Aug 11, 2025
Masson’s Trichrome staining for histology
- Samson Dowland1
- 1Charles Perkins Centre Histology Facility, The University of Sydney, NSW, 2006, Australia
- The University of Sydney

Protocol Citation: Samson Dowland 2025. Masson’s Trichrome staining for histology. protocols.io https://dx.doi.org/10.17504/protocols.io.e6nvwbwwwvmk/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: March 04, 2025
Last Modified: August 11, 2025
Protocol Integer ID: 123814
Keywords: histology, trichrome, histochemistry, staining, microscopy, pathology, histopathology, histological staining method, haematoxylin as the nuclear stain, nuclear stain, plasma stain, staining intensity, histology masson, contrast between tissue component, original red dyes for the plasma stain, fibre stain, other tissue component, tissue component, collagen yellow, collagen from muscle, collagen, embedded tissue, iron haematoxylin, green fibre stain, similar to the orginal masson, masson, contrast
Abstract
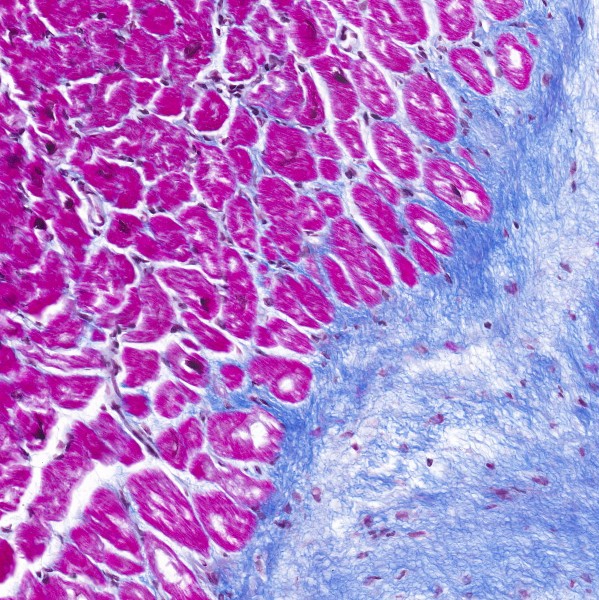
Masson's trichrome is a histological staining method commonly used to distinguish collagen from muscle, cytoplasm and other tissue components.
The method presented here remains close to the original used by Masson in 1929.
While there are many variants of the original method, there is commonality in that they all utilise three components - a nuclear stain (usually an iron haematoxylin), a fibre stain (to specifically dye the collagen) and a plasma stain (which acts as a counterstain). They also consistently use a polyacid to differentiate the plasma staining.
In this method it is noted that, if using routine formalin-fixed paraffin-embedded tissues, including the optional mordanting step will increase staining intensity and enhance contrast between tissue components. The same effect can also be achieved by using Bouin's fixative.
Nuclear stain: This method uses Weigert's haematoxylin as the nuclear stain by staining the nuclei dark purple-black. Some variants use Verhoeff's haematoxylin to additionally highlight elastin by staining it black.
Plasma stain: This method uses the original red dyes for the plasma stain, specifically a mixture of Acid fuchsin and Ponceau de xylidene. The latter has a number of alternate names, including Xylidine Ponceau, Xylidine Ponceau 2R, Ponceau 2R, Ponceau G, Ponceau Red, Acid Red 26. All have the same C.I. 16150.
Common variants use a mixture of Acid fuchsin and Biebrich scarlet, which provides a similar result, with a slightly darker red.
Heteropolyacid: Like the original, this method uses a 1% phosphomolybdic acid (PMA) solution for the heteropolyacid. Most variants use the same, but some recommend combining with phosphotungstic acid (PTA) in a 2.5% PMA + 2.5% PTA solution.
Fibre stain: The fibre stain used here is Methyl blue which stains collagen a vibrant, royal blue. This is similar to the orginal Masson's method, which was thought to use Aniline blue water soluble (a mixture of Water blue and Methyl blue). Some other variants use green fibre stains, such as Light green or Fast Green FCF. The Fast Green FCF resists fading better than Light green, which fades over time, causing sections to lose their contrast. One uncommon variant uses Metanil yellow to stain the collagen yellow.
Protocol materials
Hydrochloric AcidBanksia Scientific Company (AJAX FineChem)Catalog #AJA256
EthanolPOCD ScientificCatalog #ETHABS70WV5
EthanolHurst ScientificCatalog #EIMS95-5L
XylenePOCD ScientificCatalog #XYL5P
DPX Mountant for histologyMerck MilliporeSigma (Sigma-Aldrich)Catalog #06522
EthanolPOCD ScientificCatalog #ETHABS20M
HaematoxylinFronine Laboratory SuppliesCatalog #JL555S
Iron (III) chloride hexahydrateMerck MilliporeSigma (Sigma-Aldrich)Catalog #F2877
Acid FuchsinFronine Laboratory SuppliesCatalog #JL301
Ponceau de XylideneE. Gurr Ltd
Glacial acetic acidAjax Finechem (ThermoFisher Scientific)Catalog #AJA1-500ML
Phosphomolybdic acidMerck MilliporeSigma (Sigma-Aldrich)Catalog #221856
Methyl BlueMerck MilliporeSigma (Sigma-Aldrich)Catalog #M5528
Picric acidMay & Baker LTD
Troubleshooting
Deparrafinisation and rehydration
32m
Mordanting (optional)
1h 15m
Incubate in saturated picric acid solution at 57 °C
Picric acidMay & Baker LTD Saturated picric acid solution: 1.3 Mass Percent in distilled water
Safety information
Picric acid is an unstable explosive when dry, where it becomes highly sensitive to shock, heat and friction. Ensure it remains hydrated at all times.
