Oct 21, 2025
Kirigami Restraint of Danionella Cerebrum for Long-Term Live Imaging
- Avery Wang1,
- Adrian Wang2,
- Gordon Wang1,
- James Jaggard1,
- Philippe Mourrain1
- 1Stanford University;
- 2Stony Brook University

Protocol Citation: Avery Wang, Adrian Wang, Gordon Wang, James Jaggard, Philippe Mourrain 2025. Kirigami Restraint of Danionella Cerebrum for Long-Term Live Imaging. protocols.io https://dx.doi.org/10.17504/protocols.io.14egnrzr6l5d/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: In development
We are still developing and optimizing this protocol
Created: July 25, 2025
Last Modified: October 22, 2025
Protocol Integer ID: 223303
Keywords: Danionella Cerebellum, Microscopy, Imaging, 2 Photon Microscopy, Sleep, Calcium Imaging , Ca2+ Imaging, long term imaging of the fish brain, fish brain, term live imaging danionella cerebellum, kirigami restraint of danionella cerebrum, sensitive fish, vertebrate brain, exciting new experimental fish model, known vertebrate brain, microscopy study of the brain, known zebrafish, kirigami restraint method, transparent adult vertebrate, fish health, fish, kirigami restraint, gill, imaging, neural activity during sleep, long term imaging, danionella cerebrum, neural activity, brain, danionella adult, danionella, silicone restraint, microscopy study
Funders Acknowledgements:
NIH
Grant ID: R01NS104950
NIH
Grant ID: R01HL151576
NIH
Grant ID: P50HD109861
Abstract
Danionella Cerebellum is an exciting new experimental fish model that is a relative of the much better known zebrafish (Danio rerio). Danionella has a number of advantages for microscopy study of the brain that includes being a fully transparent adult vertebrate, and having the smallest known vertebrate brain. Danionella adults are highly social and sensitive fish that is difficult to restrain for more than several hours. Current methods often involve the use of agarose and optical adhesive, which can adversely affect fish health and require forced flow of water through its gills. We developed an kirigami restraint method that uses paper and silicone restraints to hold fish that provide over 24hr restraint and imaging that does not require external perfusion. This method allows the gentle, long term imaging of the fish brain for studies that require long-term immobilization, such as the measurement of neural activity during sleep and wake transitions.
Materials
Dow Corning - Sylgard 184 Silicone Elastomer
Aqueon - 100% Silicone Aquarium Sealant
Corning - Cover Glass No. 1.5 24x50mm
Kimberly Clark - KimWipe Delicate Task Wipers
Thorlabs - M6 Hardware Kit
Dumostar - 55 Tweezer
Ecelta - 2ASA Tweezer
Troubleshooting
Safety warnings
Avoid UV cured glues. We test a number of UV cured optical adhesives (Norland Optics: NOA68, 72, 74, MasterBond LED405MED, UV10TKMED and Bondic) and they are all toxic to Danionella. Prewashing the cured glue does appear to help, but does not completely remove the toxicity.
Ethics statement
The protocols.io team notes that research involving animals and humans must be conducted according to internationally-accepted standards and should always have prior approval from an Institutional Ethics Committee or Board.
Fabricate Coverslip Mount
2h 10m
3D print mold for mounting feet (Use Included STL file)
Prepare 1ml per mold of Sylgard 184 silicone elastomer
Prepare mold by covering one side with packing tape
Note: This makes elastic feet easier to remove from mold
Fill mold with prepared silicone elastomer
Place filled mold in oven at 55 deg C (2hr or overnight)
Note: The elastomer will cure at room temperature. In this case, overnight is recommended
2h
Remove tape and pop out silicone feet
Quick rinse with water
Glue silicone feet to coverglass with silicone aquarium glue
Note: Glue feet ~2 mm apart (see figure & STL file)
Take care not to put silicone glue between the two feet. This will obscure the imaging window. Mount.stl5KB
Mount.stl5KB
Cure silicone glue
Soak and wash with water
10m
Create Origami Restraint
Grab a Kimwipe and fold it in half
Fold one side of the Kimwipe into itself to create a side with 4 layers.
Note: This allows you to make two restrains with one set of cuts.
Cut off a 4.5mm by 90mm long strip of paper on the folded side. See pictures for reference.
On that strip of paper, cut a small triangular notch, with base ~2mm and height ~23mm.
Note: Make sure that the notch is cut into the side where the paper is connected.
On the opposite side of the strip, cut off a long tapering strip that is a triangle with base ~2mm tapering to the end.
Note: It is important that the base of the long tapering strip is shorter than the base of the opposite notch, and that the distance between the base of the strip and the base of the notch is ~3mm.
