Nov 10, 2025
Multicolor Flow Cytometry Approach for Analysis of Stromal and Thymocyte Cell Populations in the Mouse Thymus
- Tamara Menendez Medina*1,
- Zaima Mazorra Herrera*1,
- Cinthia Mariel Olexen1,
- Gisela Maria Suarez Formigo1
- 1Abu Dhabi Stem Cell Center, Abu Dhabi, United Arab Emirates. Correspondence: [email protected], [email protected], [email protected], [email protected]. * Both authors contributed equally.

Protocol Citation: Tamara Menendez Medina*, Zaima Mazorra Herrera*, Cinthia Mariel Olexen, Gisela Maria Suarez Formigo 2025. Multicolor Flow Cytometry Approach for Analysis of Stromal and Thymocyte Cell Populations in the Mouse Thymus. protocols.io https://dx.doi.org/10.17504/protocols.io.n2bvjem6bgk5/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: November 06, 2025
Last Modified: November 10, 2025
Protocol Integer ID: 231663
Keywords: Mice thymus, Thymus flow cytometry, Thymocytes, Thymus epithelial cells , thymus stromal cell, thymocyte cell populations in the mouse thymus, mature thymocyte, characterization of thymocyte, thymocyte cell population, thymocytes development, selecting thymocyte, positive selection of thymocyte, developing thymocyte, regarding thymocyte, thymic epithelial cell, thymocyte, cell fate decisions in the thymus, late stage of thymocytes differentiation, thymus organogenesi, later stages of thymocyte development, thymocytes differentiation, thymocyte development, necessary support for thymocyte development, sp thymocyte, thymic cell population, dn cells within the thymus, indispensable for thymus organogenesi, impaired thymocyte differentiation, consistent with impaired thymocyte differentiation, thymic medulla, thymus medulla, influence on thymocyte outcome, stromal cell component, stromal cell, thymocyte outcome, hallmark features of thymic aging, associated thymic involution, functional thymus, lymphocyte develop
Abstract
The thymus is a primary lymphoid organ which contains stromal cell components that provide the necessary support for thymocyte development and maturation. Thymus stromal cells include, among others, thymic epithelial cells (TEC), endothelial cells, mesenchymal/fibroblast cells, and hematopoietic populations such as dendritic cells, B cells, and macrophages. TECs are particularly critical through all the stages of T cell development, contributing to the recruitment of hematopoietic precursors from the bone marrow, directing lineage specification, supporting proliferative expansion, and orchestrating both positive and negative selection processes (Cepeda & Griffith, 2018) (Han & Zúñiga-Pflücker, 2021).
Anatomically, the thymus is organized into two distinct regions: the cortex in the outer zone, which contains the cortical TECs (cTECs) and the medulla in the inner zone, containing the medullary TECs (mTECs). cTECs are primarily involved in the positive selection of thymocytes. In contrast, mTECs participate in the negative selection, promoting central tolerance and supporting the development of Foxp3⁺ regulatory T cells and natural killer T cells (Han & Zúñiga-Pflücker, 2021) (Nitta & Suzuki, 2016).
A young and fully functional thymus is essential for generating a pool of diverse naïve T cells, which supports robust immune responses. However, during aging, the thymus undergoes progressive involution characterized by structural and molecular modifications. These age-associated changes lead to a marked decline in the production of newly generated naïve T cells. In compensation, a homeostatic expansion of existing T cell populations occurs, resulting in reduced diversity within the peripheral T cell repertoire. Functionally, this adaptation contributes to diminished responsiveness to pathogens, increased susceptibility to autoimmune conditions, and impaired tumor surveillance (Liang et al., 2022) (Han & Zúñiga-Pflücker, 2021).
The study of the thymic microenvironment and its influence on thymocyte outcome during aging provides valuable insights into the structural and functional changes that occur with senescence. One of the hallmark features of thymic aging is the decline in TEC cellularity, affecting both cTECs and mTECs subsets. TECs can be phenotypically identified with the pan-epithelial marker CD326 (EpCAM) within the CD45- thymic cell population. Further discrimination between TEC subsets could be achieved using the CD205 marker. cTECs subset could be characterized as CD45-CD326+CD205+, whereas CD45-CD326+CD205- cells correspond to mTECs (Jain & Gray, 2014). Aging is also associated with a reduction in MHC class II expression on TECs, reflecting a decline in TEC antigen-presenting ability (Gray et al., 2006). Within the TEC population, two subgroups, MHCIIlow and MHCIIhigh, can be distinguished based on the expression levels of MHC class II molecules (Jain & Gray, 2014) (Xing & Hogquist, 2014). A key molecular regulator of TEC function is the transcription factor FoxN1, which is indispensable for thymus organogenesis and TEC maintenance in adults. FoxN1 expression declines progressively with age, contributing to thymic involution and reduced naïve T cell output (Rode et al., 2015). Overexpression of FoxN1 has been shown to mitigate age-related thymic changes, restoring thymic mass, architecture, and the naïve T cell population in the spleen (Zook et al., 2011).
