Jan 08, 2026
LentiBlast Premium Lentiviral transduction enhancer
- Cedric Sapet1
- 1OZ Biosciences

External link: https://ozbiosciences.com/lentivirus-retrovirus/113-418-lentiblast-premium-transduction-enhancer.html#/
Protocol Citation: Cedric Sapet 2026. LentiBlast Premium Lentiviral transduction enhancer. protocols.io https://dx.doi.org/10.17504/protocols.io.x54v9bdwql3e/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: January 08, 2026
Last Modified: January 08, 2026
Protocol Integer ID: 238222
Keywords: Lentivirus, lentiviral transduction, transduction enhancer, viral enhancer, lentiblast premium lentiviral transduction enhancer lentiblast premium, lentiblast premium, lentiviral infection, viral fusion with cell membrane, enhancing viral fusion, viral particle, transmembrane potential change, cell membrane, membrane permeable effect, cell viability, compatible with cell viability, cell, electrostatic repulsions between membrane, cell line, membrane
Disclaimer
For Research Use Only. Not for use in humans. Not for use in diagnostic or therapeutic purposes.
Abstract
LentiBlast Premium is a novel patented chemical composition that dramatically increases lentiviral infection and transduction efficiency in any type of cells, adherent or in suspension, primary or cell lines. The properties of this reagent allow simultaneously neutralizing electrostatic repulsions between membrane and viral particles and enhancing viral fusion with cell membrane. Due to a favorable “membrane permeable effect” limiting the transmembrane potential changes, LentiBlast Premium is non-toxic and totally compatible with cell viability.
Attachments
Guidelines
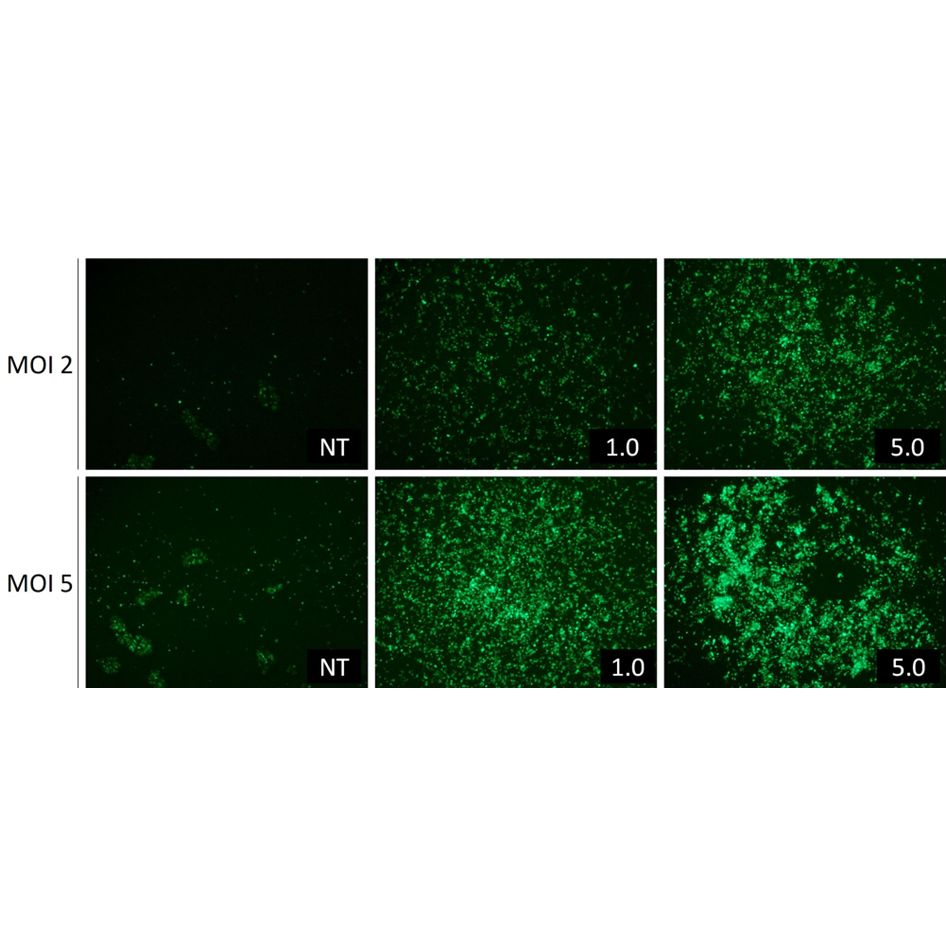
If the lentiviral transduction/infection conditions are unknown, we recommend starting with a MOI of 2 using a lentiviral vector encoding for a fluorescent protein.
Materials
LentiBlast Premium Reagent (OZ Biosciences REF LBPX500 - REF LBPX1500)
Troubleshooting
Problem
Low transduction efficiency
Solution
■ MOI used is low. Raise the lentiviral particles amount: Up to MOI 100 can be used.
■ Infectious viral titer is low. Check viral titer via transduction of HEK-293 cells with serial dilutions of lentivirus.