Unfold the cut paper. Wet the paper for next step.
Note: We normally use both restraints for added durability, but a single restraint can be used by itself.
Origami Tie Fish to Coverslip Mount
Lay the wet, cut restraint over the coverslip mount. The 3mm center should adhere to between the two silicone feet. Make sure there the paper is not obscuring the top triangle where the fish head will be.
Place the Danionella in MS222 (460μM - 120mg/L) until its mostly unconscious 30secs to 45secs. MS222 will stop the Danionella from breathing. Minimize fish time in MS222.
Transport the knocked out Danionella onto the mount. You can either directly place the fish between the silicone feet, or on top of the one of the feet and then gently flip the fish into the trough. We found the later to be easier, but both work. Note: Maneuvering the fish is better achieved with a blunt end tweezer such as a Ecelta - 2ASA tweezer.
Using tweezers, carefully without harming it, orient the Danionella into the mount dorsal down.
Place a drop of fresh fish water on the Danionella.
Using two tweezers, pull the smaller section of the paper all the way through the hole. To do this, place the tip of the narrow side, into the wedge opening of the thicker side. Lift the thicker side. Flip it over the opposite silicone feet. Pull the thin side through while still holding the thicker side. Note, there is no need to use too much force. The wet paper should adhere to the glass. If it does not remove some excess water. Now Lift the cover glass with one tweezer and use the other to flip the excess paper to the opposite side of the coverglass.
Note: Make sure it is not too tight or the Danionella will die. Adjusting the fish orientation in the forward and backward direction can be achieved by prodding the fish with a blunt tweezer. Adjusting the axial rotation of the fish can be achieved by tugging on the left and right paper.
Put mounted fish into imaging dish
Place the restrained fish dorsal side up into a dish with fish water. We use stainless steel nuts as spacers under coverglass. We use Pyrex 60mm X 25mm dishes. There is no perfusion necessary.
To secure the mounted fish, place stainless screws on top of the coverglass. The threads provide easy handling. You are now ready to image the fish with an upright microscope. The glass will allow you to image with air, silicone, glycerine, oil or water objectives. Note: If you are using a large bore imaging objective the threads might protrude too much. In that case, stainless steel nuts can be used to secure the mount.
Note: Danionella is a highly social fish. They can be severely stressed if isolated. It helps to place one or two unrestrained fish into the imaging chamber. 100nM Allopregnanolone can also be used to reduce anxiety.
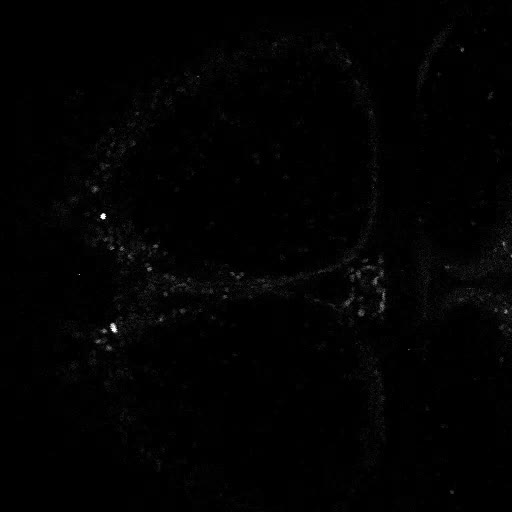
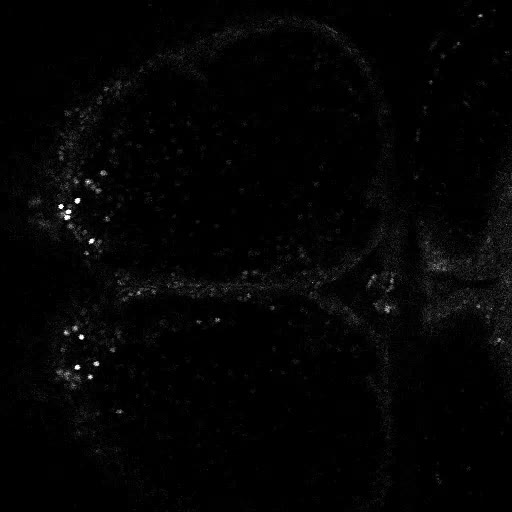
Movies of live 2 photon imaging of restrained Danionella. Movies acquired on a Leica Stellaris DIVE with at 16X 0.8NA water dipping objective. Neurons are labeled transgenically with GCaMP6s. Images (512x512) are captured bidirectionally at 2.7fps, and sped up to 30fps in the movies.