Thymic medulla, besides containing mTECs, is populated by hematopoietic antigen presenting cells (APCs) including dendritic cells, B cells, and macrophages. Through expression and presentation of self-antigens, these APCs contribute to the process of shaping the normal T cell repertoire by negatively selecting thymocytes recognizing self-antigens. Senescence studies have demonstrated age-associated declines in APCs function and cell counts (Ki et al., 2014) (Cepeda & Griffith, 2018) (Wedemeyer & Griffith, 2025) (Van Dommelen et al., 2010).
Thymocytes development starts from the double negative (DN) to simple positive (SP) T cells. At first, T cells are CD4-CD8- (DN), in the second stage they are double positive CD4+CD8+ (DP) and finally CD4+CD8- or CD4-CD8+ (SP) in the third stage. After positive selection, SP CD4+ or CD8+ T cells migrate to the thymus medulla to go through negative selection mediated by medullar TECs and APCs, where autoreactive SP T cells are deleted by apoptosis. This process of thymopoiesis results in population of peripheral blood and lymphoid sites with naive CD4+ and CD8+T cells and CD4+ regulatory T cells (Tregs) (Liang et al., 2022).
Early T-lineage progenitors (ETPs) frequency declines with aging, and their potential ability to reconstitute the thymus is also reduced (Min et al., 2005). Both DN and DP population cell numbers are significantly modified with aging. DN subset frequency increases in aged mice compared with young, whereas the percentage of DP (CD4+CD8+) subpopulations significantly diminishes with age. Additionally, thymic aging is concomitant with the abnormal accumulation of CD3+ DN cells within the thymus (Aw et al., 2010). Aging also interferes with later stages of thymocyte development. DP and SP thymocytes in aged mice display deregulated CD3 expression, which may lead to attenuated TCR-dependent stimulation (Aw et al., 2010). Consistent with impaired thymocyte differentiation in the aged thymus, T-cell receptor excision circles (TRECs) within the thymus also significantly decline with aging in mice and humans (Ortman et al., 2002).
Regarding thymocytes, the member of the high mobility group (HMG)-Box transcription factors TOX-1 has been identified as a mediator of thymic T cell expansion and the development of lymphoid inducer (Scott et al., 2019). The pattern of TOX expression in the thymus is highly regulated during the positive selection process. Downregulation of TOX-1 is observed in a later stage of positive selection, as cells take on a more mature SP phenotype (Wilkinson et al., 2002). Data about TOX expression in murine thymus during aging is scarce. Recently, it was published that Recent Thymic Emigrants (RTE) are distinguished by the expression of SOX4, IKZF2, and TOX and CD38 protein in humans. These cells significantly decrease with age (Bohacova et al., 2024). Interestingly, some TOX members have been related to T cell exhaustion in the periphery. In mice, TOX is required for the epigenetic remodeling and survival of exhausted CD8+ T cells in the chronic infection context (Yao et al., 2019). In contrast, a recent publication in human settings showed that TOX expression is related to anti-viral effective T cells (Sekine et al., 2020).
Different markers related to thymocytes development, survival, and migration have been studied in murine settings. In this case, L-selectin (CD62L) is a cell adhesion molecule that is expressed on most circulating leukocytes. The expression of CD62L is increased in the late stage of thymocytes differentiation and allows mature thymocytes to enter from circulation into lymph nodes (Weinreich & Hogquist, 2008).
Another important marker related to thymus development is the CD127 molecule (IL-7Ra chain). IL-7 augments survival of TN and SP thymocytes and is also essential for γδ T cells and thymic DC development (Lynch et al., 2009). Lymphocyte development is severely impaired in mice lacking IL-7 or IL7 receptor (IL-7R) or treated with
anti-IL-7 antibodies (Peschon et al., 1994). In both syngeneic and allogeneic mouse models, administration of IL-7 leads to enhanced T-cell reconstitution through increased thymic T-cell development and homeostatic proliferation, as well as decreased T-cell apoptosis but increased T-cell function (Alpdogan et al., 2003).
Regulatory T-cells (Tregs) were identified as sentinels of the immune response, keeping aberrant/exaggerated immune reactions in balance. After CD4 lineage commitment, some CD4 single-positive (SP) thymocytes, upon TCR stimulation, can develop into CD25+Foxp3+ Treg cells. The absolute number of Tregs in the thymus decreases with age, reflecting the reduction in cellularity that accompanies age-associated thymic involution (Chiu et al., 2007).
