Sep 10, 2024
Multicellular Circulating Co-Culture
- Bianca Cruz Pachane1,
- Pedro Henrique Teixeira Bottaro1,
- Wanessa Fernanda Altei2,
- Heloisa Sobreiro Selistre de Araujo1
- 1Universidade Federal de São Carlos - UFSCar, São Carlos, SP, Brazil;
- 2Barretos Cancer Hospital, Barretos, SP, Brazil
- Bianca Cruz Pachane: Full optimization and experimentation;
- Pedro Henrique Teixeira Bottaro: Optimization of THP-1 culture
- Wanessa Fernanda Altei: Initial development of method
- Heloisa Sobreiro Selistre de Araujo: Financial support and System acquisition

External link: https://doi.org/10.1002/jex2.70075
Protocol Citation: Bianca Cruz Pachane, Pedro Henrique Teixeira Bottaro, Wanessa Fernanda Altei, Heloisa Sobreiro Selistre de Araujo 2024. Multicellular Circulating Co-Culture. protocols.io https://dx.doi.org/10.17504/protocols.io.ewov19b47lr2/v1
Manuscript citation:
Pachane BC, Bottaro PHT, Machado AM, Castro CAd, Guerra G, Gozzer LT, Grigoli MM, Zutião AD, Fuzer AM, Cominetti MR, Altei WF, Selistre‐de‐Araujo HS (2025) Tumoural Hypoxic Extracellular Vesicles Foster a Protective Microenvironment in Triple‐Negative Breast Cancer. Journal of Extracellular Biology 4(9). doi: 10.1002/jex2.70075
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: September 10, 2024
Last Modified: September 10, 2024
Protocol Integer ID: 107221
Keywords: tumor microenvironment, role of hypoxic tumoral extracellular vesicle, hypoxic tumoral extracellular vesicle, tumoral cell, endothelial cell, suspension monocyte, cell, cell type, fibronectin, gelatin coating
Funders Acknowledgements:
São Paulo Research Foundation
Grant ID: 2021/01983-4
São Paulo Research Foundation
Grant ID: 2019/11437-7
São Paulo Research Foundation
Grant ID: 2022/12307-2
Disclaimer
DISCLAIMER – FOR INFORMATIONAL PURPOSES ONLY; USE AT YOUR OWN RISK
The protocol content here is for informational purposes only and does not constitute legal, medical, clinical, or safety advice, or otherwise; content added to protocols.io is not peer reviewed and may not have undergone a formal approval of any kind. Information presented in this protocol should not substitute for independent professional judgment, advice, diagnosis, or treatment. Any action you take or refrain from taking using or relying upon the information presented here is strictly at your own risk. You agree that neither the Company nor any of the authors, contributors, administrators, or anyone else associated with protocols.io, can be held responsible for your use of the information contained in or linked to this protocol or any of our Sites/Apps and Services.
