Feb 24, 2026
Nucleic Acid Extraction Using the MagMAX™ CORE Nucleic Acid Purification Kit on a Magnetic Particle Processor
- Nathaniel Higdon1,2,
- Leslie Blakemore1,
- Rachel Palinski1,
- Robin Holland1,
- Suelee Robbe-Austerman1
- 1Diagnostics and Biologics, National Veterinary Services Laboratories, Animal Plant Health Inspection Service, United States Department of Agriculture;
- 2Department of Veterinary Microbiology and Preventive Medicine, College of Veterinary Medicine, Iowa State University of Science and Technology
- USDA APHIS NVSL

Protocol Citation: Nathaniel Higdon, Leslie Blakemore, Rachel Palinski, Robin Holland, Suelee Robbe-Austerman 2026. Nucleic Acid Extraction Using the MagMAX™ CORE Nucleic Acid Purification Kit on a Magnetic Particle Processor. protocols.io https://dx.doi.org/10.17504/protocols.io.ewov1kypogr2/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: December 31, 2025
Last Modified: February 24, 2026
Protocol Integer ID: 236093
Keywords: MagMAX™ CORE, nucleic acid extraction, DNA purification, RNA purification, magnetic bead-based extraction, automated nucleic acid purification, guanidinium thiocyanate lysis, magnetic particle processor, KingFisher™ Flex, real-time PCR (rtPCR), sequencing, VetMAX™ Xeno™ Internal Positive Control, PCR inhibition monitoring, contamination control, diagnostic laboratory workflow, core nucleic acid purification kit, extracting nucleic acid, nucleic acid, pcr inhibition, based particle processor, particle processor, detailed contamination control, applied biosystem, time pcr, proprietary guanidinium thiocyanate lysis solution, reagent preparation step, rna, rtpcr, magnetic bead, extraction efficiency
Disclaimer
This protocol is provided for informational, diagnostic, and research purposes only. The USDA does not endorse any specific method, company, product, or reagent. It is not intended to supersede or be used as official USDA or U.S. Government determination or policy to replace institutional SOPs or regulatory requirements. The provided workflow was developed using the Applied Biosystems™ MagMAX™ CORE Nucleic Acid Purification Kit and associated equipment; however, equivalent kits or instruments might be used with appropriate validation. Users are responsible for ensuring compliance with all applicable biosafety and quality assurance guidelines. The authors make no representations that this document is complete, accurate or error free and assume no responsibility and will not be liable for any errors, omissions, damage or loss that might result from any use of this document, even if the information in the document is followed properly.
Abstract
This protocol describes a standardized, automated workflow for extracting nucleic acids (DNA/RNA) using the Applied Biosystems™ MagMAX™ CORE Nucleic Acid Purification Kit on a magnetic bead-based particle processor. The methodology employs a proprietary guanidinium thiocyanate lysis solution and magnetic microsphere beads to efficiently release, bind, and purify nucleic acids for high-sensitivity downstream applications such as real-time PCR (rtPCR) and sequencing. Integrated into the workflow is the VetMAX™ Xeno™ Internal Positive Control (IPC) to monitor extraction efficiency and PCR inhibition. Additionally, detailed contamination controls and reagent preparation steps are included to optimize performance in diagnostic and research laboratory environments.
Attachments
Guidelines
**Definitions**
- RNA Extraction: Method of purifying RNA from sample of choice
- DNA Extraction: The method of purifying DNA from sample of choice
- Extraction Control: The extraction control serves as the Negative and Positive Extraction Control in protocols where the Internal Extraction Control (IEC) is present. Successful performance of this control (Ct value undetermined or zero for disease target, Ct value falls within a pre-defined range for the IEC) indicates that neither contamination nor inhibition occurred during the nucleic acid purification process. Within this protocol, nuclease-free water serves as the Extraction Control.
- Internal Extraction Control (IEC): In this protocol, an exogenous internal positive control (Xeno™ RNA Control) will be used as the IEC. The Xeno™ RNA Control is added to the lysis buffer prior to nucleic acid purification thus all samples will contain Xeno™ RNA Control.
- Non-stick, RNase-free: Product free of enzymes that may degrade RNA with a siliconized coating to ensure RNA does not stick to treated surfaces.
- Quality Critical Reagents and Supplies: Supplies and reagents that must be purchased from the listed vendor in order to meet the requirements for quality assurance and quality control (QA/QC). These items may also require QA/QC testing prior to use.
