Jan 14, 2026
Migration of bacteria towards individual root exudate compounds
- Birgit Pruess1,
- Barney Geddes1,
- Shelley Horne1,
- Amanda Pease1,
- Fatema Akter Nisha1
- 1North Dakota State University

External link: https://doi.org/10.1139/cjm-2023-0217
Protocol Citation: Birgit Pruess, Barney Geddes, Shelley Horne, Amanda Pease, Fatema Akter Nisha 2026. Migration of bacteria towards individual root exudate compounds. protocols.io https://dx.doi.org/10.17504/protocols.io.261ge16edv47/v1
Manuscript citation:
Nisha, F.A.,J. Tagoe, A. Pease, S.M. Horne, A. Ugrinov, B. Geddes, and B.M. Prüß. 2024. Can. J. Microbiol. 70: 150-162. 10.1139/cjm-2023-0217
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working.
Created: December 24, 2025
Last Modified: January 14, 2026
Protocol Integer ID: 235806
Keywords: bacterial chemotaxis, semi-solid agar plates, Rhizobium leguminosarum, Azospirillum brasilense, seedling exudate compound, chemoattractants for rhizobium leguminosarum, migration of bacteria, rhizobium leguminosarum, individual root exudate compounds this protocol, bacteria, exudate compound, individual root exudate compound, azospirillum brasilense, tomato, own exudate, compound
Funders Acknowledgements:
Specialty Crop Blick Grant, USDA/NIFA
Grant ID: Grant 22-242
Abstract
This protocol was used to test the ability of seedling exudate compounds to serve as chemoattractants for Rhizobium leguminosarum bv. viciae and Azospirillum brasilense. Compounds were selected from the composition of our own exudates that were produced from peas, tomatoes, and cucumbers. The concept of the semi-solid swim plate was used, where bacteria migrate towards the attractant on 0.3% agar plates. The swim medium was modified from for R. leguminosarum and MMAB for A. brasilense.
Image Attribution
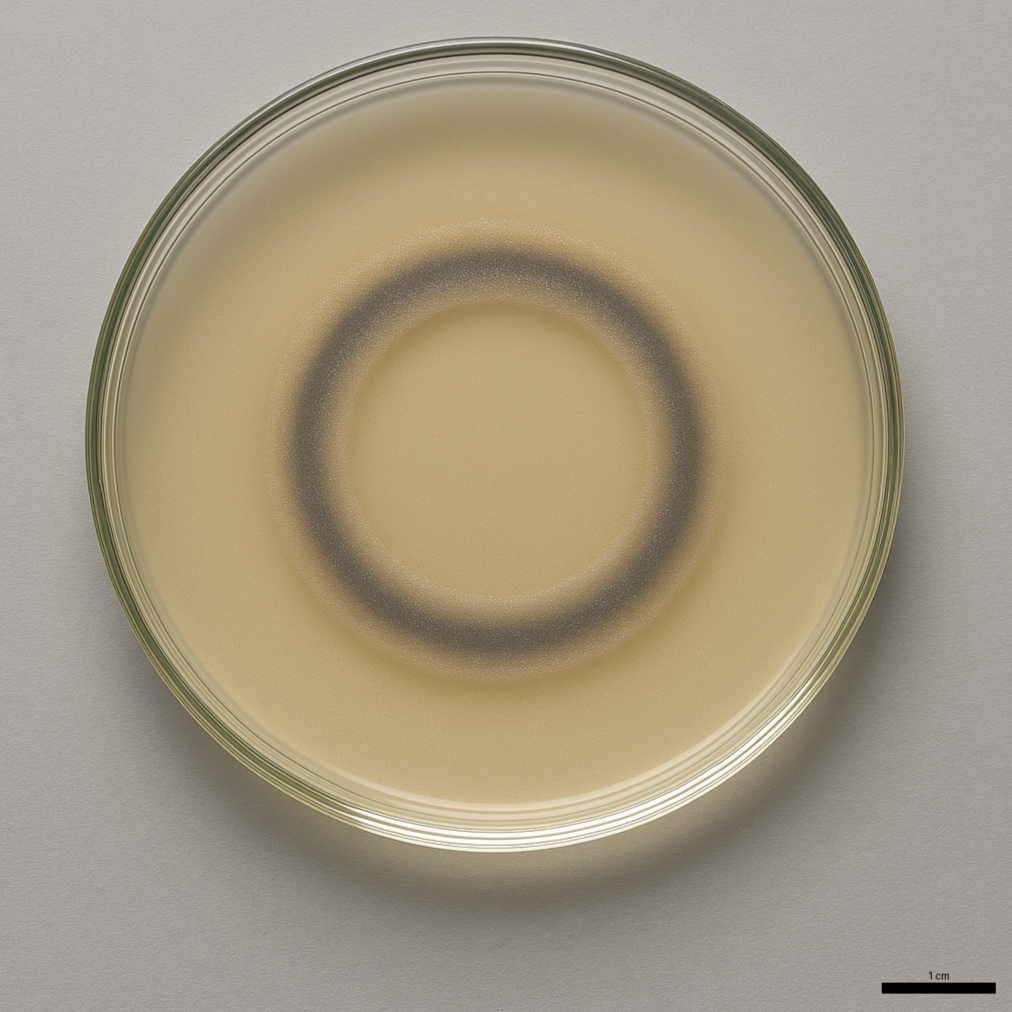
The image was generated by AI
Guidelines
You will measure the diameter of the swim ring. I use a ruler and measure the outermost edge. Some of the rings can be faint, that depends on how much the bacteria will be growing while swimming. If you let them swim long enough, you may see multiple rings. We usually measure until the rings are at lest 4 or 5 cm in diameter. If you plot the ring against time, you will see a bit of a lag, then a straight line.
