Jul 29, 2025
- Maria Dimitriu1,
- Leonard Steg1,
- Rodrigo Arzate1,
- Isabelle Mansuy1
- 1ETH Zurich

External link: https://doi.org/10.1038/s41598-025-13316-5
Protocol Citation: Maria Dimitriu, Leonard Steg, Rodrigo Arzate, Isabelle Mansuy 2025. NanoTag. protocols.io https://dx.doi.org/10.17504/protocols.io.kxygx34zkg8j/v1
Manuscript citation:
Dimitriu MA, Arzate-Mejía RG, Steg LC, Germain P, Mansuy IM (2025) NanoTag is an improved method to map interactions between DNA and proteins not requiring IgG. Scientific Reports 15(). doi: 10.1038/s41598-025-13316-5
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: December 13, 2023
Last Modified: July 29, 2025
Protocol Integer ID: 92256
Keywords: histone mark h3k4me3 genome, tagged protein, nanotag result, nanotag, ctcf in mouse embryonic stem cell, wide profiling of dna, chromatin, most current methods for genome, nanotag background, proteins such as chip, mouse embryonic stem cell, gene regulation, bound protein, binding protein, protein interactions in cell
Funders Acknowledgements:
ETH Zurich
Grant ID: ETH-10 15-2
Abstract
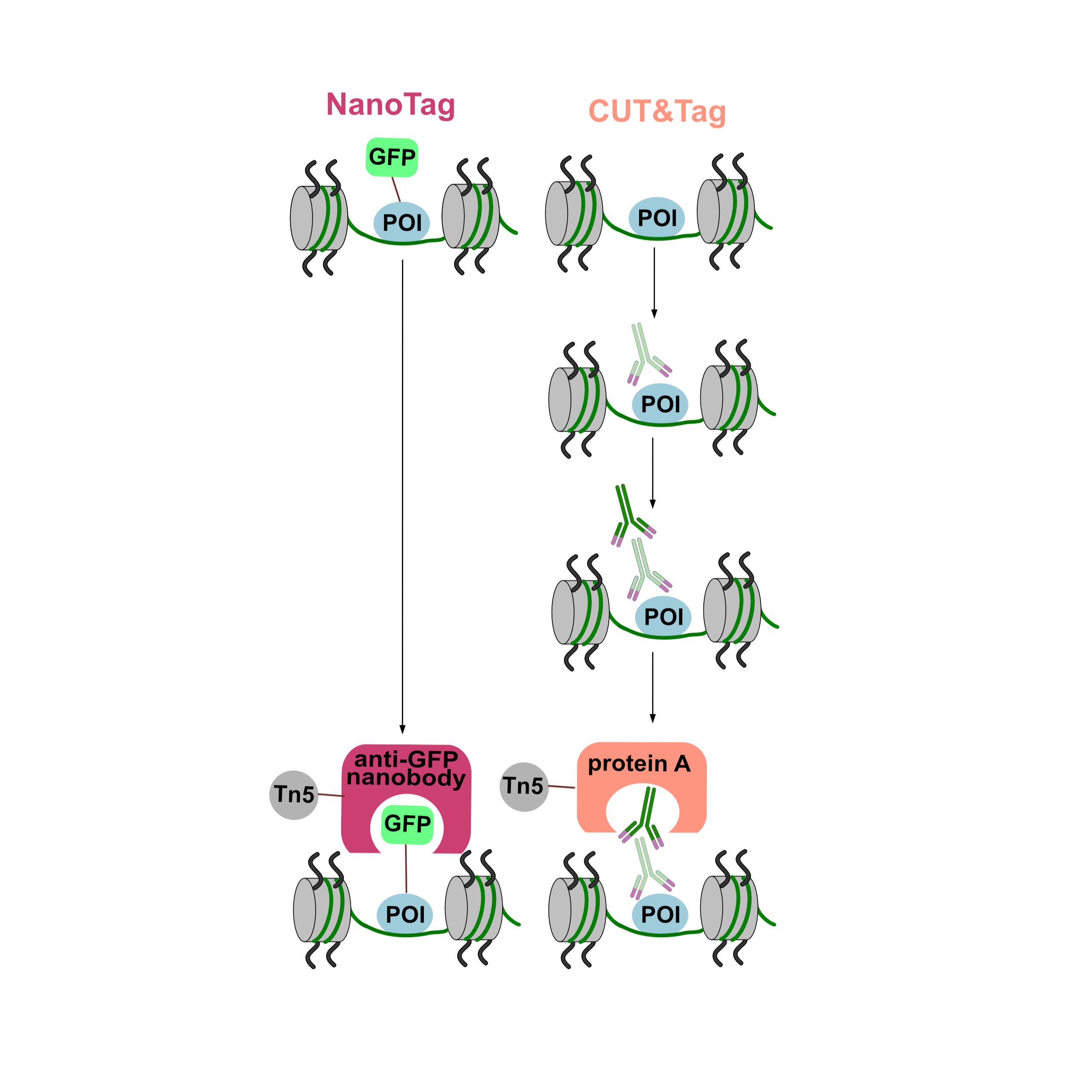
Background: Genome-wide profiling of DNA-protein interactions in cells can provide important information about mechanisms of gene regulation. Most current methods for genome-wide profiling of DNA-bound proteins such as ChIP-seq and CUT&Tag use conventional IgG antibodies to bind the target protein(s). This limits their applicability to targets with available high affinity and specificity antibodies and prevents their use for other targets. Here we describe NanoTag, an IgG-free method derived from CUT&Tag to profile DNA-protein interactions. NanoTag is based on a fusion between an anti-GFP nanobody and Tn5 transposase that can map GFP-tagged proteins associated with chromatin in a fast, cost-effective and animal-free manner.
Results: We used NanoTag to indirectly profile the histone mark H3K4me3 genome-wide via its binding partner TATA box-binding protein-associated factor 3 (TAF3) and the transcription factors Nanog and CTCF in mouse embryonic stem cells (mESCs). NanoTag results show high inter-replicate reproducibility, high signal-to-noise ratio and strong correlation with CUT&Tag datasets, validating its accuracy and reliability.