Abstract
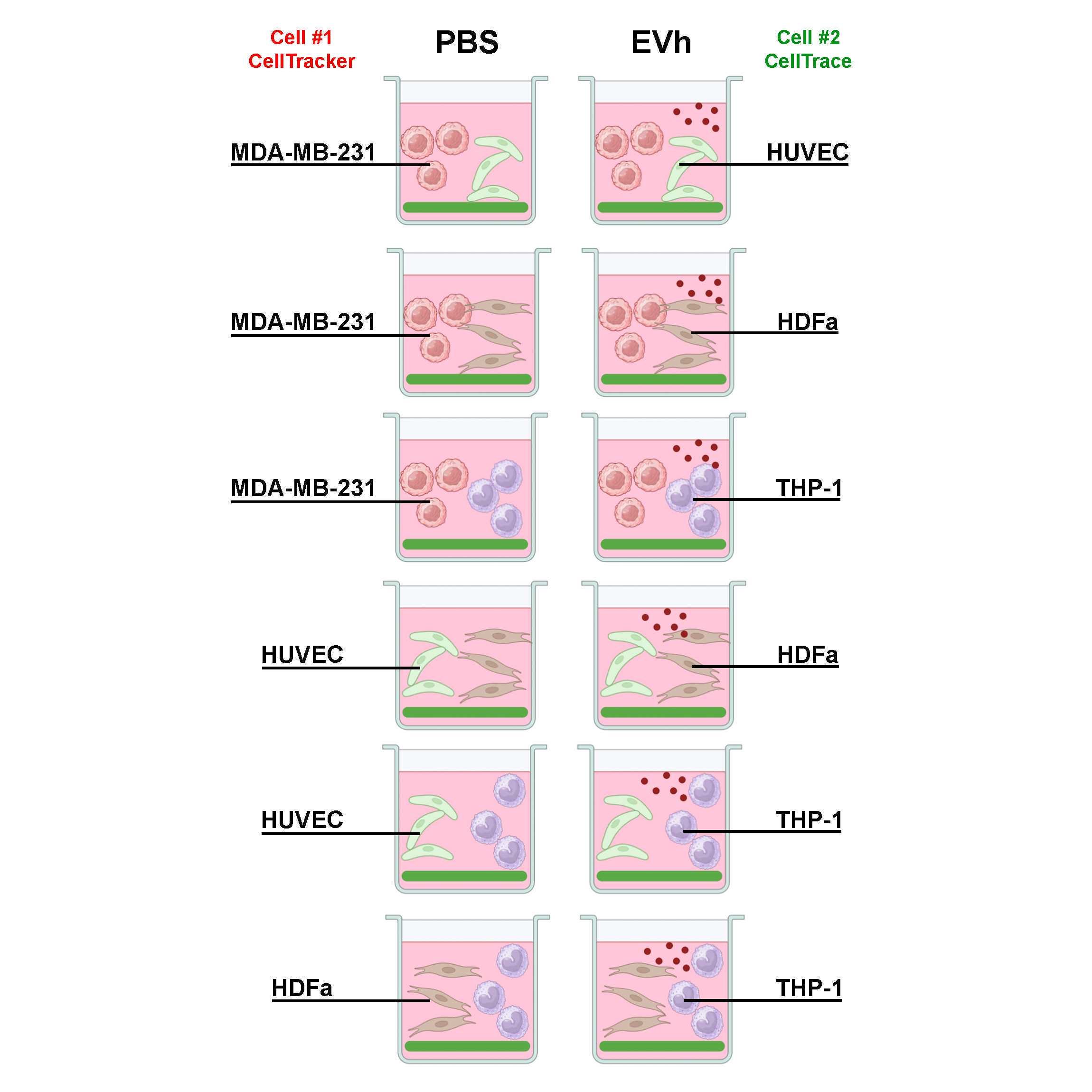
A novel method to study the tumor microenvironment (TME) in vitro, using the quasi-vivo technology from Kirstall to survey the individual responses in cell types common to the TME. We have developed a strategy that allows a tumoral cell (MDA-MB-231) seeded in gelatin coating, an endothelial cell (HUVEC) seeded in Matrigel coating, and a dermal fibroblast (HDFa) seeded in fibronectin to be cultured in tandem, alongside suspension monocytes (THP-1). The goal was to investigate how cells would behave in this setting and evaluate the role of hypoxic tumoral extracellular vesicles in the development of the TME.
Image Attribution
The diagram was created using Adobe Photoshop. Original photograph by Bianca Pachane.
Materials
Materials:
- Round glass coverslips 13mm ø
- 24-well clear plates with flat bottom
- Histological slides, Exacta.
