Sep 12, 2025
Plant extraction for LC-MS Analysis
Forked from Lyophilized plant extraction for LC-MS Analysis
- Tito Damiani1,
- Kateřina Kučerová2
- 1IOCB Prague;
- 2IOCB
- Pluskal Lab

Protocol Citation: Tito Damiani, Kateřina Kučerová 2025. Plant extraction for LC-MS Analysis. protocols.io https://dx.doi.org/10.17504/protocols.io.81wgbkj5qgpk/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: June 16, 2025
Last Modified: September 12, 2025
Protocol Integer ID: 220309
Keywords: Plant, Metabolomics, LC-MS, lyophilization, plant extraction for lc, extracting metabolite, plant extraction, plant material for lc, lyophilized plant material, ms analysis, transfer to lc
Abstract
This protocol describes a rapid and efficient method for extracting metabolites from lyophilized plant material for LC-MS analysis. Samples are homogenized, extracted with ethanol–water, dried, and reconstituted in water–acetonitrile before final centrifugation and transfer to LC-MS vials. The method is optimized for small-scale use and ensures compatibility with LC-MS workflows.
Attachments
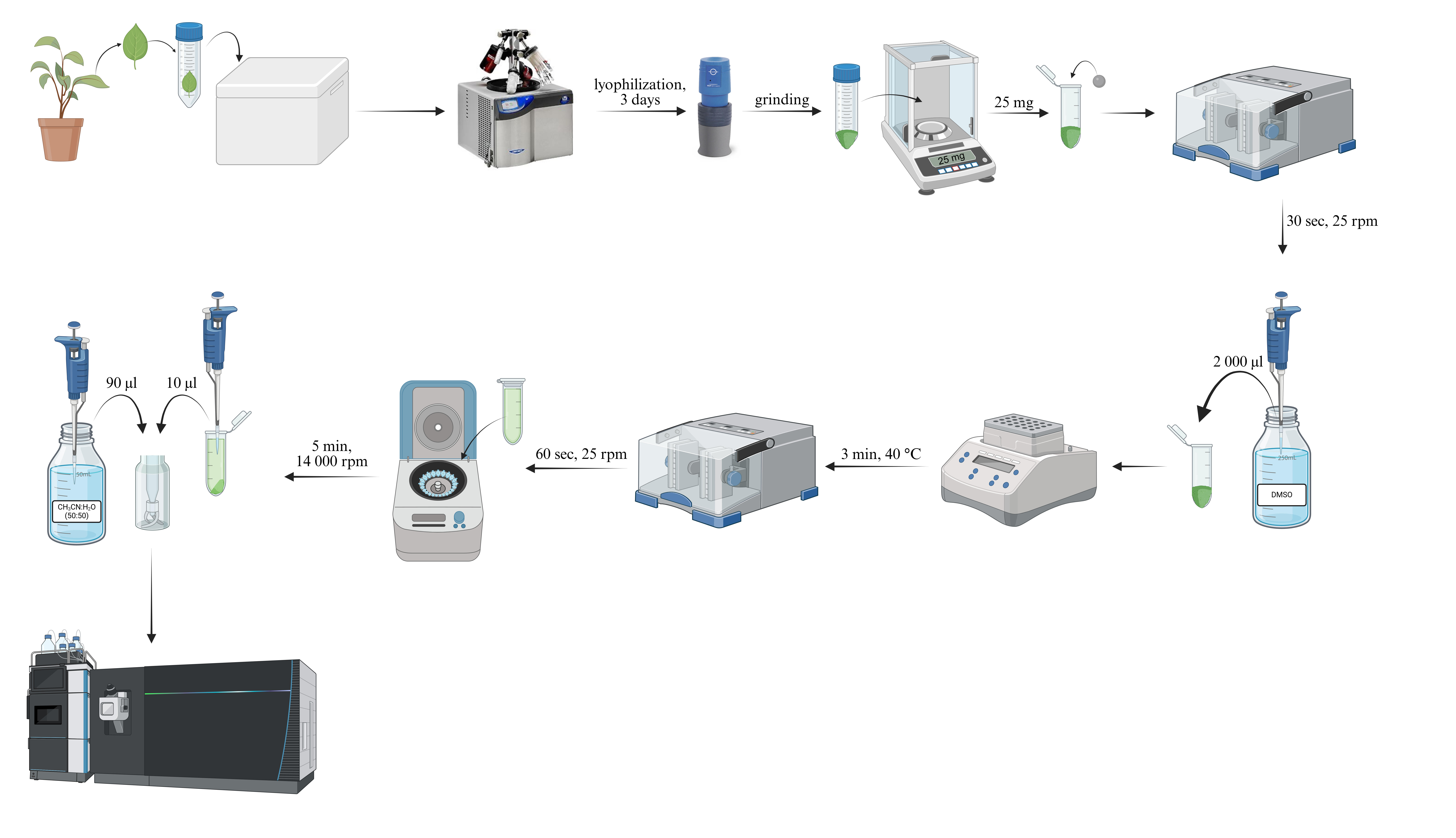
Workflow.png
4.4MB
Materials
Solvents
- DMSO
- CH3CN:H2O (50:50)
Experimental Supplies
- 2.0 ml Eppendorf tubes
- LC-MS vials
- Stainless Steel grinding beads
- Dry ice
Labels
- For weighted material
- For LC-MS vials
Instruments
- Analytical balance
- Analytical mill
- Thermal mixer
- TissueLyser
- Pipette (10–1000 μL) + tips
- Centrifuge
- Vortex
- LC-MS system
Troubleshooting
Plant collection, transportation, lyophilisation
Collect a portion of fresh plant material (leaf, root, stem, flower, fruit) into a 50 mL Falcon tube
Close the tubes tightly
Put the tubes in dry ice immediately for transportation to the lab
Unscrew the tubes
Cover the top and sides with one piece of thin net fabric
Attach the fabric to the tube with a rubber band
Insert the tubes into the lyophiliser flasks
Run the lyophilisation for 3 days (follow lyophiliser instructions)
Insert all the lyophilised material into a lab grinder
Grind the material to obtain a powder
Store the material in dry room in a tightly closed Falcon tube.
Plant extraction
4h 45m
Weight 25 ± 2.5 mg of lyophilized plant material into a 2.0 Eppendorf tube with round bottom
20m
Add a metal bead to each tube
30s
Homogenize in TissueLyser 30 sec, 25 rpm
30s
Add 2 000 μL of DMSO (HPLC grade) and vortex to mix
10m
Incubate 3 min at 40 °C
3m
Homogenise in TissueLyser 60 sec, 25 rpm
1m
Centrifuge 5 min, 14 000 rpm
5m
Transfer 10 μL of supernatant into LC-MS vials with insert and add 90 μL of CH3CN-H2O solution (50:50, v/v)
20m