Costimulatory and coinhibitory molecules affect cell fate decisions in the thymus by enhancing or dampening signaling through the TCR. Studies have shown that PD-1 is expressed by developing thymocytes (Nishimura et al., 1996). A recent publication has reported that PD-1 limits the production of IL-2 within the thymus, thereby restraining the development of Tregs (Caruso et al., 2024).
The present protocol describes the development of flow cytometry panels tailored for the identification and characterization of thymocytes and thymus stromal cells. The panels could be useful to compare young and aged mice in senescence studies, offering valuable insights into age-associated changes in thymic architecture and immune function.
Materials
Biological species
- C57BL/6J mice (The Jackson Laboratories)
Reagents and solutions
- RPMI 1640 Medium GlutaMAX
- Fetal Bovine Serum, heat inactivated
- Bovine serum albumin solution 30%
- Phosphate Buffered Saline (PBS)
- Penicillin-Streptomycin for cell culture
- PBS/ Azide: PBS pH7.2 with Sodium Azide 0.01%
- FACS buffer: 1% BSA in PBS
- sRPMI: RPMI 1640 culture medium supplemented with 10% Fetal bovine serum and 1% Penicillin/Streptomycin
- Trypan blue solution
- PerFix-nc kit (Beckman Coulter, catalogue number B10825).
- OptiLyse C, No-Wash Lysing Solution (Beckman-Coulter, catalogue number A11894).
- LIVE/DEAD Fixable Aqua Dead Cell Stain Kit (Invitrogen, catalogue number L34965).
- TruStain FcX PLUS (anti-mouse CD16/32) antibody (clone S17011E) (Biolegend, catalogue number 156603).
- Rat anti-mouse, PE-Texas Red/ CD45 monoclonal antibody (Clone 30-F11) (Invitrogen, catalogue number MCD4517).
- Rat anti-mouse, PE/CD205 (DEC-205) monoclonal antibody (clone NLDC-145) (Biolegend, catalogue number 138213).
- Rat anti-mouse, eFluor 450/ CD326 (EpCAM) monoclonal antibody (clone G8.8) (Invitrogen, eBioscience, catalogue number 48-5791-82).
- Rat anti-mouse, APC/IA-IE (MHC class II) monoclonal antibody (clone M5/114.15.2) (BioLegend, catalogue number: 107614).
- Rabbit anti-human (reactivity to Human, Mouse and Rat), unconjugated FOXN1 polyclonal antibody (Bioss, catalogue number BS-6970R).
- Goat anti-rabbit, Alexa Fluor 488-IgG H&L (Abcam, catalogue number ab150077).
- Rat anti-mouse Brilliant Violet 785/ CD3 monoclonal antibody (clone 17A2) (Biolegend, catalogue number 100232)
- Rat anti-mouse PCY7/CD62L monolonal antibody (clone MEL-14) (Invitrogen, catalogue number 25-0621-82)
- Armenian hamster anti-mouse eFluor450/ PD-1 monoclonal antibody (clone J43) (eBioscience, catalogue number 48-9985-82)
- Rat anti-mouse PE/CD25 monoclonal antibody (clone PC61.5) (Invitrogen, catalogue number 12-0251-82)
- Rat anti-mouse Qdot605/ CD4 monoclonal antibody (clone RM4-5) (Invitrogen, catalogue number Q10092)
- Rat anti-mouse BV650/ CD8 monoclonal antibody (clone 53-6.7) (Biolegend, catalogue number 100742)
- Rat anti-mouse CD127-AF700 monoclonal antibody (clone A7R34) (eBioscience, catalogue number 56-1271-82)
- Rat anti-mouse eF660/ Tox1 monoclonal antibody (clone TXRX10) (Invitrogen, catalogue number 50650282)
- Rat anti-mouse AF488/ FoxP3 monoclonal antibody (clone FJK-16s) (Invitrogen, catalogue number 53577382)
Laboratory supplies
- Curved and straight forceps
- Dissection scissors
- Pippete micro tip with filter, sterile (20 μL, 200 μL and 1000 μL)
- Disposable CultureTubes, Non-Sterile, 12 x 75 mm tubes, Polypropylene, 5 mL
- Strainer Cap for FlowTubes with 35µm strainer mesh
- 15 mL Conical Sterile Polypropylene Centrifuge Tubes
- 50 mL Conical Sterile Polypropylene Centrifuge Tubes
- 1.5 mL microcentrifuge tubes
- 96-well U-bottom cell culture plate
- 70 um Cell Strainer for 50ml Tube
- 0.22 μm syringe filter
- Biohazard bags 30 mL sterile plastic containers
- 3-mL sterile syringe
- Serological pipettes 5ml
Equipment
- Adjustable micropipettes
- Serological pipette controller
- Refrigerated Centrifuge with rotor for 1.5 to 2 mL microtubes