- Sterile forceps
- Petri dishes
- 0.22 pore syringe filters
Reagents and Solutions:
- Parafilm™ M Laboratory Wrapping Film, 4 in. W x 125 ft. L; (10cm x 38m)Thermo FisherCatalog #1337410
- Poly-l-lysine, 0.1% (wt/vol)Merck MilliporeSigma (Sigma-Aldrich)Catalog #P8920
- Glutaraldehyde solution (50% in solution)Merck MilliporeSigma (Sigma-Aldrich)Catalog #G6403
- Gelatin From Pig Skin, Fluorescein ConjugateInvitrogen - Thermo FisherCatalog #G13187
- Corning® Matrigel®CorningCatalog #354277
- FibronectinGibco - Thermo Fisher ScientificCatalog #33016-015
- 1X PBS (Phosphate-buffered saline )
- OptiMEM™ I Reduced Serum MediaGibco - Thermo Fisher ScientificCatalog #31985070
- Trypan Blue Solution 0.4% Sterile-filtered Merck MilliporeSigma (Sigma-Aldrich)Catalog #T8154
- CellTracker™ Red CMTPX DyeThermo FisherCatalog #C34552 - reconstituted in DMSO
- ParaformaldehydeMerck MilliporeSigma (Sigma-Aldrich)Catalog #P6148 - PFA 4% in deionized water, pH 7.6, sterile
- Triton X-100Merck MilliporeSigma (Sigma-Aldrich)Catalog #T8787-50ML - Triton X-100 0.1% (v/v) in deionized water
- Bovine Serum AlbuminMerck MilliporeSigma (Sigma-Aldrich)Catalog #A7030 - 1% BSA-PBS
- Anti-VEGF Receptor 2 antibodyAbcamCatalog #ab39256 - 1:50 dilution in 1% BSA-PBS
- BD Transduction Laboratories™ Purified Mouse Anti-β-CateninBD BiosciencesCatalog #610154 - 1:50 dilution in 1% BSA-PBS
- Anti-Collagen II antibodyAbcamCatalog #ab85266 - 1:50 dilution in 1% BSA-PBS
- Anti-Collagen III antibody [1E7-D7/Col3]AbcamCatalog #ab23445 - 1:50 dilution in 1% BSA-PBS
- Goat Anti-Mouse IgG H&L (FITC)AbcamCatalog #ab6785 - 1:1,000 dilution in 1% BSA-PBS
- Goat Anti-Rabbit IgG H&L (Alexa Fluor® 568)AbcamCatalog #ab175696 - 1:1,000 dilution in 1% BSA-PBS
- Phalloidin + DAPI (1 µl Phalloidin-iFluor 647 ReagentAbcamCatalog #ab176759 + 0.76 µl 4,6-Diamidino-2-Phenylindole, Dihydrochloride (DAPI)Thermo Fisher ScientificCatalog #D1306 in 5 ml PBS)
- FluoromountMerck MilliporeSigma (Sigma-Aldrich)Catalog #F4680
Cell lines and growth media:
- MDA-MB-231 (ATCC™ CRM-HTB-26®) - Leibovitz L-15 10% FBS
- HDFa (ATCC™ PCS-201-012®) - DMEM 10% FBS 1% pen/strep
- HUVEC (ATCC™ CRL-1730®) - DMEM 10% FBS 1% pen/strep
- THP-1 (ATCC™ TIB-202®) - RPMI 1640 10% FBS 1% pen/strep
Equipments:
- Biological cabinet
- Cell incubator (37 ºC, 5% CO2)
- QV500 System, Kirstall
- Cell counter - TC20 Cell Counter, Bio-Rad - Catalog #1450011
- Epifluorescence microscope - ImageXpress Micro XLS, Molecular Devices - Catalog #500496
Protocol materials
1X PBS (Phosphate-buffered saline )
Parafilm™ M Laboratory Wrapping Film, 4 in. W x 125 ft. L; (10cm x 38m)Thermo FisherCatalog #1337410
CellTracker™ Red CMTPX DyeThermo FisherCatalog #C34552
OptiMEM™ I Reduced Serum MediaGibco - Thermo Fisher ScientificCatalog #31985070
Trypan Blue Solution 0.4% Sterile-filtered Merck MilliporeSigma (Sigma-Aldrich)Catalog #T8154
Poly-l-lysine, 0.1% (wt/vol)Merck MilliporeSigma (Sigma-Aldrich)Catalog #P8920