Materials
- Revised RB minimal media (RB):**
- 7.3 mM K2HPO4, 3 mM KH2PO4, 1 mM MgSO4·7H2O, 1 mM (NH4)2SO4, 0.1 mM CaCl2·2H2O, 0.1 mM NaCl, 0.2% mannitol, supplemented with filter sterilized 0.01 mM Na2MoO4, 0.001 mM FeSO4, 0.2% carbon based putative chemoattractant, 1 μg/mL thiamine, 1 μg/mL biotin, 1 μg/mL pyridoxin HCl, and 10 μg/mL CoCl2 after autoclaving.
- Carbon based seedling exudate compounds (RB carbon):**
- 7.3 mM K2HPO4, 3 mM KH2PO4, 1 mM MgSO4·7H2O, 1 mM (NH4)2SO4, 0.1 mM CaCl2·2H2O, 0.1 mM NaCl, 1 mM carbon based putative chemoattractant, supplemented with filter sterilized 0.01 mM Na2MoO4, 0.001 mM FeSO4, 0.2% carbon based putative chemoattractant, 1 μg/mL thiamine, 1 μg/mL biotin, 1 μg/mL pyridoxin HCl, and 10 μg/mL CoCl2 after autoclaving.
- Amino acid seedling exudate compounds (RB amino acid):**
- 7.3 mM K2HPO4, 3 mM KH2PO4, 1 mM MgSO4·7H2O, 0.1 mM CaCl2·2H2O, 0.1 mM NaCl, 1 mM amino acid based putative chemoattractant, supplemented with filter sterilized 0.01 mM Na2MoO4, 0.001 mM FeSO4, 0.2% carbon based putative chemoattractant, 1 μg/mL thiamine, 1 μg/mL biotin, 1 μg/mL pyridoxin HCl, and 10 μg/mL CoCl2 after autoclaving.
Troubleshooting
Safety warnings
NA
Ethics statement
No humans or animals are involved in this protocol.
Before start
Be aware of what you want to do. You can use different bacteria, but will have to look up the minimal medium for them.
Migration of bacteria towards individual root exudate compounds
Test the ability of bacteria to migrate towards individual root exudate compounds. Select carbon sources and amino acids that suit the biological question for your experiment.
Test R. leguminosarum bv. viciae 3841 on RB carbon and RB amino acid minimal swim agar. RB carbon minimal swim agar contains all components of RB minimal medium, except D-mannitol and glycine, supplemented with 1 mM of a carbon source (e.g., fructose). For the composition of RB, see materials section.
Supplement organic acids (e.g., citric acid) with the sodium salt of the acid and determine the pH of the solution. Adjust the pH when needed.
RB amino acid minimal swim agar contains all components of RB minimal medium, except D-mannitol and glycine, supplemented with 1 mM of an amino acid (e.g., asparagine, arginine). The agar concentration is 0.3%.