**Personnel qualifications**
Personnel performing the procedure outlined in this protocol must be familiar with the preparation and proper handling of samples and reagents. It is advised that personnel be aware of the calibration, maintenance, and use of instruments included in this protocol before performing this protocol.
**Contamination prevention**
Laboratory gloves should be worn throughout the procedure and changed frequently if contamination of gloves or work area is suspected or when moving between laboratory areas. The use of gloves can help protect reagents and samples from contaminating agents/samples and/or cross-contamination which can affect results.
**Areas and Equipment**
Under traditional guidance having two separate preparation areas and equipment for the nucleic acid extraction procedure can be beneficial.
- One area designated as a "clean" area and used for the preparation of reagents. Within the clean area there should be a biological safety cabinet (BSC) or flow hood prep station designated for clean work only. The clean area is separated from the other area, ideally in a separate room and not just a separate BSC or flow hood. Virus, diagnostic samples, PCR amplicon, or template nucleic acid should never be introduced into the clean area. A designated "clean" set of calibrated pipettes and tips, nuclease-free water, tubes for reagent preparation, racks, and an ice container/cold block, for clean use only and should never leave this area. Exception being the pipettes when undergoing calibration.
- Separate cooler(s)/freezer(s) for clean reagent storage.
- Fresh gloves should always be worn when working with clean reagents and equipment.
- A second Sample/Extraction area which has a BSC or flow hood with a set of calibrated pipettes, 96-well magnetic particle processor, plate shaker and other equipment and reagents that are used for handling samples and extraction procedures.
**Preparation of equipment/instrumentation**
Calibration of Equipment
- Calibration of all equipment must be completed and certified according to the manufacturer’s instructions and/or the respective institution’s SOP.
Disinfection of Equipment
- Disinfection of equipment must be performed prior to beginning the extraction procedure and after performing the extraction process. Working areas should be sprayed/wiped with appropriate disinfectants, such as 10% bleach, commercial bleach wipes, Virkon® S, 70% Ethanol, or appropriate cleaning agent for sample and or pathogen being handled. Appropriate contact time for disinfectant should be used and then area should be wiped clean prior to placing sample(s) or reagents in the hood. Should the disinfectant be corrosive, hazardous, etc. a followed up 70% ethanol spray and wipe may be performed to remove any residues. Additionally, to appropriate nucleases/DNases/RNases treatment may be performed to the working surfaces and pipettes to further reduce possible contamination from occurring.
Materials
**Reagents**
- Applied Biosystems™ MagMAX™ CORE Nucleic Acid Purification Kit
(Product Code A32700: 100 reactions; Product Code A32702: 500 reactions)
- MagMAX™ CORE Lysis Solution, 50 mL; 275mL
- MagMAX™ CORE Binding Solution, 45mL; 220mL
- MagMAX™ CORE Magnetic Beads, 2.2mL; 11mL
- MagMAX™ CORE Proteinase K, 1.25mL; 5mL
- MagMAX™ CORE Wash Solution 1, 60mL; 300mL
- MagMAX™ CORE Wash Solution 2, 60mL; 300mL
- Elution Buffer, 12mL; 55mL
Note: While the MagMAX™ CORE Nucleic Acid Purification Kit is the suggested product for this extraction (based on volumes provided in the kit), the identical components are also found in other MagMAX™ family nucleic acid extraction products. It is acceptable to use the solutions/reagents from other MagMAX™ family products.
- VetMAX™ Xeno™ Internal Positive Control (IPC) RNA (10,000 copies/ µl) (1 x 1250 µl) (Life Technologies Cat No. A29761 or A29763)
- Internal extraction control (IEC): In this protocol, exogenous internal positive control (Thermo Fisher Scientific catalog No. A29761 or A29763, Xeno™ Internal RNA Control), 10,000 copies/µl stock
- Negative extraction control (NEC): For this protocol, nuclease-free water.
- Disinfectant: Bleach (5% sodium hypochlorite): Freshly prepared 10% bleach solution or commercial bleach wipes or equivalent disinfectant of choice appropriate for sample and purposes
**Supplies**
- S-blocks, 2.2 ml or equivalent.
- Microplates, 96-well, 200 µl or equivalent.
- Deep Well Plate, 96-well, 2.5 mL or equivalent
- Tip Comb/ Rod-Cover, 96-well, appropriate size for magnetic head.
- Pipette tips, aerosol resistant, nuclease-free
- Sterile containers such as sterile reagent reservoirs or conical tubes
- Snap cap microcentrifuge tubes, 1.5 mL or 2 mL
- Laboratory gloves, powder-free
- 96-well plate covers (examples parafilm, aluminum foil plate covers, reusable hard plastic plate covers, or adhesive PCR plate seals seals)
**Equipment**
- Calibrated pipettes and multichannel pipettes
- Orbital plate shaker (recommended, but optional).