Glutaraldehyde solution (50% in solution)Merck MilliporeSigma (Sigma-Aldrich)Catalog #G6403
Anti-Collagen III antibody [1E7-D7/Col3]AbcamCatalog #ab23445
ParaformaldehydeMerck MilliporeSigma (Sigma-Aldrich)Catalog #P6148
BD Transduction Laboratories™ Purified Mouse Anti-β-CateninBD BiosciencesCatalog #610154
Goat Anti-Mouse IgG H&L (FITC)AbcamCatalog #ab6785
FluoromountMerck MilliporeSigma (Sigma-Aldrich)Catalog #F4680
Gelatin From Pig Skin, Fluorescein ConjugateInvitrogen - Thermo FisherCatalog #G13187
Corning® Matrigel®CorningCatalog #354277
Bovine Serum AlbuminMerck MilliporeSigma (Sigma-Aldrich)Catalog #A7030
Anti-VEGF Receptor 2 antibodyAbcamCatalog #ab39256
Anti-Collagen II antibodyAbcamCatalog #ab85266
Goat Anti-Rabbit IgG H&L (Alexa Fluor® 568)AbcamCatalog #ab175696
4,6-Diamidino-2-Phenylindole, Dihydrochloride (DAPI)Thermo Fisher ScientificCatalog #D1306
FibronectinGibco - Thermo Fisher ScientificCatalog #33016-015
Triton X-100Merck MilliporeSigma (Sigma-Aldrich)Catalog #T8787-50ML
Phalloidin-iFluor 647 ReagentAbcamCatalog #ab176759
Troubleshooting
Safety warnings
Light-sensitive assay. Work under sterile conditions.
Before start
Fluorescent gelatin preparation: Under sterile conditions, solubilize the fluorescent gelatin stock at 37 °C with warmed PBS following the manufacturer's instructions for a concentration of 5 mg/mL . Aliquot in microtubes and maintain at -20 °C until time of use.
Before use, thaw gelatin at 37 °C for 00:30:00 . Dilute stock to a 0.2 mg/mL working solution with warmed PBS and maintain at 37 °C until use.
Matrigel preparation: Thaw Matrigel vial at 4 °C and maintain On ice until use. Dilute a 1:1, v/v aliquot of stock Matrigel in cold sterile PBS for use.
Cell culture: Maintain cells in culture during at least two passages after thawing.
QV500: Autoclave all parts before use.
Preparation of matrix-coated coverslips
20m
Clean round glass coverslips (13 mm ø) with 70% ethanol wipes before use. Maintain slips in a clean container.
Prepare a 0.5% solution of glutaraldehyde in H2O and maintain it at 4 °C protected from light.
Glutaraldehyde solution (50% in solution)Merck MilliporeSigma (Sigma-Aldrich)Catalog #G6403
Under sterile conditions, prepare a surface with a piece of Parafilm fixed in place. Add 20 µL droplets of poly-L-lysine interspaced in the Parafilm.
Poly-l-lysine, 0.1% (wt/vol)Merck MilliporeSigma (Sigma-Aldrich)Catalog #P8920
Parafilm™ M Laboratory Wrapping Film, 4 in. W x 125 ft. L; (10cm x 38m)Thermo FisherCatalog #1337410
Drop coverslips atop the droplets and incubate at Room temperature for 00:20:00 minimum.
20m
Using forceps, transfer the coverslips to a 24-well plate with the coating facing upwards.
Wash coverslips twice with500 µL PBS.
1X PBS (Phosphate-buffered saline )
Cross-link coating with 500 µL of cold 0.5 % (v/v) glutaraldehyde for 00:15:00 at Room temperature
15m
Prepare a Petri dish with the bottom covered in Parafilm.