To make the RB minimal carbon swim plates: add all the components from the Materials section, RB carbon until NaCl. Add agar. Then autoclave. Supplement 248.75 ml of this warm solution with filter sterilized solutions of the following:
250 μl of 1 M carbon source, that gives you 1 mM in the end.
250 μl of 0.01 mg/ml CoCl2
250 μl of 1 mg/ml thiamine
250 μl of 1 mg/ml biotine
250 μl of 1 mg/ml pyridoxin HCl
Pore ~20 ml into a petri dish and let sit until semi-sold. Inoculate next day with R. leguminosarum bv. viciae 3841 or other Rleg strain. You will need 100 μl of a culture to be spotted into the center of the plate.
To make the RB minimal amino acid swim plates: add all the components form the Materials section, RB amino acids until NaCl. Then autoclave, notice there is no ammonium sulfate. Supplement 245 ml of this warm solution with filer sterilized solutions of the following.
5 ml of 1 M amino acid, that gives you 1 mM in the end.
250 μl of 0.01 mg/ml CoCl2
250 μl of 1 mg/ml thiamine
250 μl of 1 mg/ml biotine
250 μl of 1 mg/ml pyridoxin HCl
Pore ~20 ml into a petri dish and let sit until semi-solid. Inoculate next day with R. leguminosarum bv. viciae 3841 or other Rleg strain. You will need 100 μl of a culture to be spotted into the center of the plate.
Production of the Rleg culture: Liquid inoculant cultures were grown in RB minimal media with mannitol for 3 to 4 days. The bacteria were pelleted at 10,000 ×g for five minutes and resuspended in fresh media with no nitrogen or carbon source. Inocula were standardized to an OD600 of 1 or as close as possible (±0.2).
Construct the composition of the minimal swim agar for A. brasilense Sp7 similarly. Use MMAB minimal medium as a base. MMAB carbon lacks malic acid, MMAB amino acid lacks malic acid and NH4Cl. Supplement with 1 mM carbon source for the carbon minimal swim plates and 1 mM amino acid for the amino acid minimal swim plates. Use 0.3% agar for the swim plates. For the composition of MMAB, see Table 1.
Production of the Abra culture: Liquid inoculant cultures were grown in TY liquid media for two to three days at 30°C. The bacteria were pelleted at 10,000 ×g for five minutes and resuspended in minimal MMAB without nitrogen or carbon source. Inocula were standardized to an OD600 of 1 or as close as possible (±0.2).
As a negative control for the R. leguminosarum bv. viciae 3841 or A. brasilense Sp7 experiments, produce swim plates from RB or MMAB minimal medium that do not contain the carbon or amino acid source. You can also use a mutant that does not swim.
Data collection: The bacteria will 'eat' up the nutrients in the center of the plate and migrate towards the gradient that they themselves produced. That means you will see a concentric ring forming around the inoculation spot. For Rleg, that can take several days. For Abra, expect about a day. Measure the diameter of the ring in regular time interval so you get 6 to 8 data points. If you plot the diameter against time, you should get a linear increase in diameter. Calculate cm or mm/h.
Statistical analysis. Conduct each experiment in at least 4 replicates. Present data as bar plots or box and whiskers. Perform statistical analysis with ANOVA, followed with Tukey that compared data from each carbon source or amino acid to the negative control.
Protocol references
Swim plates: Wolfe and Berg, 1989, PNAS. Migration of bacteria in semi-solid agar.
RB medium: Sourjik and Schmidt, 1996, Mol. Microbiol. Different roles of CheY1 and CheY2 in the chemotaxis of Rhizobium meliloti.
MMAB medium: Vanstockem et al., 1987. Appl. Environm. Microbiol. Transposon mutagenesis of Azospirillum brasilense and Azospirillum lipoferum: Physical analysis of Tn5 and Tn5-Mob insertion mutants
R. leguminosarum bv. viciae 3841: Johnston and Beringer, 1975. J. Gen. Microbiol. Identification of the Rhizobium strains in pea root nodules using genetic markers.
A. brasilense Sp7: Tarrand et al., 1987. Can. J. Microbiol. A taxonomic study of the Spirillum lipoferum group, with descriptions of a new genus, Azospirillum gen. nov. and two species, Azospirillum lipoferum (Beijerinck) comb. nov. and Azospirillum brasilense sp. nov.
Acknowledgements
Dr. Gladys Alexandre for providing Azospirillum brasilense Sp7
