Aug 01, 2025
Pole descent
Forked from Pole Test Assessment
- Anastasiya Moiseyenko1
- 1California Institute of Technology

Protocol Citation: Anastasiya Moiseyenko 2025. Pole descent. protocols.io https://dx.doi.org/10.17504/protocols.io.8epv5k9mjv1b/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: July 31, 2025
Last Modified: August 01, 2025
Protocol Integer ID: 223829
Keywords: ASAPCRN, Behavior, Mouse, PD, Neurobehavior, pole test, pole, mice, testing, parkinson's disease, motor function, pole descent, pole test assessment, mouse models of parkinson, pole descent pole descent, parkinson, assessment of gross motor function, mouse model, time for mice, gross motor function, mice
Funders Acknowledgements:
Aligning Science Across Parkinson's
Grant ID: ASAP-020495
Aligning Science Across Parkinson's
Grant ID: ASAP-000375
Heritage Medical Research Institute
Disclaimer
The protocols.io team notes that research involving animals and humans must be conducted according to internationally-accepted standards and should always have prior approval from an Institutional Ethics Committee or Board.
Abstract
Pole descent
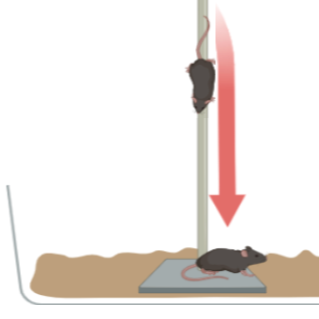
This test measures the time for mice to descend a 0.5m pole, an assessment of gross motor function for mouse models of Parkinson's disease
Image Attribution
Mouse climbing down pole placed inside cage
Materials
1. A metal pole with a diameter of 1cm and height of 50-60cm, this should come on a base. (See attached photo).
2. Foam/PVC liner, we use a cabinet liner similar to what can be found here. Adhesive Grip for Pole Test.
3. Standard Mouse cages.
4. Stopwatch/Timer.
5. GoPro/Means of recording each trial.

Figure 1. Example of pole test apparatus used by our lab.
Troubleshooting
Information
A 0.5m rod, 1cm in diameter, attached to a base and wrapped in adhesive foam (i.e. shelf liner) to increase grip is used. Training and testing is done on two consecutive days. Animals are placed onto the pole and trained to descend back into their home cage. Mice are given 3 trails on the training day and 2 trials on testing day. Experiments can be done inside a biosafety cabinet for animals with sensitive microbiomes.
Set up
Habituate animals to behavior testing room for 1 hour prior to assay start.
Wipe down the apparatus with animal-safe cleaning agent before beginning any trials and between each cage.
Remove cagemates from home cage into a clean cage and place pole apparatus into home cage.
Training Day
Place the animal at the bottom of the home cage with the pole for approximately 1 minute to allow exploration.
Training day consists of 3 trials for each mouse.
Trial 1: Place the mouse approximately ⅓ of the way up the pole, facing head down, and let freely descend.
Use gentle tail-touches to encourage movement if animal will not move. Readjust animal on beam to face head down if animal starts to climb up.
In between each trial allow the mouse rest time in the home cage for ~30 seconds.
Trial 2: Place the animal ⅔ of the way up the pole and let freely descend. Adjust animal on pole as needed.
Trial 2: Place the animal at the top of the pole and let freely descend. Adjust animal on pole as needed.
Test day
On the next day, set up the apparatus the same way as for training. Testing day consists of 2 trials for each mouse. Rotating through all animals for one trial, then moving onto the next trial, allows animals to rest.
Animals are timed starting at the moment the animal's tail is released at top of the pole and ending when one of the hindlimbs touches the base of the pole, with a maximum time of 30 seconds.
Trial 1: Place mouse face down at the top of the pole and allow to descend freely.
Trial 2: Repeat step 12.
Clean the pole and base before moving onto the next cage.
Protocol references
1. Sampson, T. R. et al. Gut Microbiota Regulate Motor Deficits and Neuroinflammation in a Model of Parkinson’s Disease. Cell 167, 1469-1480.e12 (2016).