Parafilm™ M Laboratory Wrapping Film, 4 in. W x 125 ft. L; (10cm x 38m)Thermo FisherCatalog #1337410
Apply spaced 20 µL droplets of the matrices to the Parafilm-covered surface:
- Fluorescent gelatin: 0.2 mg/mL in PBS, kept at 37 °C until time of use;
- Matrigel solution: 50 % (v/v) in PBS, kept On ice until time of use;
- Fibronectin solution: 1 mg/mL in PBS, kept On ice until time of use;
Remove the coverslips from the 24-well plate and drop them atop the droplets, with the coating facing down. Incubate at 4 °C Overnight , protected from light.
The next day, remove the slips from the Petri dish using a forceps and transfer them, with the coating facing up, to a fresh 24-well plate.
Wash coverslips twice with500 µL PBS.
1X PBS (Phosphate-buffered saline )
Slips can be stored at 4 ºC for up to a week, wrapped in aluminium foil.
Pre-condition matrix coating with 500 µL of culture media for 00:30:00 at 37 °C :
- Fluorescent gelatin: Leibovitz L-15 10% FBS
- Matrigel solution: DMEM 10% FBS 1% pen/strep
- Fibronectin solution: DMEM 10% FBS 1% pen/strep
30m
Cell seeding
Subculture cells as usual. Resuspend cell pellets in growth media and count cells using the trypan blue exclusion method.
- MDA-MB-231 - Leibovitz L-15 10% FBS
- HUVEC - DMEM 10% FBS 1% pen/strep
- HDFa - DMEM 10% FBS 1% pen/strep
Trypan Blue Solution 0.4% Sterile-filtered Merck MilliporeSigma (Sigma-Aldrich)Catalog #T8154
Remove the pre-conditioning medium from the 24-well plate.
Seed cells in the 24-wells following the table below:
| A | B | C | D | |
| Coating: | Cell Type: | Density: | Culture Media: | |
| Fluorescent gelatin | MDA-MB-231 | 50,000 cells/ml | 1 ml Leibovitz L-15 10% FBS | |
| Matrigel | HUVEC | 50,000 cells/ml | 1 ml DMEM 10% FBS 1% pen/strep | |
| Fibronectin | HDFa | 20,000 cells/ml | 1 ml DMEM 10% FBS 1% pen/strep |
Incubate cells at 37 °C 5% CO2 Overnight for adhesion.
ps: seal the MDA-MB-231 plate with parafilm to protect from CO2 exposure.
THP-1 staining and seeding
5m
The day after cell seeding:
Transfer THP-1 suspension to a conical tube and centrifuge at 1200 rpm, 00:05:00 . Discard supernatant.
5m
Resuspend cell pellet in OptiMEM 1% pen/strep and count cells using the trypan blue exclusion method.
OptiMEM™ I Reduced Serum MediaGibco - Thermo Fisher ScientificCatalog #31985070
Trypan Blue Solution 0.4% Sterile-filtered Merck MilliporeSigma (Sigma-Aldrich)Catalog #T8154
Dilute cell suspension to a 1x106 cells/ml density in OptiMEM 1% pen/strep.
OptiMEM™ I Reduced Serum MediaGibco - Thermo Fisher ScientificCatalog #31985070
Add 1 µL of CellTracker™ CMPTX per ml of cell suspension for a 5 micromolar (µM) final concentration. Pipette well to mix.
CellTracker™ Red CMTPX DyeThermo FisherCatalog #C34552
Incubate the cell suspension at37 °C % CO2 for 00:30:00 , protected from light.
30m
Dilute cell suspensions with 4 mL OptiMEM and centrifuge at 1200 rpm, 00:05:00
OptiMEM™ I Reduced Serum MediaGibco - Thermo Fisher ScientificCatalog #31985070
5m
Resuspend cell pellets in 1 mL OptiMEM and recount cells using the trypan blue exclusion method.
OptiMEM™ I Reduced Serum MediaGibco - Thermo Fisher ScientificCatalog #31985070 Trypan Blue Solution 0.4% Sterile-filtered Merck MilliporeSigma (Sigma-Aldrich)Catalog #T8154
In a 15-ml conical tube, prepare a 1x105 cell/ml THP-1 suspension in 15 mL OptiMEM 1% pen/strep . Maintain protected from light.