Conclusions: NanoTag provides a novel, flexible and cost-effective IgG-free method to generate high resolution DNA-binding profiles in cells and tissues.
Materials
This protocol requires having prepared GFP nanobody-Tn5 transposomes with a known concentration.
For details on preparation of the protein and transposomes, see the last section of the protocol.
Materials required for the protein purification:
HEGX buffer
20 mM HEPES (pH 7.5), 800 mM NaCl, 1 mM EDTA, 0.1% Triton X-100, 10% Glycerol, protease inhibitors
Tn5 dialysis buffer
100 mM HEPES (pH 7.2), 200 mM NaCl, 0.2 mM EDTA, 2mMDTT, 0.2% (v/v) Triton X-100, 20% (v/v) glycerol
Oligonucleotides
Tn5ME-A: TCGTCGGCAGCGTCAGATGTGTATAAGAGACAG
Tn5ME-B: GTCTCGTGGGCTCGGAGATGTGTATAAGAGACAG
Tn5-ME-rev: /5Phos/CTGTCTCTTATACACATCT
Equipment needed for the purification of GFP nanobody-Tn5:
42°C water bath
Shaker incubator
5-liter Erlenmeyer flasks
500-ml centrifuge tubes
Refrigerated centrifuge with swinging-bucket rotor
Laboratory balance
Probe sonicator with 10-mm tip (e.g., Branson Sonifier)
15- and 50-ml conical centrifuge tubes
Econo-Pac Chromatography Columns (BioRad, cat. no. 7321010)
Membrane Dialysis, 12 to 14 kD (Spectrum Labs, cat. no. 132706)
PCR tubes
NanoTag buffers:
Wash buffer
20 mM HEPES pH 7.5, 150 mM NaCl, 0.5 mM Spermidine, protease inhibitors
Binding buffer
20 mM HEPES-KOH pH 7.9, 10 mM KCl, 1 mM CaCl2, 1 mM MnCl2
Dig-wash buffer
0.05% Digitonin, 2 mM EDTA in Wash buffer (nanobody-Tn5 will be diluted in this buffer and added to samples)
Dig-med buffer
20 mM HEPES pH 7.5, 300 mM NaCl, 0.5 mM Spermidine, 0.01% Digitonin, protease inhibitors
Tagmentation buffer
10 mM MgCl2 in Dig-med buffer
Elution buffer
1 mM Tris-HCl pH 8.0, 0.1 mM EDTA
Equipment needed for performing the NanoTag protocol and library preparation:
Centrifuge
Nutator
End-over-end rotator
Thermocycler
Lightcycler
Thermomixer/water bath
Troubleshooting
Cell collection
17m
Remove media from culture dish and wash with D-PBS.