- Ice bucket with ice, or cold block that has been cooled
- Vortex touch mixer or equivalent
- Refrigerator, 2-8°C
- Freezer (-30 ± 5°C, -20 ± 5°C)
- Ultralow Freezer (-70 ± 10°C, -80 ± 10°C)
- Dark Colored Specimen Transport Box or equivalent sealable and leak proof container
- Class II, HEPA filtered biological safety cabinets (BSC) (UV germicidal light optional)
- Magnetic Particle Processor (Examples: BioSprint® 96 (Qiagen), KingFisher™ 96 or KingFisher™ Flex™ (Thermo Scientific) or MagMAX™ Express (Ambion/Applied Biosystems) or equivalent)
Note: This protocol is built around the KingFisher™ Flex™ magnetic particle processor. This process also uses deep well plates and thus needs the deep well magnetic head. The Flex does not automatically come with deep well head and thus procurement of a deep well head will be required if one is not available.
Troubleshooting
Before start
Prior to beginning the extraction procedure annotation of all pertinent information should be performed. Included is a useful extraction worksheet (Nucleic Acid Extraction using the MagMAX™ CORE Nucleic Acid Purification Kit on a Magnetic Particle Processor Worksheet) for recording information. Be sure to include lot numbers and sample positions on the 96-well plate for record maintenance, ISO accreditation, and troubleshooting.
Programing the Magnetic Particle Processor
Ensure that the appropriate program(s) have been downloaded and uploaded to the magnetic particle processor. Downloadable files can be found in attached documents and/or on the manufacturer website which can be found in the references section.
- KingFisher Flex heated script: MagMAXCORE_Flex.bdz
Reagent Preparation
Notes:
- With the exception of the addition of the Xeno
™ Internal Control all reagent preparation should be, and is highly recommended, to be performed in the clean room. - It is highly recommended that prior to beginning the extraction procedure that the laboratorian annotate all the pertinent information in the extraction worksheet (Nucleic Acid Extraction using the MagMAX™ CORE Nucleic Acid Purification Kit on a Magnetic Particle Processor Worksheet) including but not limited to lot numbers, reagent kit numbers, expiration dates, etc., and sample positions on the 96-well plate.
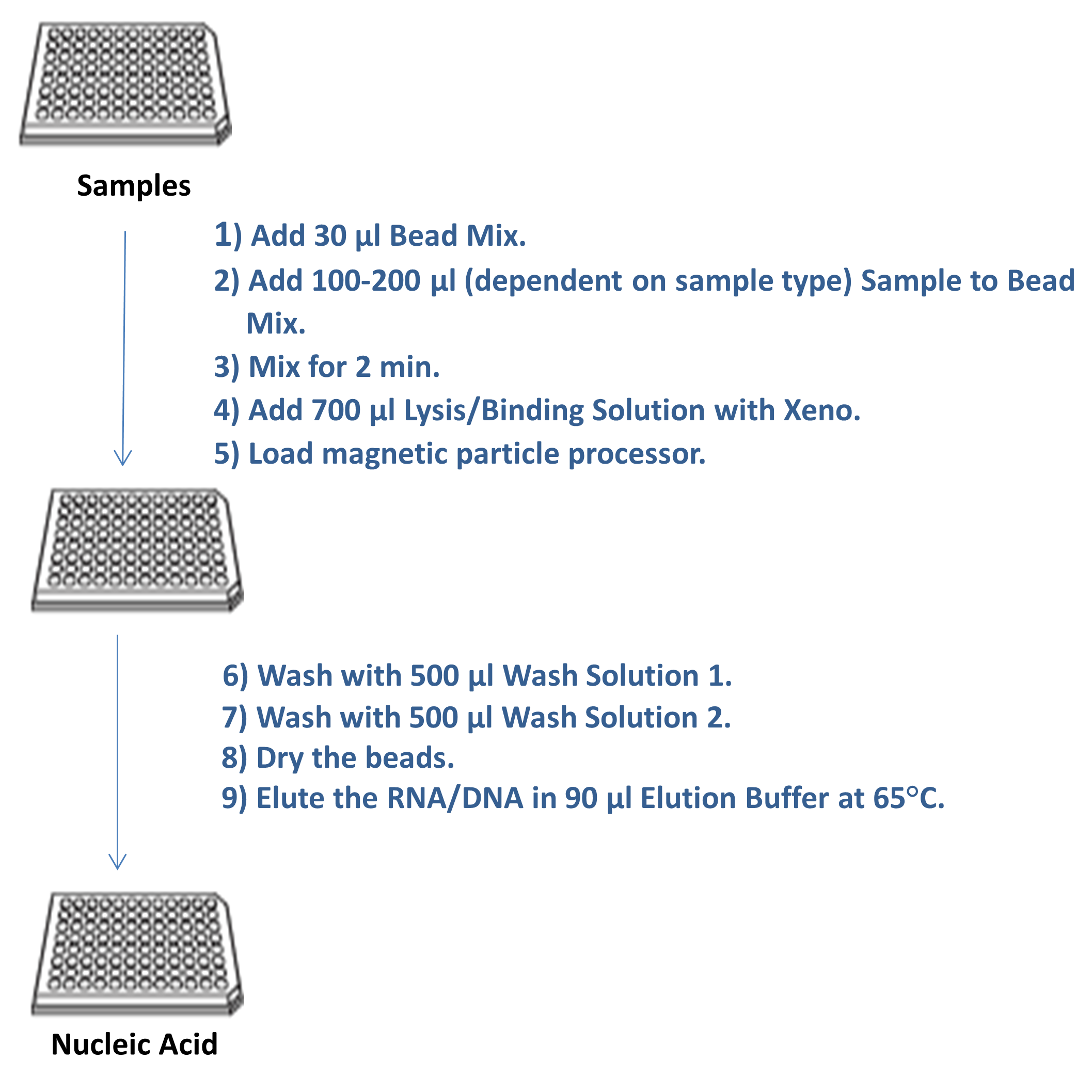
Plate Arrangement and Reagent Volumes
| Plate Number/Plate Type | Reagent/Solution Mix | Volume Per Well (μL) | |
| Plate 1 (Deep Well) | Proteinase K/Magnetic Bead Mix (PK/Beads) | 30 | |
| Sample | 100-200 | ||
| Lysis/Binding/Xeno™ Solution | 700 | ||
| Plate 2 (Deep Well) | Wash 1 | 500 | |
| Plate 3 (Deep Well) | Wash 2 | 500 | |
