Aug 02, 2025
Isolation of brain eGFP-cholinergic interneurons by dissociation and flow cytometry
- Yu-En Lin1,2,
- Shahzad Khan1,2,
- Claire Y Chiang1,2,
- Ebsy Jaimon1,2,
- Suzanne Pfeffer1,2
- 1Stanford University School of Medicine;
- 2Aligning Science Across Parkinson's

Protocol Citation: Yu-En Lin, Shahzad Khan, Claire Y Chiang, Ebsy Jaimon, Suzanne Pfeffer 2025. Isolation of brain eGFP-cholinergic interneurons by dissociation and flow cytometry. protocols.io https://dx.doi.org/10.17504/protocols.io.dm6gpmbydgzp/v1
Manuscript citation:
L.C. Foo, 2013, Cold Spring Harb Protoc; 2013; doi:10.1101/pdb.prot074229
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: July 31, 2025
Last Modified: August 06, 2025
Protocol Integer ID: 223737
Keywords: ASAPCRN, Neuron dissociation, ChAT neuron isolation, cholinergic interneurons by dissociation, expressing cholinergic interneuron, cholinergic interneuron, isolation of brain egfp, dissected mouse dorsal striatum tissue, mouse dorsal striatum tissue, single cell isolation of gfp, brain egfp, single cell isolation, cytometry the method
Funders Acknowledgements:
Aligning Science Across Parkinson's
Grant ID: ASAP-000463
Abstract
The method enables single cell isolation of GFP-expressing cholinergic interneurons from dissected mouse dorsal striatum tissue. The protocol is based on a method of Lynette Foo (2013).
Materials
- Cell strainers (100µm and 40µm, Fisher Scientific
- HBSS (Hank's Balanced Salt Solution) (w/o Ca2+, Mg2+, and Phenol Red) (GIBCO)
- Enzyme Stock Solution (ESS) (see below)
- Low-ovomucoid (low-OVO) inhibitor solution (see below)
- High-ovomucoid (high-OVO) inhibitor solution (see below)
- Papain (Worthington Biochemical Cat#: LS003126)
- DNase I (Worthington Biochemical Cat#: LS002139) (prepare 0.4% w/v in 1mL EBSS)
- 0.22μm filter
- Surgical tools
- 1mm acrylic coronal adult mouse brain slicer matrix
- control mouse
- eGFP-Chat mouse
- Dissection Buffer (DB) - HBSS
- Digestion Buffer (DgB) - ESS + 5 units/ml DNase I + 1mM L-cysteine; add 20 units/ml papain later and use within 20 minutes of addition of papain
- HBSS + 0.02% BSA (w/v)
- FACS buffer (HBSS + 1% BSA)
- Gentle MACS C-tube
- Gentle MACS dissociator
- Fluorescence-activated cell sorter (FACS) (We use a SONY SH800)
Enzyme Stock Solution (prepare 200ml as follows):
20ml, 10x EBSS (Eagle's Balanced Salt Solution; Sigma-Aldrich E7510)
2.4 ml, 30% D-glucose (final 0.46%)
5.2 ml, 1 M NaHCO3 (final 26 mM)
2ml, 50 mM EDTA (final 0.5 mM)
Bring the volume to 200 mL with ddH2O and filter-sterilize using a 0.22μm filter
High-Ovomucoid (High-Ovo) 10X Stock Solution (prepare 200ml as follows):
6 g BSA (Sigma-Aldrich A8806) dissolved in 150 mL Dulbecco's (D)-PBS (Cytiva)
6 g trypsin inhibitor (Worthington LS003086)
~1.5 mL 1 N NaOH to adjust pH to 7.4
-Bring the volume to 200 mL with D-PBS, filter-sterilize (0.22μm filter), aliquot, store at −20°C
Low-Ovomucoid (Low-OVO) 10X Stock Solution (prepare 200ml as follows):
3 g BSA (Sigma-Aldrich A8806) dissolved in 150 mL D-PBS
3 g trypsin inhibitor (Worthington LS003086)
~1 mL 1 N NaOH to adjust pH to 7.4
Bring the volume to 200 mL with D-PBS, filter-sterilize (0.22μm), aliquot, store at −20°C
Inhibitor Stock Solution (prepare 500ml as follows):
50 mL,10x EBSS (Sigma-Aldrich E7510)
6 mL 30% D+-glucose (final 0.46%)
13mL, 1 M NaHCO3 (final 26 mM)
Bring the volume to 500 mL with ddH2O, filter-sterilize using a 0.22μm filter
Troubleshooting
Solution Preparation
Prepare solutions: ESS, ISS, 10X High OVO stock, 10X Low OVO stock, DNase (0.4%).