2m
Dissociate cells using Accutase and count cells.
Note
Use of trypsin to dissociate cells will disrupt the binding of the cells to the concanavalin A beads in subsequent steps, so use of Accutase is crucial.
10m
Wash cells with 1 mL Wash buffer and resuspend cells at 1 million cells/mL of Wash buffer. 400 µL per sample. Transfer in a 2 mL tube. Use 400,000 cells per NanoTag sample.
5m
Take 400 µL of cell suspension (equivalent to 400k cells) per sample.
Binding cells to beads
15m
Prepare concanavalin A beads:
Withdraw 10 uL of bead slurry per sample (i.e. use 170 µL for 16 sample), transfer it into 1.6 mL Binding buffer and mix by pipetting.
1m
Place tube on magnet until solution is clear. Remove the liquid, then remove tube from magnet and resuspend in 1.5 mL Binding buffer. Mix by pipetting.
2m
Place tube on magnet until solution is clear, then remove the liquid and resuspend the slurry in the original volume (170 µL for 16 samples) of Binding buffer. Keep the beads at room temperature until cells/nuclei are ready.
Add the correct amount of beads to the corresponding amount of cell suspension (10 µL of bead slurry and 400 µL of cell suspension) per sample while gently vortexing.
Mix by pipetting and incubate on an end-over-end rotator for 00:15:00 . During the incubation, continue with step 8.
15m
Tagmentation
1h 5m
During incubation of step 6 mix the correct amount of GFP nanobody-Tn5 transposomes (2 µL per sample for 23 micromolar (µM) protein) and Dig-wash buffer (100 µL per sample).
After the incubation from step 6 aliquot the bead-cell suspension to the corresponding amount of tubes.
Place tubes on the magnet until solution is clear. Remove the liquid, remove tubes from magnet and resuspend beads in 100 µL of cold GFP nanobody-Tn5-containing Dig-wash buffer by gently pipetting.
Incubate on a nutator Overnight at 4 °C to allow the GFP nanobody-Tn5 to bind to its target.
The next day place tubes on the magnet until solution is clear and remove the liquid. Wash beads with 1 mL of Dig-med buffer. Incubate on nutator for 00:05:00 .
5m
Repeat step 11 once to remove any anti-GFP nanobody-Tn5 protein that is unspecifically bound.
Resuspend beads in 300 µL of Tagmentation buffer and incubate at 37 °C for 01:00:00 .
1h
Tagmentation stop and DNA extraction
1h 14m
Add 10 µL 0.5M EDTA, 3 µL 10% SDS and 2.5 µL of 20 mg/mL Proteinase K to each sample. Vortex tubes on full speed for 2-3 seconds.
Incubate at 55 °C for 01:00:00 .
1h
Add 300 µL Phenol:Chloroform:Isoamyl Alcohol and full speed vortex for 2s.
Transfer solution to a phase-lock tube and centrifuge 16000 x g, 00:03:00 .
3m
Transfer the aqueous (top) layer to a fresh 1.5 mL tube containinng 750 µL 100% ethanol, pipet up and down to mix and keep tubes On ice .
Centrifuge 16000 x g, 4°C, 00:10:00 . Pour off the liquid and quickly drain on a paper towel.
10m
Add 1 mL 100% ethanol and centrifuge 16000 x g, 4°C, 00:01:00 . Pour off the liquid and let tubes dry on a paper towel for about 10 minutes.
1m
When tubes are dry add 25 µL Elution buffer and full-speed vortex to dissolve the DNA.
Store tagmented DNA at -20 °C .