OptiMEM™ I Reduced Serum MediaGibco - Thermo Fisher ScientificCatalog #31985070
QV500 assembly and preparation
10m
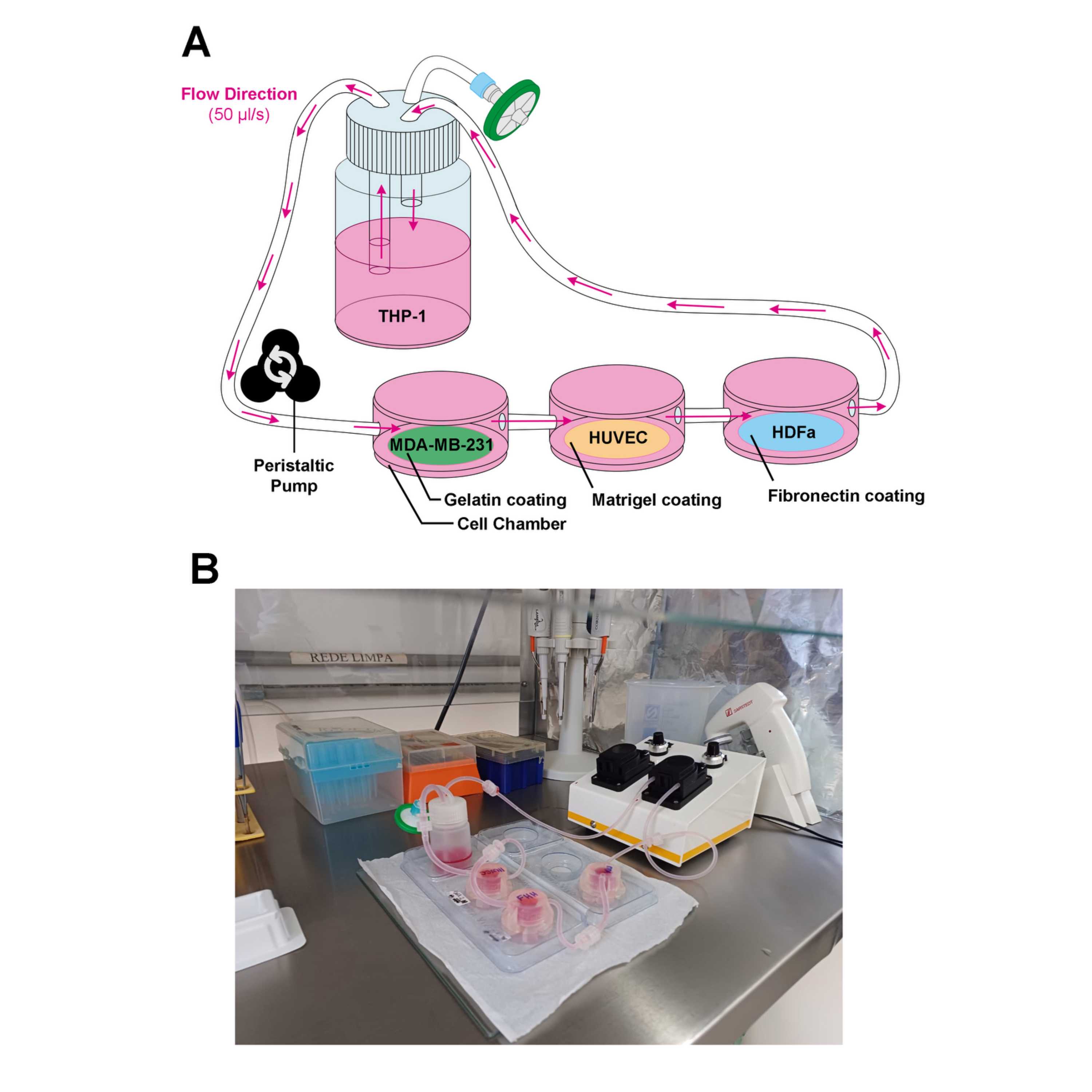
QV500 consists of:
- (1) Peristaltic pump
- (3) Culture chambers with connecting tubings
- (1) 15-ml reservoir with 1 inlet, 1 outlet and 1 air filter piece.