- Refrigerated Centrifuge with rotors for 15- and 50-mL conical tubes
- Centrifuge with rotor for ELISA plates
- 4°C laboratory Refrigerator
- -20°C Laboratory Freezer
- -80°C Laboratory Freezer
- Water bath
- Vortex Mixer
- Biosafety cabinet, Class II, Type A2
- Flow Cytometer (Beckman Coulter DxFLEX)
Software
- FlowJo (BD Biosciences, Version 10)
Troubleshooting
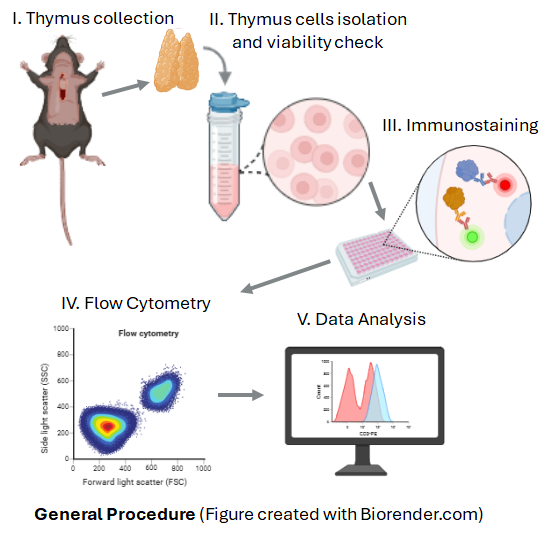
I- Thymus collection
Euthanize mice in accordance with national regulations on laboratory animal welfare, in accordance with internationally accepted standards for the care and use of laboratory animals, and after approval of the corresponding local ethic committees.
For the present protocol mice were anesthetized with an intraperitoneal injection of ketamine (120 mg/kg) and xylazine (16 mg/kg), a dosage commonly used for deep anesthesia prior to terminal procedures in rodent models (Cossarizza et al., 2021). Once unresponsiveness was confirmed, euthanasia was performed by decapitation (Leary et al., 2020). The procedure was carried out by a trained veterinary physician and following national and institutional animal care guidelines.
Note: Maintain sterile technique throughout all the procedure to prevent contamination. Always follow institutional safety guidelines when handling biological materials.
Position the mouse on its back and secure each limp to the dissection board. Spray with 70% ethanol to sterilize the mouse body.
Open the thoracic cavity with scissors, carefully exposing the heart and the thymus without cutting major blood vessels.
Extract the thymus gently using forceps, removing surrounding fat and connective tissue.
Transfer the thymus to a plastic container pre-filled with sRPMI medium and wash twice with sRPMI. Keep the organ in sRPMI at 4 °C until processing.
II- Thymus cell isolation and preparation for flow cytometry
Place a 70 μm cell strainer on top of a 50-mL tube. Place the thymus onto the strainer and gently press it through using the plunger of a 3-mL syringe. Rinse with sRPMI in between the mashing to facilitate dissociation. Cells will pass through the strainer, while clumps and debris are retained. At the end, rinse the strainer with sRPMI, adding up to a total volume of 10 mL.
Centrifuge the cell suspension at 300 x g for 5 minutes at 2-8°C. Discard the supernatant and gently dissociate the cell pellet in the remaining liquid. Add 5 mL of sRPMI to the resuspended cells.
Perform cell count and viability assessment using Trypan blue exclusion staining. Prepare a 1:1 dilution of the cell suspension with Trypan blue. Load the diluted sample onto a hemocytometer, count viable (unstained) and non-viable (blue-stained) cells. Calculate the total cell count and viability percentage.
Note: Before starting the immunostaining, all antibodies should be titrated to determine the optimal working concentration. For each antibody prepare additional control samples: no stain, single antibody staining, multi-antibody staining, and fluorescence minus one (FMO) staining controls. Fluorochrome combinations should be carefully selected based on the instrument and available lasers. Consultation with flow cytometry core personnel is strongly recommended to minimize spectral overlap. Fluorochrome combinations used in the present protocol were optimized for use on cytometers with at least three lasers: blue (488 nm), red (640 nm), and violet (405 nm).
III- Immunostaining
LIVE/DEAD staining protocol
Centrifuge the cells at 300 x g for 5 minutes. Discard the supernatant and resuspend the cell pellet with PBS at a concentration of 107 cells/mL.
Transfer the cells to a 96-well U-bottom plate, dispensing 200 µL per well (2 x 106 cells per well). Add 50 µL PBS to each well. Centrifuge at 300 x g for 5 minutes and discard the supernatant.
Repeat the wash by adding 250 µL of PBS to each well, centrifuging and discarding the supernatant.
