Jan 19, 2026
Practical guidelines for producing non-replicating canine adenovirus vectors
- denis omara1,
- denis omara1
- 1Medical Research Council/Uganda Virus Research Institute & London School of Hygiene and Tropical Medicine (MRC/UVRI & LSHTM) Uganda Research Unit.
- Makerere University

Protocol Citation: denis omara, denis omara 2026. Practical guidelines for producing non-replicating canine adenovirus vectors. protocols.io https://dx.doi.org/10.17504/protocols.io.5qpvo131dg4o/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: January 19, 2026
Last Modified: January 19, 2026
Protocol Integer ID: 238888
Keywords: Canine adenovirus type 2 (CAV-2) vector, Expression, Expansion, Purification, Titration., canine adenovirus type, adenovirus vector, alternative adenoviral vector, field of alternative adenoviral vector, human adenovirus serotype, other vaccine development platform, vaccine development, recombinant viral genome, vaccine design, vaccine application, vaccine platform, release of the recombinant viral genome, suitable for vaccine application, vaccine efficacy, gene therapy, expressing gene, genes in mammalian cell, virus replication, existing immunity, virus stock expansion, virus stock, mammalian cell
Abstract
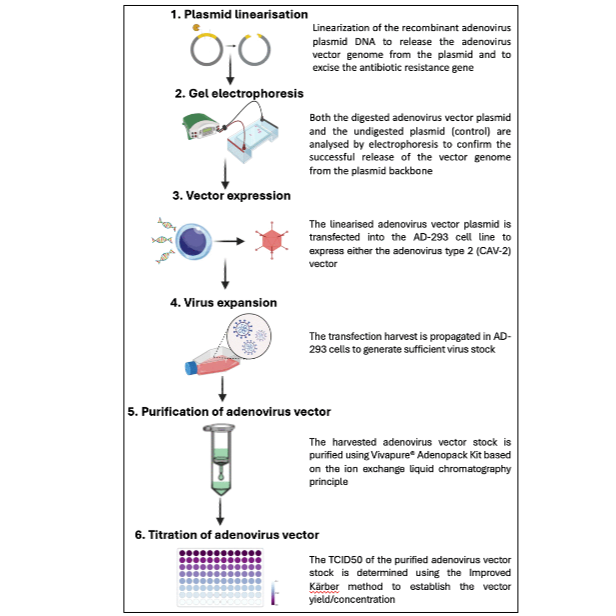
Adenovirus vectors have been used for vaccine development, gene therapy, among others, because of their ease of genetic manipulation and high efficiency in delivering and expressing genes in mammalian cells without causing significant cytotoxicity or integrating into the host genome. Adenovirus vectors are relatively immunogenic, hence avoiding the need for adjuvant when used in vaccine design, unlike other vaccine development platforms. Human adenovirus serotype 5 (HAdV-5) has traditionally been employed in vaccine platforms; however, its clinical utility is limited by widespread pre-existing immunity in human populations, which can reduce vaccine efficacy. As a promising alternative, canine adenovirus type 2 (CAV-2) vector offers several advantages, including low seroprevalence in humans, efficient infection of respiratory epithelial and neuronal cells, and long-lasting transgene expression. These features render the CAV-2 vector particularly suitable for vaccine applications requiring repeated administrations or booster doses. In this protocol, we describe methods for the expression, expansion, purification, and titration of a non-replicative CAV-2 vector in the AD-293 cell line. The procedure involves the release of the recombinant viral genome from the pUC19 plasmid backbone, followed by virus stock expansion in a complementing AD-293 cell line, which is able to express the E1 protein to aid virus replication. The virus stock was purified through ion-exchange liquid chromatography and titered using the Improved Kärber method. This work contributes to the growing field of alternative adenoviral vectors with implications for translational research by providing step-by-step procedures for producing a canine adenovirus type 2 (CAV-2) vector.
Troubleshooting
SOP for transfection, infection, expansion, and large-scale production of modified Adenovirus using AD293 cell line
SOP for Vivapure Adenopack 20 RT Virus Purification
SOP for virus titration using Improved Kärber Method
