Jan 22, 2020
Version 1
Measurement of Symbiodiniaceae cell density in Exaiptasia pallida tentacles V.1
- giada tortorelli1,
- Roy Belderok2,
- Simon Davy3,
- Geoff McFadden1,
- Madeleine van Oppen1
- 1University of Melbourne;
- 2University of Amsterdam;
- 3Victoria University of Wellington
- Aiptasia Symbiodiniaceae Model System

Protocol Citation: giada tortorelli, Roy Belderok, Simon Davy, Geoff McFadden, Madeleine van Oppen 2020. Measurement of Symbiodiniaceae cell density in Exaiptasia pallida tentacles. protocols.io https://dx.doi.org/10.17504/protocols.io.2i6gche
Manuscript citation:
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: May 02, 2019
Last Modified: January 22, 2020
Protocol Integer ID: 22846
Keywords: Exaiptasia pallida, model system, Symbiodiniaceae, symbiosis, microscopy, fluorescence., measurement of symbiodiniaceae cell density, symbiodiniaceae cell density, symbiodiniaceae uptake, colonization rate in exaiptasia pallida, hospite cell density, exaiptasia pallida, fluorescent microscopy, advantage of fluorescent microscopy, fluorescence, red chlorophyll auto, symbiont coverage, cell, fluorescent
Abstract
To monitor Symbiodiniaceae uptake and measure their colonization rate in Exaiptasia pallida, we extended the methods presented in earlier works by including a calibration curve that allows symbiont coverage to be expressed as in hospite cell density. Our approach takes advantage of fluorescent microscopy and symbionts red chlorophyll auto-fluorescence.
Materials
- Red Sea Water (RSW)
- Filtered Red Sea Water (FRSW)
- MgCl2 solution - 1:1 mixture of 0.37 M MgCl2 and FRSW
- 4% PFA/PBS
- 1x PBS
- 90% glycerol/PBS
- 1.5 ml tubes
- Pasteur pipettes
- Scalpels
- Watchglass
- Dissecting microscope
- Microscope slides and coverslips
- Clear nail polish
Troubleshooting
Safety warnings
Wear PPE at ll times.
Paraformaldehyde is highly toxic, hence handling must be performed in a fume hood.
SAMPLING & FIXING TENTACLES (DAY 1)
Anaesthetize the anemone by submersion in the MgCl2 solution for about 00:35:00 in the dark.
Gently move the anaesthetized anemone on a watchglass on the microscope, and try to have as less liquid as possible on the watchglass (this will facilitate tentacles excision).
With the microscope, focus on the tentacles. Use a scalpel to excise the needed number of tentacles (excise few extra tentacles as backup). Cut the tentacles as close as possible to the oral disk area but do not compromise the body or the oral disk of the anemone.
Add a drop of Red Sea Water (RSW) to the watchglass: this will facilitate the collection of the tentacles with a pasteur pipette. Then, submerge tentacles into a 1.5 tube filled with cold (4 °C ) 4% PFA/PBS solution. Keep the tubes at 4 °C for 01:00:00 .
Wash tentacles in PBS x2 times every 00:10:00 .
Leave fixed tentacles in PBS at 4 °C overnight.
SLIDES PREPARATION FOR TENTACLES ANALYSIS @ THE MICROSCOPE (DAY 2)
Place a small drop of 90% glycerol/PBS on a microscope slide and spread.
With a pasteur pipette, move the fixed tentacles from the tube onto the slide (try to have as less liquid as possible on the slide). Do not overlap tentacles. Gently remove extra PBS from the slide with a pasteur pipette or a kimwipe.
Apply coverslip onto the slides (do not apply any pressure to the tentacles, or sample would get ruined). Seal coverslip onto the slide with clear nail polish.
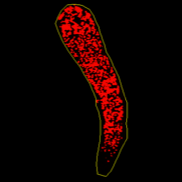
Store slides at 4 °C in the dark, and analyse samples for fluorescence on the same day. Microscope settings for a Leica M205 FA dissecting microscope, software LAS X Life Science Leica (a. brightfield, b. GFP LP fluorescence), are attached.
IMAGE ANALYSIS ON FIJI
Set Fiji:
- set measurements -> area, limit to threshold, max number of decimals (9)
Open Brightfield (BF) image on Fiji
Plugins -> macros -> run Step 1
This macro will:
- open the ROI manager with the "show all" option selected
Define the ROI (tentacle) with the polygon tool and add to the ROI manager (shortcut T), then measure the tentacle area (shortcut M)
Open the respective fluorescent (GFP-LP) image on Fiji
Plugins -> macros -> run Step 2
This macro will:
- split the color channels of the GFP-LP image
- keep the red channel (the symbiont chlorophyll auto-fluorescence)
- open the colour threshold window (with settings: default, red, dark background)
- restore the ROI (tentacle) created on the respective BF image
- delete the rest of the image and keep just the image content within the ROI
Manually adjust threshold to cover all the symbiont fluorescence shown in the selected red channel (it appears grey now). Apply threshold and press M to measure the area covered by the threshold (symbiont coverage).
Save measurements as .csv file.
DATA ANALYSIS
The data obtained are: TENTACLE AREA (mm2) and FLUORESCENT AREA (mm2)
Keep in mind the unit used to take the images at the microscope, because that would be the unit Fiji uses to take and express the measurements. In case the unit used is not mm2, then you need to convert your value.
The number of cells in each tentacle was plotted against the red fluorescent surface area within the same tentacle to establish a calibration curve allowing transformation of red fluorescent surface area into algal cell number.
Since these measures are proportional (R2= 0.948), we generated a calibration curve to extrapolate the number of symbiont cells represented by the measured fluorescence coverage with a simple equation: y= 1049.86 + 146.11x.
Hence, calculate the number of symbiont cells in the fluorescent area (CELLS IN FLUO) as:
CELLS IN FLUO= 1049.86+(146.11* FLUORESCENT AREA)
Then, calculate the number of symbiont cells (N SYMBIONTS) in the tentacle as:
N SYMBIONTS= CELLS IN FLUO/TENTACLE AREA