- (1) 1.6 mm pump tubing
- (2) plastic chamber supports
Check the experimental diagram for better understanding.
Assemble the system under the biological hood with autoclaved parts:
Add 15 mL sterile PBS to the reservoir and close the lid.
1X PBS (Phosphate-buffered saline )
Attach the 0.22 µm pore syringe filter to the air outlet (blue piece).
Add 1 mL sterile PBS to each chamber and close the lids.
1X PBS (Phosphate-buffered saline )
Attach the 1.6 mm pump tubing to the peristaltic pump.
Connect the inlet and outlet tubings with the chambers to form a close circuit.
Circulate the PBS for 00:10:00 at max flow rate.
10m
Drain the system and disassemble the parts.
Repeat steps until step #29 with 15 ml OptiMEM to condition the system.
OptiMEM™ I Reduced Serum MediaGibco - Thermo Fisher ScientificCatalog #31985070
Multicellular Circulating Co-Culture
1d
Remove the growth media from cells seeded in coverslips ( ).
Assemble the MDA-gelatin coverslip in one QV500 culture chamber; the HUVEC-Matrigel coverslip in the second culture chamber; and the HDFa-FN coverslip in the last culture chamber.
Add 1 mL 1 ml OptiMEM 1% pen/strep to each chamber and close the lids, connecting the tubings following the order described above.
OptiMEM™ I Reduced Serum MediaGibco - Thermo Fisher ScientificCatalog #31985070
Add the 15 mL THP-1 cell suspension in OptiMEM 1% pen/strep to the reservoir.
Add EVh (109 particles/ml) or the equivalent treatment volume in PBS to the reservoir and close the lid.
Connect the tubings to close the system. Circulate the media at a 50 µl/s flow rate and incubate the whole system for 24:00:00 at 37 °C 5% CO2.
1d
The next day, disconnect the system and carefully drain the compartments. Collect the conditioned media in a conical tube and spin at 1200 rpm, 4°C, 00:10:00 . Transfer the supernatant to fresh tubes and freeze at -80 °C for further testing.
10m
Transfer the coverslips to a fresh 24-well plate for fixing and staining ( )
Wash and decontaminate the QV500 parts for further use.
Cell Fixing
Fix cells in coverslips with 500 µL warmed 4% PFA atRoom temperature for 00:10:00
10m
Wash coverslips twice with500 µL PBS.
1X PBS (Phosphate-buffered saline )
Permeabilize cells with 500 µL 0.1% Triton X-100 at Room temperature for 00:05:00
5m
Wash coverslips twice with500 µL PBS.
1X PBS (Phosphate-buffered saline )
For MDA-MB-231 coverslips only: skip the immunofluorescence protocol and
Immunofluorescence of HUVEC and HDFa Coverslips
Block non-specific bindings with 500 µL 1% BSA-PBS for 01:00:00 at 4 °C
1h
Prepare the primary antibody dilutions:
For HUVECs:
- Anti-VEGF Receptor 2 antibodyAbcamCatalog #ab39256 - 1:50 dilution in 1% BSA-PBS
- BD Transduction Laboratories™ Purified Mouse Anti-β-CateninBD BiosciencesCatalog #610154 - 1:50 dilution in 1% BSA-PBS
For HDFa:
- Anti-Collagen II antibodyAbcamCatalog #ab85266 - 1:50 dilution in 1% BSA-PBS
- Anti-Collagen III antibody [1E7-D7/Col3]AbcamCatalog #ab23445 - 1:50 dilution in 1% BSA-PBS
Place 20 µL of spaced droplets of the antibodies in a Petri dish covered with Parafilm (as seen on ).
