This method can be used to explore the motor function of the fundus under a variety of experimental conditions or disease states while largely preserving its three-dimensional shape.
Materials
Silk suture: 8-0, cat# 2023 , Mani Inc, Japan
Organ bath: cat# 158505, Radnoti LLC, Covina, CA
Pressure transducer: PX260, Edwards Lifesciences, Irvine, CA
Infusion pump: PHD Ultra syringe pump, cat# 70-3007 or 70-3005, Harvard Apparatus, Holliston, MA
Stimulator: RADSTIM Stimulator, cat# RS1000, Radnoti LLC, Covina, CA, USA
Data acquisition system: DataQ Instruments, Akron, OH
WinDaq software: Version 2.87, DataQ Instruments, Akron, OH
Prostaglandin F2α: cat# 4214, Tocris, Minneapolis, MN
Atropine: cat# A0132, Sigma-Aldrich, St. Louis, MO
Guanethidine: cat# G-8520, Sigma-Aldrich, St. Louis, MO
L-NAME: cat# N5751, Sigma-Aldrich, St. Louis, MO
SNP: cat# 71780, Sigma-Aldrich, St. Louis, MO
Troubleshooting
Isolation of Gastric Fundus
1h 9m
Euthanize mice according to IACUC approved method (i.e., CO2 asphyxiation).
5m
After abdominal incision, excise the stomach by transecting the lower esophagus and the pylorus.
1m
Quickly place stomach in Sylgard-coated dissecting dish filled with cold Krebs solution and continuously bubbled with carbogen (95% O2 and 5% CO2).
Kreb's Solution:
- NaCl 120mM
- KCl 5.9mM
- NaHCO3 25mM
- Na2H3PO4 1.2mM
- MgCl2 • 6H2O 1.2mM
- CaCl2 2.5mM
- dextrose 11.5mM
.
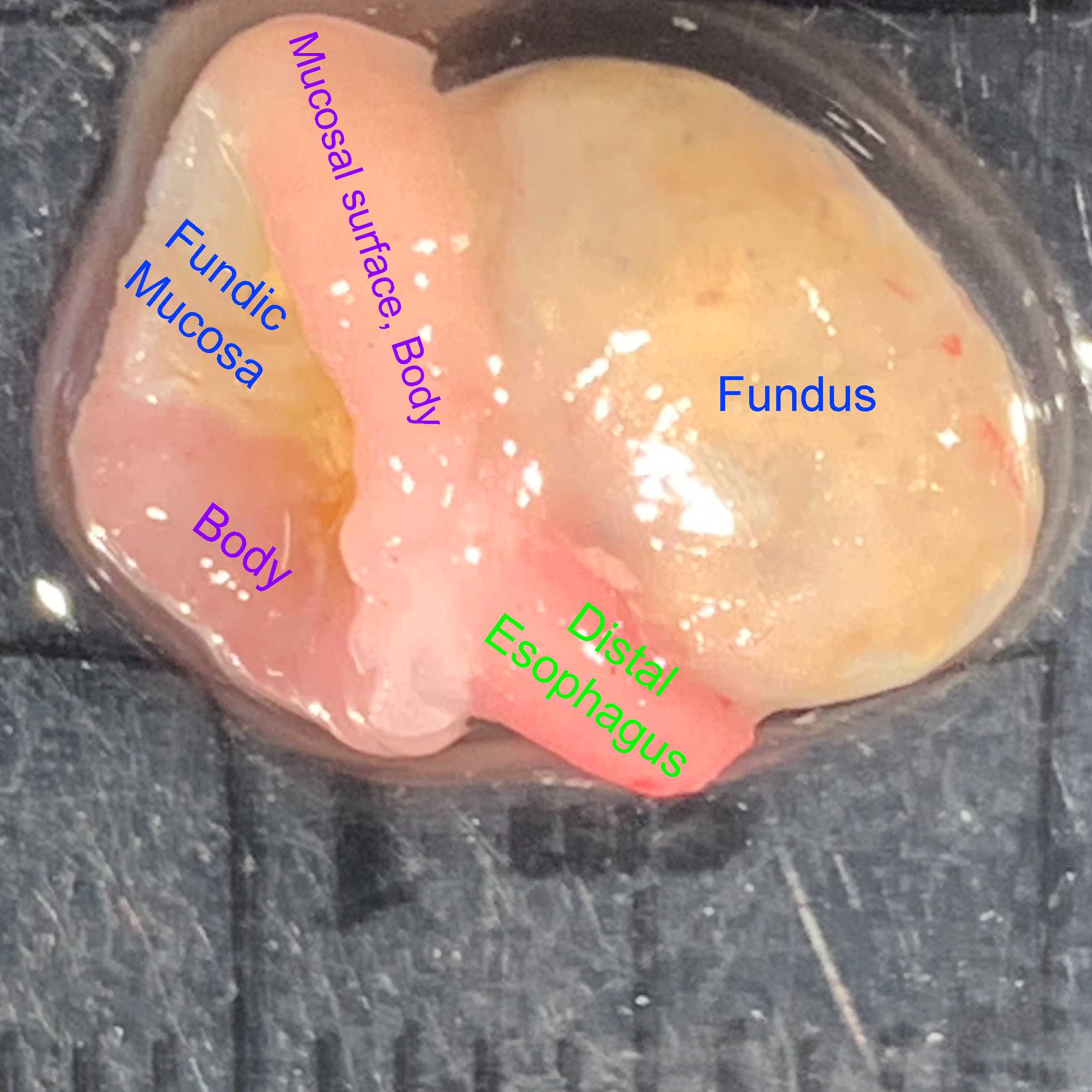
Isolate the fundic portion of the stomach from the remaining regions by cutting through the corpus below the gastroesophageal junction, leaving about 3mm of corporal tissue attached to the fundus.