Solution Equilibration
Aliquot and equilibrate the ESS (22 mL) for 1 hour as follows:
Open the container and transfer to a 34°C incubator with 5% CO2/95% O2.
After equilibration, prepare 15 mL Digestion Buffer and transfer to a 34°C water bath; do not add papain for now.
Aliquot and equilibrate the ISS (2 X 21 mL and 1 X 10 mL) for 1 hour as in STEP 2.1.
After equilibration of ISS, transfer to room temperature.
Prepare High- and Low-OVO inhibitor solutions (10, 21, 21 mL for one sample) (3 TOTAL solutions)
High OVO solution: mix 10 mL ISS + 2 mL 10X High OVO stock + 20 µL 0.4% DNase
2, Low OVO solutions: for each of these mix 21 mL ISS + 1.5 mL 10X Low OVO stock + 100 µL 0.4% DNase; label them as “Tube 1 Low OVO” and “Tube 2 Low OVO”.
When the mice are ready to be sacrificed (within 20 min), add papain to Digestion Buffer (pre-warmed to 34°C in a water bath).
Brain dissection
Add 10 mL HBSS to a 6cm petri dish on ice.
Mice are euthanized by CO2.
Brains are quickly dissected, and whole brains placed in dissection buffer in the 6cm petri dish on ice.
Brains are quickly placed into a 1mm acrylic coronal adult mouse brain slicer matrix and 1-2mm sections are cut using standard razor blades.
The dorsal striatum is carefully dissected from each brain (2 pieces per brain).
Dissociation of cells
Brain sections are placed into pre-warmed digestion buffer containing papain (3 ml/brain) in a 6cm petri dish then transferred to a Gentle MACS C-tube and chopped with a Gentle MACS dissociator (“02_02” setting for mouse brain).
The homogenate is incubated for 45 minutes at 37°C in an incubator and then triturated with a 5ml pipette (up and down gently 10 times) every 15 minutes during this incubation. The sample does not appear dissociated but fewer chunks will be observed after 45 minutes.
Brain samples are further dissociated using Gentle MACS “01_02” setting for mouse brain.
Transfer the digested sample into a 50-mL conical tube, wait for the material to settle, then aspirate and discard the excess liquid.
Wash the cells by adding 4 mL of low-ovo inhibitor solution from Tube 1. Wait for the cells to settle, and aspirate and discard the excess liquid.
Repeat step 15 three times; the remaining low-ovo volume of tube 1 should be ~5 mL (4 x 4 = 16, 21-16 = 5 mL).
Add 4 mL of low-ovo inhibitor solution from Tube 2 to the conical tube for trituration.
Use a 5-mL serological pipette to quickly suck up and release the sample + low-ovo inhibitor solution. As the tissue dissociates, the low-ovo inhibitor solution will become cloudy. Be careful not to introduce bubbles.
Allow the tissue to settle. A cloudy, single-cell suspension will form above the chunks of undissociated tissue. Collect the dissociated cells with a 1-mL pipette and add them to 5 mL fresh low-ovo inhibitor solution remaining in Tube 1 from Step 16.
Repeat the trituration (Steps 17-19) four times, until almost all the brain chunks have dissociated into single cells. (4 + 4 x 4 = 21, tube 2 will be almost empty after this)
10 mL of High OVO solution is added carefully to the bottom of the single-cell suspension and the tubes are centrifuged at 110g for 5 minutes.
The supernatant is removed, and the cell pellet resuspended in 3 mL, HBSS + 0.02% BSA, pipetting up and down with a 1-mL pipette. After the cells are resuspended, add 9 mL HBSS + 0.02% BSA.
Pre-wet 100µm and 40µm cell strainers by adding 1 mL, HBSS + 0.02% BSA
The cells are passed through a 100µm cell strainer and then a 40µm cell strainer.
Cells are centrifuged at 100Xg for 10 minutes. The supernatant is removed and 1ml of FACS buffer (HBSS + 1% BSA) is added.