Library preparation
5m
To identify the optimal amount of cycles to perform during the library amplification, perform a qPCR:
Create a master mix by adding 2 µL of i5 primer and 2 µL of i7 primer, 0.15 µL 100X SYBR Green dye, 0.15 µL water and 0.85 µL NEBNext PCR Master mix per sample.
Add 12.5 µL of master mix to each well of a qPCR plate. Add 2.5 µL of tagmented DNA to each well and run the qPCR using the following program:
Cycle 1: 72 °C for 5 min (gap filling)
Cycle 2: 98 °C for 30 sec
Cycle 3: 98 °C for 10 sec
Cycle 4: 63 °C for 30 sec
Cycle 5: 72 °C for 60 sec
Repeat Cycles 3-4-5 20 times
Check the amount of cycles necessary to achieve 1/3 of the maximum fluorescence for each sample and perform library amplification using that amount of cycles.
Note
The amount of cycles varies by target but usually between 18 and 21 cycles are necessary to ensure a minimum library concentration of 2 nM after clean-up.
20231221_3edit.jpg
Once the amount of cycles has been determined according to step 23 amplify libraries:
Transfer 21 µL of tagmented DNA from each sample into a PCR tube. Add 2 µL of i5 index, 2 µL of i7 index (use different i5 and i7 combinations for each library) and 25 µL of NEBNext PCR master mix, mix and briefly spin down the sample then perform the following PCR program:
Cycle 1: 72 C for 5 min (gap filling)
Cycle 2: 98 °C for 30 sec
Cycle 3: 98 °C for 10 sec
Cycle 4: 63 °C for 10 sec
Repeat Cycles 3-4 for the amount of times determined in step 23
72°C for 1 min and hold at 8 °C
Add 60 µL AMPure beads to each library. Pipet up and down 10x to mix. Briefly spin down and incubate for 10 min. Place on magnet and allow solution to clear.
Remove the liquid and wash beads 2x with 200 µL of 80% ethanol with the tubes still on the magnet. After the second wash remove the liquid using 2 pipetting steps.
Add 20 µL Tris-HCl pH 8.0 and full-speed vortex. Incubate for 5 min. Pleace on magnet and allow solution to clear. Transfer liquid to fresh tubes.
Check the library quality using a fragment analyzer (e.g. Tapestation).
Note
Example of Tapestation profile of a NanoTag library targeting TAF3 in mESC.
Sequencing
NanoTag libraries can be sequenced using Illumina platforms. Before sequencing, serially dilute the libraries to equal molarity and pool to the desired molarity according to the sequencing platform specifications. We recommend adding 10% PhiX spike-in to the pool and performing paired-end sequencing using 50bp reads.
Preparing GFP nanobody-Tn5 transposomes
Transform LysY/lq competent E. coli with pMD1 plasmid encoding GFP nanobody-Tn5 with a intein-CBD tag.
Inoculate 2L of LB (+100ug/mL Ampicilin) with 1 colony and induce overexpression of GFP nanobody-Tn5 with 0.25 mM IPTG overnight at 18 °C .
The next day collect the bacterial medium, centrifuge and freeze the pellet.
Resuspend the pellet in 40 mL HEGX buffer and sonicate for 2.5 min using 10s cycles at 70% amplitude.
Centrifuge for 30 min at 20,000xg.
Collect the supernatant and add 1.1 mL of 10% PEI (pH 7.5) to 40 mL of supernatant.
Centrifuge for 20 min at 20,000xg.
Bind the supernatant tp chitin beads for 2h at 4 °C in a disposable column. Wash thoroughly with 10 column volumes of HEGX buffer.
Induce cleavage of the GFP nanobody-Tn5 from its intein tag by incubating the column overnight at 4 °C in HEGX buffer + 100 mM DTT.
Collect the eluate. Prepare the oligonucleotides and add the oligonucleotides to the eluate and incubate for 1h at 30 °C .
Resuspend oligonucleotides at 200 uM in water.
Incubate for 5 min at 95 °C in a thermocycler and cool down over
Dialyze overnight in Tn5 dialysis buffer.
Concentrate the protein using 30kDa Amicon centrifugation filter. Store the concentrated protein in Tn5 storage buffer, prepare aliquots, flash freeze in liquid nitrogen and store at -80 °C .