Resuspend the cell pellet in 200 µL of a 1:100 dilution (in PBS) of the Live Aqua Dead-Fixable Cell Staining Reagent (and previously prepared in DMSO following the manufacturer recommendations).
Incubate the cells for 30 minutes at 4°C, protected from light.
Add 50 µL PBS, centrifuge, and discard the supernatant.
Perform a final wash with 250 µL of PBS, centrifuge, and discard the supernatant.
Fc receptor blocking
Resuspend cell pellets in 100 µL of PBS/ Azide solution.
Add 0.5 µg (1 µL) of TruStain FcX‱ PLUS (anti-mouse CD16/32) antibody per well following manufacturer instructions.
Incubate for 10 minutes at 4°C protected from light.
Wash the cells by adding 150 µL of FACS buffer, centrifuge, and discard the supernatant.
Repeat the wash with 250 µL of FACS buffer, centrifuge, and discard the supernatant.
Proceed to stain the thymocytes and the thymic stromal cell populations separately.
Thymocytes staining
After the Fc receptor blocking step, add 50 µL of FACS buffer containing appropriately diluted (based on previous titrations) primary, fluorescent-conjugated antibodies to each well for surface staining: PE-TxRed/ CD45, Brilliant Violet 785/ CD3, PCY7/ CD62L, eFluor450/ PD-1, PE/ CD25, Qdot605/ CD4, BV650/ CD8, AF700/CD127. Incubate mixes for 15- 30 min at room temperature protected from light.
Add 250 µL of PBS to each well, transfer the contents to 5 mL flow cytometry tubes, add an additional 750 µL of PBS to each tube (to complete 1 mL final volume) following instructions from the PerFix-nc kit (Beckman Coulter), centrifuge at 300 x g for 5 minutes, and discard the supernatant.
Resuspend cell pellets in 100 µL of PBS.
Add 10 µL of the Fixative Reagent (Reagent 1 of the PerFix-nc kit).
Vortex and incubate for 15 minutes at room temperature (18 – 25 °C).
Vortex and add 300 µL of the Permeabilizing Reagent (Reagent 2 of the PerFix-nc kit) to each tube and
vortex.
Add fluorophore-conjugated antibodies for intracellular staining: eF660/ Tox1 and AF488/ FoxP3 at adequate concentration each (based on previous titration). Vortex and incubate for 15 – 30 min at room temperature.
Add 3 ml of Final 1X Reagent (prepared by diluting the 10X concentrated Final Solution, Reagent 3 of the PerFix-nc kit, with type II water), vortex.
Centrifuge at 300 x g for 5 minutes and resuspend cell pellets in 250 uL of Optilyse C.
Filter all samples through 70 μm cell strainers immediately before acquisition on the Dx Flex Flow cytometer.
Thymic Stromal Cells staining
After the Fc receptor blocking step, add 50 µL of FACS buffer containing appropriately diluted (based on previous titrations) primary, fluorescent-conjugated antibodies to each well for surface staining: PE-Texas Red/ CD45, PE/ CD205, eFluor‱ 450/ CD326 (EpCAM) and APC/ IA-IE (MHCII). Incubate mixtures for 15- 30 min at room temperature protected from light.
Add 250 µL of PBS to each well, transfer the contents to 5 mL flow cytometry tubes, add an additional 750 µL of PBS to each tube (to complete 1 mL final volume) following instructions from the PerFix-nc kit (Beckman Coulter), centrifuge at 300 x g for 5 minutes, and discard the supernatant. Resuspend cell pellets in 100 µL of PBS.
Add 10 µL of the Fixative Reagent (Reagent 1 of the PerFix-nc kit).
Vortex and incubate for 15 minutes at room temperature (18 – 25 °C).
Vortex and add 300 µL of the Permeabilizing Reagent (Reagent 2 of the PerFix-nc kit) to each tube and vortex.
Add the unconjugated FoxN1 antibody at an adequate concentration (based on a previous titration). Vortex and incubate for 15 – 30 min at room temperature.
Add the secondary antibody, anti-rabbit-AF488 conjugate following the manufacturer recommendations, and incubate 20 minutes at room temperature.
Add 3 ml of Final 1X Reagent (prepared by diluting the 10X concentrated Final Solution, Reagent 3 of the PerFix-nc kit, with type II water), vortex.
Centrifuge at 300 x g for 5 minutes and resuspend cell pellets in 250 uL of Optilyse C.
Filter all samples through 70 μm cell strainers immediately before acquisition on the Dx Flex Flow cytometer.
IV- Flow cytometry operation
The no stain sample is used to set laser voltages, the single-color staining samples are used to compensate fluorochrome emission overlap, and the FMO samples are used to provide empirical evidence that the instrument was well compensated. Instrument compensation is essential for proper interpretation of the results.