| Plate 4 (Deep Well or Standard Microplate) | Elution Buffer | 90 | |
| Plate 5 (Deep Well or Standard Microplate) | Tip Comb |
Table 1: Plate arrangement on the magnetic particle processor-96 turntable for MagMax™ CORE Nucleic Acid Purification. Reagent type, mix, and volumes.
In the clean room, prepare the reagent plates (except the lysis/binding/sample plate 1) using the volumes listed in table 1. Plates may be loosely covered with an appropriate seal to prevent cross contamination during movement from the clean room to the extraction room.
Aliquot the appropriate volumes of working stock solutions of Wash Solution 1 (500 µL per sample), Wash Solution 2 (500 µL per sample), and Elution Buffer (90 µL per sample) into separate sterile containers.
- Adding an additional 10-15% volume of each reagent is advised to cover losses from pipetting errors
- Using low bind pipettes can aid in the prevention of reagent loss and further reduce losses form pipetting
- It is advised to use foil seals or have some type of container to prevent light from reaching reagents. Wash 1 solution is light sensitive.
In the clean room add 20 µL of MagMax™ CORE Magnetic Beads to 10 µL of MagMax™ CORE Proteinase K Solution, per sample, in an appropriately sized sterile container.
Gently vortex PK/Beads mix and add 30 µL to each of the appropriate wells in a deep well 96-well plate.
While in the clean room add 350 µL of MagMax™ CORE Lysis Solution per reaction to 350 µL MagMax™ CORE Binding Solution per reaction to an appropriately sixed sterile container.
- The lysis solution is light sensitive, and it is advised to have a container to prevent light from reaching the solution. A simple and cheap container to prevent light can be a glove which the container can be placed in.
- Lysis/Binding Solution is stable for 24 hr after mixing.
- Lysis solution contains a guanidinium thiocyanate compound which is toxic, may cause skin burns or eye damage. Do not mix with bleach or acidic solutions.
Place all reagent plates into appropriate transport container and move to extraction/sample room.
Sample and Reagent Preparation
2m
In the extraction room remove samples from the freezer or refrigerator.
- It is advisable to ensure that all caps/lids are secured and clean on the outer surface of sample containers. Should the outer surface be dirty or the sample was not secured properly, and a leak has occurred clean the outer surface with bleach, bleach wipes, ethanol, or appropriate disinfectant. Make sure to use care and check labels and information on the tube before cleaning as disinfectant may remove writing or affect labels on the sample container. It is recommended that all containers be dried after cleaning.