Picric acid reacts with metals to form unstable picrate salts which are more explosive. Do not use metal containers, lids or racks.
Picric acid is toxic if swallowed, inhaled or absorbed through the skin.
Consult the MSDS and follow all recommended safety precautions.
1h
Cool to Room temperature
10m
Wash until no longer yellow
5m
Nuclear staining
13m
Stain in Weigert’s Haematoxylin (combine solution A & B immediately before use)
Solution A: 1 g Haematoxylin in 100 ml of 95% Ethanol
Solution B: 1.6 g Ferric chloride hexahydrate (Iron III chloride; FeCl3.6H2O) + 95 ml distilled water + 1 ml concentrated HCl
HaematoxylinFronine Laboratory SuppliesCatalog #JL555S CI: 75290
EthanolHurst ScientificCatalog #EIMS95-5L
Iron (III) chloride hexahydrateMerck MilliporeSigma (Sigma-Aldrich)Catalog #F2877
Hydrochloric AcidBanksia Scientific Company (AJAX FineChem)Catalog #AJA256
10m
Wash with running tap water to remove excess stain
2m
Differentiate with acid alcohol (1-2 dips) to clear the slides
[1% Hydrochloric Acid (HCl) in 70% Ethanol]Hydrochloric AcidBanksia Scientific Company (AJAX FineChem)Catalog #AJA256 EthanolPOCD ScientificCatalog #ETHABS70WV5
Wash in tap water
1m
Examine slides under microscope to assess nuclear staining. Nuclei should be dark purple-black and cytoplasm or collagenous tissue components should be grey. If nuclei are under-stained, return to step 5. If sections are over-stained, return to step 7.
Plasma stain
11m
Stain in Acid Fuchsin + Ponceau de xylidine solution (make fresh, lasts 2-3 days)
Solution: 1 g Acid Fuchsin + 1 g Ponceau de xylidine + 99 ml distilled water + 1 ml Glacial acetic acid
Acid FuchsinFronine Laboratory SuppliesCatalog #JL301 CI: 42685
Ponceau de XylideneE. Gurr Ltd CI: 16150
Glacial acetic acidAjax Finechem (ThermoFisher Scientific)Catalog #AJA1-500ML
5m
Wash in distilled water
Heteropolyacid (differentiation of plasma stain)
11m
Differentiate and mordant in 1% PMA solution 2 – 6 mins (check progress of differentiation under the microscope every 2 mins, until collagen is colourless)
PMA Solution: 1 g Phosphomolybdic acid + 100 ml distilled water
Phosphomolybdic acidMerck MilliporeSigma (Sigma-Aldrich)Catalog #221856
6m
Drain – do not rinse
Fibre stain
2m 30s
Stain in Methyl blue solution
Solution: 2 g Methyl blue + 2.5 ml Glacial acetic acid + 100 ml distilled water (solution can be re-used, lasts many years)
Methyl BlueMerck MilliporeSigma (Sigma-Aldrich)Catalog #M5528 CI: 42780
Glacial acetic acidAjax Finechem (ThermoFisher Scientific)Catalog #AJA1-500ML
2m
Wash in distilled water
Differentiate with 1% Glacial acetic acid
Glacial acetic acidAjax Finechem (ThermoFisher Scientific)Catalog #AJA1-500ML
30s
Dehydration, clearing and mounting
11m 30s
95% ethanol
EthanolHurst ScientificCatalog #EIMS95-5L
30s
100% ethanol
EthanolPOCD ScientificCatalog #ETHABS20M
Note: ensure whole slide and slide rack is fully immersed in ethanol to remove all traces of water, preventing water contamination of the xylene
30s
100% ethanol
EthanolPOCD ScientificCatalog #ETHABS20M
Note: ensure whole slide and slide rack is fully immersed in ethanol to remove all traces of water, preventing water contamination of the xylene
30s
100% Xylene
XylenePOCD ScientificCatalog #XYL5P
5m
100% Xylene
XylenePOCD ScientificCatalog #XYL5P
5m
Coverslip with a resinous mounting medium, such as DPX
DPX Mountant for histologyMerck MilliporeSigma (Sigma-Aldrich)Catalog #06522
Results
Expected result
Nuclei: Purple-black
Cytoplasm: Red
Keratin: Red
Erythrocytes: Red
Muscle: Red
Collagen: Blue