Flow Cytometry
On the Sony SH800 cell sorter, select the 488 filter during set up and use FSC-3%, BSC-29.5%, 38% 488 EM-gain. We use the 100µm sorting chip.
Using your negative control (non-eGFP expressing) sample, run 10,000 events (cells) through the sorter to analyze the cell population.
Using a graph showing FSC-A on the X-axis and BSC-A on the Y-axis, select a circular region that represents the desired population of "larger" cells (right side of the graph). We will call this population A. This population will contain cholinergic neurons that have larger then average soma diameters.
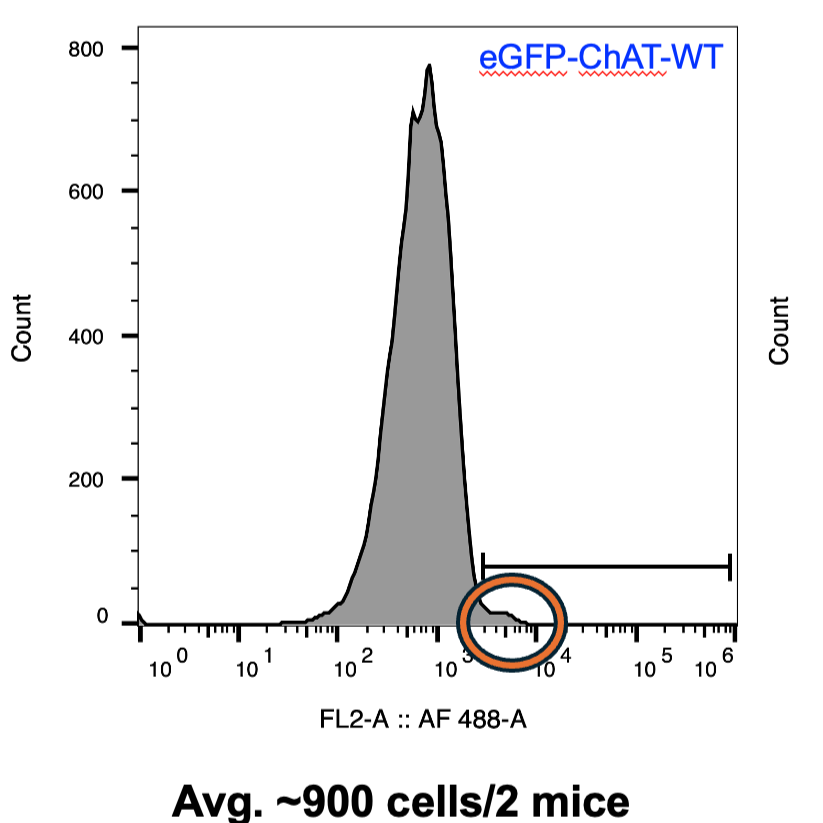
Using another graph containing events only from population A, display the 488-signal on the X-axis and number of events on the Y-axis. This should display a histogram with a normal distribution. This is the intrinsic GFP-signal from your cell population. Draw a linear gate on this graph starting from just off the right side of this histogram toward the high 488-end of the graph (right side).
Run your eGFP-positive sample through the sorter, at least a total of 5,000 cells. Do not collect these cells, this is only used for analysis to determine the population to collect. Display the same graphs displaying the population A gate and linear gate from your negative control sample (these should auto-populate).
Using the graph showing 488-signal on the X-axis and number of events or cells on the Y-axis, you should see a histogram of normal distribution similar to that of the negative control sample, with an additional small shoulder to the right (see protocol image). This small peak should be contained within the linear gate that was drawn in step 29; the rest of the histogram should not be within this boundary. If it is, adjust the gate accordingly.
Initiate sorting and collect the population delineated by the linear gate described above. Run your full sample through the sorter and collect all the cells in this population into your desired buffer for downstream applications. For mass spectrometry we collect cells directly into RIPA lysis buffer.
Protocol references
Foo, L.C. (2013) Purification of Astrocytes from Transgenic Rodents by Fluorescence-Activated Cell Sorting. Cold Spring Harb Protoc; 2013; doi:10.1101/pdb.prot074229
Acknowledgements
This protocol is an expansion of the method of Lynette C. Foo (2013).