■ Cells are difficult to transduce or non permissive to virus serotype: perform a spinoculation step after addition of LBP + viral particles or use Magnetofection with ViroMag RL as an alternative to chemical enhancers.
■ No effect of LentiBlast Premium is observed. Concentrations of LentiBlast is too low, try increasing volume of LBP
Safety warnings
Do not use LentiBlast Premium with another viral enhancer or adjuvant.
Before start
We recommend minimizing freeze-thaw cycles to preserve LentiBlast Premium integrity.
Protocol | lentiviral transduction enhancement with LentiBlast Premium
3h
Cell culture prior to transduction: the day before transduction, prepare the cells according to table 1 below. Cells should be 20-50 % confluent at the time of transduction.
| A | B | |
| Tissue culture dish | Cell number | |
| 96-well plate | 3-8 x 1.10e3 | |
| 24-well plate | 2-4 x 1.10e4 | |
| 6-well plate | 1-2 x 1.10e5 |
Table 1: Suggested cell number for lentiviral transduction per well.
CLASSIC PROTOCOL
LentiBlast Premium is 100 times concentrated (100X), we recommend trying 3 final concentrations of 0.5X, 1X and 2X final corresponding to 0.5, 5 and 10 µL of LentiBlast Premium per condition respectively in a 24-well plate.
Virus preparation: dilute virus into culture medium sufficient for 4 samples (50 µL per sample).
Note
If the lentiviral transduction/infection conditions are unknown, we recommend starting with a MOI of 2 using a lentiviral vector encoding for a fluorescent protein.
Dispatch equal volume of viral suspension into 4 tubes.
LentiBlast Premium: add 0, 0.5, 5 and 10 µL of LentiBlast Premium to each tube.
0 µL : Lentiviral particles only
Mix vials by inverting.
Add viral suspensions to cells and incubate 24 H under standard culture conditions.
Note
Optionally change medium.
Incubate cells 24 to 96 H: incubate the cells under standard culture conditions.
CENTRIFUGATION PROTOCOL
For hard-to-transduce cells, it is recommended to add a centrifugation step to the standard protocol. Cells are prepared on the day of transduction, counted, pelleted and suspended in Lentivirus/LentiBlast Premium mixes.
Detach and seed cells into 5 wells, refer to table 1 for suggested cell density.
Follow steps 2.1 to 2.4 of the standard protocol and add LentiBlast Premium/lentivirus mixes to cells.
Centrifuge the plate 900 rpm x 90 min.
900 rpm, 25°C, 01:30:00
3h
Incubate cells overnight and proceed to the remaining steps of the standard protocol.
Protocol references
- Zhang P. – 2025. Systematic evaluation of GAPs and GEFs identifies a targetable signalling hub in cancer (titre abrégé d’après la notice). Cancer Discovery 15(12):2530‑2553. PDX cells, lentivirus. DOI: 10.1158/2159‑8290.CD‑24‑0972.
- Pellegrini JM. – 2025. SLAMF7 and SLAMF8 receptors shape human plasmacytoid dendritic cell responses (titre d’après PDF JCI). Journal of Clinical Investigation 135(8):e182467. CAL‑1, rétro/lentivirus. DOI: 10.1172/JCI182467.
- Yan J. – 2025. Titre non reproduit ici. Leukemia (article en ligne, T lymphocytes, lentivirus). Réf.: doi:10.1038/s41375‑025‑02652‑0.
- Burton OT. – 2024. Titre non reproduit ici. Immunity S1074‑7613(24)00277‑2. Hematopoietic stem cells, rétro/lentivirus. DOI: 10.1016/j.immuni.2024.06.xxx (voir PubMed).
- Liu X. – 2024. Titre non reproduit ici. Journal of Clinical Investigation e175619. Acute myeloid leukemia (AML), rétro/lentivirus. PMID: 38713535.
- Ma L. – 2022. Tet‑mediated DNA demethylation regulates specification of hematopoietic stem and progenitor cells (AGM ex vivo). Science Advances 8(9):eabm3470. AGM ex vivo, rétro/lentivirus. DOI: 10.1126/sciadv.abm3470.
- Claireaux M. – 2022. Titre non reproduit ici. Nature Communications 13(1):521. CD4+ T lymphocytes, rétro/lentivirus. DOI: 10.1038/s41467‑021‑27890‑x (voir PubMed).
- Nassour J. – 2023. Titre non reproduit ici (telomeres & cancer). Nature 614(7949):767‑773. HMEC (et autres), rétro/lentivirus. DOI: 10.1038/s41586‑022‑05645‑4 (voir notice auteur).
- Patel AS. – 2021. Titre non reproduit ici (small cell lung cancer). Science Advances 7(5):eabc2578. SCLC, lentivirus. DOI: 10.1126/sciadv.abc2578.
- Arnoult N. – 2017. Titre non reproduit ici. Nature 549:548‑552. HT1080, rétro/lentivirus. DOI: 10.1038/nature24023.
Acknowledgements
Purchaser Notification | Conditions of Sale
This product is sold in accordance with our general conditions of sale that you can find on our website: https://ozbiosciences.com/content/3-terms-and-conditions.