Run and record all samples, including all controls, after compensation adjustment.
V- Data Analysis
Thymocytes gating strategy (Cossarizza et al., 2021) (Figure 1)
Thymocytes were first gated based on forward scatter (FSC-A) and side scatter (SSC-A) properties (Figure 1 Panel A).
Doublets were excluded by FSC-A versus FSC-H gating and SSC-A versus SSC-H (Figure 1 Panels B and C, respectively).
Viable cells were identified using the viability dye (Live Aqua, negative gate) (Figure 1 Panel D).
After singlets and viable cells selection, two strategies of gating were performed. One strategy gated CD4 and CD8 expression on CD45+CD3+ population to distinguish more mature cells (Figure 1 Panel E, F and G) and the other strategy gated CD4 and CD8 expression on viable cells (Figure 1 Panel H). In both strategies, four types of cells were defined as: Double positive DP (CD4+CD8+), Double negative
DN (CD4-CD8-), Simple positive SP (CD4-CD8+ or CD4+CD8-).
Precursors and Thymic Tregs were distinguished on CD4+viable cells. In this case, precursors T regs were defined as FOXP3+ cells while Thymic T regs were stained as CD25+FOXP3+ T cells (Figure 1 Panel I and J, respectively).
The rest of the markers (TOX-1, CD62L, PD-1 and CD127) were analyzed inside DN, DP and SP populations. As examples expression of TOX-1 in DP population and PD-1 in CD3+CD8+ populations is shown (Figure 1 Panel K and L, respectively).
Thymic stromal cells gating strategy (Jain & Gray, 2014)(Gray et al., 2002)(Gray et al., 2008)(Rode et al., 2015)(Figure 2).
All thymus cells were first gated based on forward scatter (FSC-A) and side scatter (SSC-A) properties as previously described (Jain & Gray, 2014) (Gray et al., 2002) (Figure 2 Panel A).
Doublets were excluded by FSC-A versus FSC-H gating and SSC-A versus SSC-H (Figure 2 Panels B and C).
Viable cells were identified using the viability dye (Live Aqua, negative gate) (Figure 2 Panel D).
After singlets and viable cell selection, non-hematopoietic and hematopoietic cells were defined as CD45- and CD45+, respectively (Figure 2 Panel E).
Within the CD45+ subpopulation, hematopoietic APCs were identified as MHCII positive with the surface marker IA-IE (IA-IE+) (Figure 2 Panel F). Within the CD45- subpopulation, TECs were identified as CD326+ cells (Figure 2 Panel G).
MHCII expression into TECS was analyzed with the surface marker IA-IE, and cells were classified as MHCII(IA-IE)low or MHCII(IA-IE)high (Figure 2 Panel H).
FoxN1 expression into TECS was analyzed and cells were classified as FoxN1- or FoxN1+ (Figure 2 Panel I).
Division of TECS into subpopulations was assessed with the surface marker CD205 as CD326+CD205- (mTECs) and CD326+CD205+ (cTECs) (Figure 2 Panel J).
Protocol references
Alpdogan, O., Muriglan, S. J., Eng, J. M., Willis, L. M., Greenberg, A. S., Kappel, B. J., & van den Brink, M. R. (2003). IL-7 enhances peripheral T cell reconstitution after allogeneic hematopoietic stem cell transplantation. The Journal of Clinical Investigation, 112, 1095–1107. https://doi.org/10.1172/JCI200317865.
Aw, D., Silva, A. B., & Palmer, D. B. (2010). The Effect of Age on the Phenotype and Function of Developing Thymocytes. Journal of Comparative Pathology, 142, S45–S59. https://doi.org/10.1016/j.jcpa.2009.10.004.
Bohacova, P., Terekhova, M., Tsurinov, P., Mullins, R., Husarcikova, K., Shchukina, I., Antonova, A. U., Echalar, B., Kossl, J., Saidu, A., Francis, T., Mannie, C., Arthur, L., Harridge, S. D. R., Kreisel, D., Mudd, P. A., Taylor, A. M., McNamara, C. A., Cella, M., … Artyomov, M. N. (2024). Multidimensional profiling of human T cells reveals high CD38 expression, marking recent thymic emigrants and age-related naive T cell remodeling. Immunity, 57, 2362-2379.e10. https://doi.org/10.1016/j.immuni.2024.08.019.
Caruso, B., Weeder, B. R., Thompson, R. F., & Moran, A. E. (2024). PD-1 Limits IL-2 Production and Thymic Regulatory T Cell Development. ImmunoHorizons, 8, 281–294. https://doi.org/10.4049/immunohorizons.2300079.