5m
Gently remove the stomach contents.
1m
Immobilize the fundus on the bottom of the dissecting dish using insect pins inserted into the edges of the corpus.
3m
With the aid of a stereoscope, insert a 4F double lumen catheter into the esophageal stump and through the gastroesophageal junction, taking care not to evert the mucosa. Secure in place with suture. Flush both lumens with Kreb's solution to ensure that air bubbles have been removed.
15m
Close the distal edges of the fundus with a horizontal mattress suture using 8-0 silk (8-0 ophthalmic silk, cat# 2023, Mani Inc., Japan), keeping the mucosal surfaces in apposition and using the reflected edge of the corpus to support the stitches. The boundary between the fundus and body can be identified by the difference in mucosal color.
30m
Close the region below the base of the esophagus with a purse-string suture.
10m
Test for fluid leakage: Attach a 1mL syringe to the catheter and slowly infuse Kreb's solution into the fundic lumen. Verify the absence of bubbles from the suture line.
5m
Measurement of pressure in the fundus
Position the intact fundus between platinum electrodes in a 5ml organ bath (cat# 158505, Radnoti LLC, Covina, CA) that has been filled with Kreb's solution, maintained at 37°C and continuously aerated with carbogen.
5m
Attach one lumen of the catheter to a calibrated pressure transducer (PX260, Edwards Lifesciences, Irvine, CA) and the other lumen to an infusion pump (PHD Ultra syringe pump, cat# 70-3007 or 70-3005, Harvard Apparatus, USA). Monitor intraluminal pressure continuously using a Data-Q acquisition system driven by WINDAQ software (DATAQ Instruments, Inc. USA). Many commercially available acquisition systems are adequate for this purpose.
5m
Equilibrate tissue for 45-60 minutes.
1h
Generation of nerve-mediated relaxation responses in contracted fundus
After equilibration, slowly infuse Kreb's solution into fundus at 10μl/minwhile continuously measuring intralumenal pressure.
10m
With 100μl instilled into the lumen, pre-contract the fundus with prostaglandin F2α (10μM) under non-adrenergic, non-cholinergic conditions (i.e., in the presence of atropine 1μM, and guanethidine 10μM).
5m
Once the isovolumetric pressure in response to PGF2α reaches a plateau, generate nerve-mediated relaxation responses using electrical field stimulation (EFS, 30V, 0.5ms pulse duration, 10 seconds) over a range of frequencies (2-30Hz) delivered by an electronic stimulator (RADSTIM Stimulator, cat# RS1000, Radnoti, Covina, CA, USA).
10m
Post-junctional relaxation responses can be examined by exposing the fundus to a nitric oxide donor such as sodium nitroprusside (SNP 10μM).
5m
Withdraw the infused fluid from the lumen and wash the tissue by changing the Kreb's solution in the organ bath 2-3 times over 15 minutes.
15m
Evaluation of nitric oxide-mediated relaxation in the fundus
To evaluate the contribution of nitric oxide to the relaxation response, add a nitric oxide inhibitor (Nω-nitro-L-arginine methyl ester, 10μM or Nω-nitro-L-arginine, 10μM) to the organ bath for 15 minutes prior to evoking neurogenic relaxation responses as above (repeat steps 14-18).
40m
Pressure measurement in response to contractile stimuli
Slowly infuse Kreb's solution into fundus at 10μl/min while continuously measuring intralumenal pressure.
10m
To evaluate receptor-independent contractile efficiency of the intact fundus, expose the preparation to high extracellular potassium by replacing the solution in the organ bath with a modified Kreb's solution in which the concentration of KCl is increased to 120mM, with a proportional reduction in the concentration of NaCl.
5m
Measure weight of fundus
5m
At the end of the experiment, remove the catheter, open the fundus along the suture line and weigh the tissue after blotting excess liquid on filter paper.
5m