- It is recommended to thaw frozen samples on ice if frozen or place onto a cold bock or on ice inside a BSC for use.
The lysis and binding solution should be prepared last to reduce the amount of time the sample is in the lysis buffer and to prevent loss of reagent due to evaporation and reduce light exposure.
Vortex each sample briefly at high speed to mix the sample thoroughly prior to addition to the 96-deep well plate.
- Keep in mind sample type and pathogen and fragility. Vortexing may not be appropriate in every situation.
For all sample types and controls, with the exception of whole blood, add 200 µL of sample to the deep well plate containing the aliquoted 30 µL PK/Beads in the appropriate wells.
- Note: A sample volume of 200 µL was found to improve sensitivity in some cases. If the volume of the original sample is insufficient it is acceptable to use 100 µL.
- It is recommended that the sample volume used to be indicated on the appropriate worksheet.
For whole blood samples, add 100 µL of sample to the 30 µL of PK/Bead mix in the appropriate wells.
Return any remaining samples and controls to ice until they can be frozen/refrigerated.
Once all samples and controls have been aliquoted seal the plate with aluminum foil seal (or equivalent material) to prevent cross contamination and allow the plate to be removed from the BSC/hood for shaking.
Decontaminate the plate and remove from the BSC.
Shake plate for 2 minutes on an orbital shaker at moderate speed (recommended).
- Not recommended but mixing via pipetting up and down is an acceptable method.
- If worried about sample aerosols or droplets near the well opening or on the seal wait a few seconds after shaking and/or centrifuge at low speed.
2m
To Lysis/Binding solution add 2 µL of Xeno™ RNA Internal Control per total number of reactions and then vortex the mixture.
Immediately after vertexing the Lysis/Binding/Xeno™ solution pipette 700 µL into each sample/control well and seal the plate.
- An additional shake can be performed to ensure full mixing of all solutions.
- A very rapid centrifuge can be performed to ensure that no sample is on the seal.
Proceed at once to the magnetic particle processor.
Running the Automated Program
30m
Turn on the machine. At the display screen on the instrument, locate and select the desired program name. Push start.
The machine will then re-orient the turntable and check the head. The turntable will then orient itself so that the plates may be loaded in appropriate order of the chosen program. The instrument will display a message at the display screen to load plate "X" (X indicates the plate name or number of the plate).
Open the sliding door to be able to insert the plates. Insert the proper plate in the presented slot and click the start button to move onto the next plate. The turntable will move to the next plate section each time you press the start button.
- Total time may slightly vary and may take between 25 to 30 minutes. This is also dependent on program used and if additional wash steps have been incorporated into the program.
30m
After the last plate is loaded close the sliding door. Press the start button again to initiate the program. At the end of the program, remove Elution plate, Plate 4. It should be directly positioned at the front of the sliding door.
Seal plate immediately for transportation/movement with a new cover.
To remove remaining plates that had been used simply press the start button and the turntable will move to the next plate. Keep pressing start to move through all plates.
Press Stop after all plates have been removed and wait for the turntable to stop moving and then power down the machine.
Your Elution plate now contains your extracted DNA/RNA. Either use the extracted nucleic acid or store at -60°C or less for future use. Samples may be aliquoted into other plates or stored into individual tubes if desired for use.
Disinfect all exposed surfaces of the machine after each run and record any run data and sample information required on the appropriate forms.
Protocol references
MagMAX‱ CORE Nucleic Acid Purification Kit with reference downloads for KingFisher‱Flex‱ programming specifications:
Applied Biosystems‱ MagMAX‱ CORE Nucleic Acid Purification Kit User Guide
Thermo Fisher Scientific. Available at:
KingFisher‱ Flex Magnetic Particle Processor User Manual
Thermo Fisher Scientific. Available at:
BindIt Software for KingFisher Instruments User Manual
Thermo Fisher Scientific. Available at:
Applied Biosystems MagMAX‱ Express Instrument Comparisons
Thermo Fisher Scientific. Available at:
VetMAX‱ Xeno‱ Internal Positive Control RNA Product Information
Thermo Fisher Scientific. Available at:
For Programing the magnetic particle processor any .kf2 files can only be used directly with the KingFisher 96 and MagMAX Express-96 platforms. If using a different magnetic particle processor, please refer to the included software user manual for specific instructions and/or refrenct technical notes. Should files for KingFisher Flex, BindIt software be required. Contact Thermo Fisher Scientific to request/buy appropriate software.