Cepeda, S., & Griffith, A. V. (2018). Thymic stromal cells: Roles in atrophy and age-associated dysfunction of the thymus. Experimental Gerontology, 105, 113–117. https://doi.org/10.1016/j.exger.2017.12.022.
Chiu, B. C., Stolberg, V. R., Zhang, H., & Chensue, S. W. (2007). Increased Foxp3+ Treg cell activity reduces dendritic cell co-stimulatory molecule expression in aged mice. Mechanisms of Ageing and Development, 128, 618–627. https://doi.org/10.1016/j.mad.2007.09.002.
Cossarizza, A., Chang, H. D., Radbruch, A., Abrignani, S., Addo, R., Akdis, M., Andrä, I., Andreata, F., Annunziato, F., Arranz, E., Bacher, P., Bari, S., Barnaba, V., Barros-Martins, J., Baumjohann, D., Beccaria, C. G., Bernardo, D., Boardman, D. A., Borger, J., … Yang, J. (2021). Guidelines for the use of flow cytometry and cell sorting in immunological studies (third edition). European Journal of Immunology, 51, 2708–3145. https://doi.org/10.1002/eji.202170126.
Gray, D. H. D., Chidgey, A. P., & Boyd, R. L. (2002). Analysis of thymic stromal cell populations using flow cytometry. Journal of Immunological Methods, 260, 15–28. https://doi.org/10.1016/s0022-1759(01)00493-8.
Gray, D. H. D., Fletcher, A. L., Hammett, M., Seach, N., Ueno, T., Young, L. F., Barbuto, J., Boyd, R. L., & Chidgey, A. P. (2008). Unbiased analysis, enrichment and purification of thymic stromal cells. Journal of Immunological Methods, 329, 56–66. https://doi.org/10.1016/j.jim.2007.09.010.
Gray, D. H. D., Seach, N., Ueno, T., Milton, M. K., Liston, A., Lew, A. M., Goodnow, C. C., & Boyd, R. L. (2006). Developmental kinetics, turnover, and stimulatory capacity of thymic epithelial cells. Blood, 108, 3777–3785. https://doi.org/10.1182/blood-2006-02.
Han, J., & Zúñiga-Pflücker, J. C. (2021). A 2020 View of Thymus Stromal Cells in T Cell Development. The Journal of Immunology, 206(2), 249–256. https://doi.org/10.4049/jimmunol.2000889.
Jain, R., & Gray, D. H. D. (2014). Isolation of thymic epithelial cells and analysis by flow cytometry. Current Protocols in Immunology, 107, 3.26.1-3.26.15. https://doi.org/10.1002/0471142735.im0326s107.
Ki, S., Park, D., Selden, H. J., Seita, J., Chung, H., Kim, J., Iyer, V. R., & Ehrlich, L. I. R. (2014). Global transcriptional profiling reveals distinct functions of thymic stromal subsets and age-related changes during thymic involution. Cell Reports, 9, 402–415. https://doi.org/10.1016/j.celrep.2014.08.070.
Leary, S., Anthony, R., Cartner, S., Grandin, T., Greenacre, C., Gwaltney-Brant, S., McCrackin, M. A., Meyer, R., Miller, D., Turner, T., & Yanong, R. (2020). American Veterinary Medical Association (AVMA) Guidelines for the Euthanasia of Animals (2020 Edition). https://icwdm.org/wp-content/uploads/2021/03/2020avma-euthanasia-guidelines.pdf
Liang, Z., Dong, X., Zhang, Z., Zhang, Q., & Zhao, Y. (2022). Age-related thymic involution: Mechanisms and functional impact. Aging Cell, 21, e13671. https://doi.org/10.1111/acel.13671.
Lynch, H. E., Goldberg, G. L., Chidgey, A., Van den Brink, M. R. M., Boyd, R., & Sempowski, G. D. (2009). Thymic involution and immune reconstitution. Trends in Immunology, 30, 366–373. https://doi.org/10.1016/j.it.2009.04.003
Min, H., Montecino-Rodriguez, E., & Dorshkind, K. (2005). Effects of aging on early B-and T-cell development. Immunological Reviews, 205, 7–17. https://doi.org/10.1111/j.0105-2896.2005.00263.x.
Nishimura, H., Agata, Y., Kawasaki, A., Sato, M., Imamura, S., Minato, N., Yagita, H., Nakano, T., & Honjo, T. (1996). Developmentally regulated expression of the PD-1 protein on the surface of double-negative (CD4-CD8-) thymocytes. International Immunology, 8, 773–780. https://doi.org/10.1093/intimm/8.5.773.
Nitta, T., & Suzuki, H. (2016). Thymic stromal cell subsets for T cell development. Cellular and Molecular Life Sciences, 73, 1021–1037. https://doi.org/10.1007/s00018-015-2107-8.