Parafilm™ M Laboratory Wrapping Film, 4 in. W x 125 ft. L; (10cm x 38m)Thermo FisherCatalog #1337410
Remove the coverslips from the 24-well plate and drop them atop the droplets, with the cells facing down. Incubate at 4 °C for 18:00:00 , protected from light.
18h
The next day, remove the slips from the Petri dish using a forceps and transfer them, with the coating facing up, to a fresh 24-well plate.
Wash coverslips twice with500 µL PBS.
1X PBS (Phosphate-buffered saline )
Prepare the secondary antibody dilutions:
- Goat Anti-Mouse IgG H&L (FITC)AbcamCatalog #ab6785 - 1:1,000 dilution in 1% BSA-PBS
- Goat Anti-Rabbit IgG H&L (Alexa Fluor® 568)AbcamCatalog #ab175696 - 1:1,000 dilution in 1% BSA-PBS
Place 20 µL of spaced droplets of the antibodies in a Petri dish covered with Parafilm (as seen on ).
Parafilm™ M Laboratory Wrapping Film, 4 in. W x 125 ft. L; (10cm x 38m)Thermo FisherCatalog #1337410
Remove the coverslips from the 24-well plate and drop them atop the droplets, with the cells facing down. Incubate at Room temperature for 01:00:00 , protected from light.
1h
Remove the slips from the Petri dish using a forceps and transfer them, with the coating facing up, to a fresh 24-well plate.
Wash coverslips twice with500 µL PBS.
1X PBS (Phosphate-buffered saline )
Cell Counterstaining and Slide Assembly
20m
Add 500 µL of DAPI + Phalloidin-647 mixture to each well and incubate for 00:20:00 .
> 1 µl Phalloidin-iFluor 647 ReagentAbcamCatalog #ab176759 + 0.76 µl 4,6-Diamidino-2-Phenylindole, Dihydrochloride (DAPI)Thermo Fisher ScientificCatalog #D1306 in 5 ml PBS
20m
Wash coverslips twice with500 µL PBS.
1X PBS (Phosphate-buffered saline )
Remove the coverslips from the 24-well plate and assemble them in histological slides with mounting media. Allow the media to dry for at least 04:00:00 .
FluoromountMerck MilliporeSigma (Sigma-Aldrich)Catalog #F4680
4h
Once dry, seal coverslips using clear nail polish and store at4 °C until time of analysis.
Cell Imaging by Epifluorescence HTS
Using the microscope ImageXpress Micro XLS+ (Molecular Devices), check the template for the Corning 3603 plate and the filters for DAPI (nuclei), FITC (gelatin), TxRed (CMPTX) and Cy5 (phalloidin-647).
Set laser intensity to a minimum of 10 ms and increase gradatively if necessary.
Check the wells using the 4X objective.
Change into the 20x objective and adjust the laser focus. Select 9 sites per well minimally.
Acquire the plate. Export metadata for analysis.
Repeat and step #64 with the 40x objective.
For representative images, use the 40x objective and adjust the laser focus.
Select the sites of interest and acquire.
Export image channels and combinations.
Imaging analysis
Gelatin degradation quantification, cell morphology analysis, quantification of immunofluorescence probing, and assembly of representative images were performed using FIJI.
Software
ImageJ/Fiji
NAME
Windows 7
OS
National Institutes of Health
DEVELOPER
SOURCE LINK
Please refer to the following protocol for the complete pipeline of analysis.
Protocol references
PACHANE, Bianca Cruz et al. Small Extracellular Vesicles from Hypoxic Triple-Negative Breast Cancer Cells Induce Oxygen-Dependent Cell Invasion. International Journal of Molecular Sciences, [s. l.], v. 23, n. 20, p. 12646, 2022.
EVEN-RAM, Sharona; ARTYM, Vira. Extracellular Matrix Protocols: Second Edition. [S. l.]: Humana Press, 2009.