Ortman, C. L., Dittmar, K. A., Witte, P. L., & Le, P. T. (2002). Molecular characterization of the mouse involuted thymus: aberrations in expression of transcription regulators in thymocyte and epithelial compartments. International Immunology, 14, 813–822. https://doi.org/10.1093/intimm/dxf042.
Peschon, J. J., Morrissey, P. J., Grabstein, K. H., Ramsde11, F. J., Maraskovsky, E., Gliniak, B. C., Park, L. S., Ziegler, S. F., Williams, D. E., Ware, C. B., Meyer, J. D., & Davison, B. L. (1994). Early Lymphocyte Expansion Is Severely Impaired in Interleukin 7 Receptor-deficient Mice. Journal of Experimental Medicine, 180, 1955–1960. https://doi.org/10.1084/jem.180.5.1955.
Rode, I., Martins, V. C., Küblbeck, G., Maltry, N., Tessmer, C., & Rodewald, H.-R. (2015). Foxn1 Protein Expression in the Developing, Aging, and Regenerating Thymus. The Journal of Immunology, 195, 5678–5687.
https://doi.org/10.4049/jimmunol.1502010.
Scott, A. C., Dündar, F., Zumbo, P., Chandran, S. S., Klebanoff, C. A., Shakiba, M., Trivedi, P., Menocal, L., Appleby, H., Camara, S., Zamarin, D., Walther, T., Snyder, A., Femia, M. R., Comen, E. A., Wen, H. Y., Hellmann, M. D., Anandasabapathy, N., Liu, Y., … Schietinger, A. (2019). TOX is a critical regulator of tumour-specific T cell differentiation. Nature, 571, 270–274. https://doi.org/10.1038/s41586-019-1324-y.
Sekine, T., Perez-Potti, A., Nguyen, S., Gorin, J.-B., Wu, V. H., Gostick, E., Llewellyn-Lacey, S., Hammer, Q., Falck-Jones, S., Vangeti, S., Yu, M., Smed-Sörensen, A., Gaballa, A., Uhlin, M., Sandberg, J. K., Brander, C., Nowak, P., Goepfert, P. A., Price, D. A., … Buggert, M. (2020). TOX is expressed by exhausted and polyfunctional human effector memory CD8 + T cells. Science Immunology, 5, eaba7918. https://doi.org/10.1126/sciimmunol.aba7918.
Van Dommelen, S. L., Rizzitelli, A., Chidgey, A., Boyd, R., Shortman, K., & Wu, L. (2010). Regeneration of dendritic cells in aged mice. Cellular and Molecular Immunology, 7, 108–115. https://doi.org/10.1038/cmi.2009.114.
Wedemeyer, S. A., & Griffith, A. V. (2025). Thymic B cells in aging and autoimmune disease. Frontiers in Immunology, 16, 1595805. https://doi.org/10.3389/fimmu.2025.1595805.
Weinreich, M. A., & Hogquist, K. A. (2008). Thymic emigration: when and how T cells leave home 1. Journal of Immunology, 181, 2265–2270. https://doi.org/10.4049/jimmunol.181.4.2265.
Wilkinson, B., Chen, J. Y. F., Han, P., Rufner, K. M., Goularte, O. D., & Kaye, J. (2002). TOX: An HMG box protein implicated in the regulation of thymocyte selection. Nature Immunology, 3, 272–280. https://doi.org/10.1038/ni767
Xing, Y., & Hogquist, K. A. (2014). Isolation, identification, and purification of murine thymic epithelial cells. Journal of Visualized Experiments, 90, e51780. https://doi.org/10.3791/51780.
Yao, C., Sun, H. W., Lacey, N. E., Ji, Y., Moseman, E. A., Shih, H. Y., Heuston, E. F., Kirby, M., Anderson, S., Cheng, J., Khan, O., Handon, R., Reilley, J., Fioravanti, J., Hu, J., Gossa, S., Wherry, E. J., Gattinoni, L., McGavern, D. B., … Wu, T. (2019). Single-cell RNA-seq reveals TOX as a key regulator of CD8+ T cell persistence in chronic infection. Nature Immunology, 20, 890–901. https://doi.org/10.1038/s41590-019-0403-4.
Zook, E. C., Krishack, P. A., Zhang, S., Zeleznik-Le, N. J., Firulli, A. B., Witte, P. L., & Le, P. T. (2011). Overexpression of Foxn1 attenuates age-associated thymic involution and prevents the expansion of peripheral CD4 memory T cells. Blood, 118, 5723–5731. https://doi.org/10.1182/blood-2011-03-342097.
Acknowledgements
We thank the staff of the animal facilities at UAEU for their support and assistance in animal handling. We also acknowledge ADSCC for technical support and resources contributing to this work.

