Aug 21, 2025
Infiltration and rubbing with BSMV or FoMV vectors for VMRG
- Macarena Mellado-Sanchez1,
- Faye Oddy1,
- Peggy McGroary1,
- Guilherme Menegol Turra1,2,
- Dana Macgregor1
- 1Rothamsted Research;
- 2UFRGS
- Dana Macgregor: https://orcid.org/0000-0003-0543-0408, [email protected];

Protocol Citation: Macarena Mellado-Sanchez, Faye Oddy, Peggy McGroary, Guilherme Menegol Turra, Dana Macgregor 2025. Infiltration and rubbing with BSMV or FoMV vectors for VMRG. protocols.io https://dx.doi.org/10.17504/protocols.io.5jyl8drr6g2w/v1
Manuscript citation:
Yuan, C., et al. (2011). "A High Throughput Barley Stripe Mosaic Virus Vector for Virus Induced Gene Silencing in Monocots and Dicots." PLOS ONE 6(10): e26468. into Nicotiana benthamiana plants and then rub inoculation of the virus-infected sap into target plants to induce Virus Induced Gene Silencing (VIGS).
Lee, W.-S., et al. (2015). "Virus induced gene silencing (VIGS) for functional analysis of wheat genes involved in Zymoseptoria tritici susceptibility and resistance." Fungal Genetics and Biology 79: 84-88.
Mellado-Sánchez, M., et al. (2020). "Virus-mediated transient expression techniques enable gene function studies in black-grass." Plant Physiology 183(2): 455-459.
Mellado-Sánchez, M., et al. (2020). "Virus-mediated transient expression techniques enable genetic modification of Alopecurus myosuroides." bioRxiv: 2020.2001.2028.923466
Bouton, C., et al. (2018). "Foxtail mosaic virus: A Viral Vector for Protein Expression in Cereals." Plant Physiology 177(4): 1352-1367.
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: October 21, 2024
Last Modified: August 21, 2025
Protocol Integer ID: 110604
Keywords: Infiltration, VIGS, Agroinfiltration, Barley Stripe Mosaic Virus Vector, Foxtail Mosaic Virus Vector, Rub inoculation, modified barley stripe mosaic virus vector, high throughput barley stripe mosaic virus vector, barley stripe mosaic virus vectors from yuan, virus induced gene silencing, foxtail mosaic virus, virus mediated overexpression, inoculation of the virus, mosaic virus, functional analysis of wheat gene, viral vector, wheat gene, viral vector for protein expression, nicotiana benthamiana plant, virus, sap into target plant, fungal genetics, infected sap, fomv vectors for vmrg, target plant, vig, gene function study, genetic modification of alopecurus, plant physiology, fomv vector, genetic modification, gene, monocot
Funders Acknowledgements:
Dana MacGregor
Grant ID: Smart Crop Protection Industrial Strategy Challenge Fund (grant no. BBS/OS/CP/000001)
Abstract
This protocol details the infiltration of modified Barley Stripe Mosaic Virus vectors from Yuan, C., et al. (2011). "A High Throughput Barley Stripe Mosaic Virus Vector for Virus Induced Gene Silencing (VIGS) in Monocots and Dicots." PLOS ONE 6(10): e26468. into Nicotiana benthamiana plants and then rub inoculation of the virus-infected sap into target plants to induce Virus Induced Gene Silencing / VIGS or Virus Mediated Overexpression / VOX.
Further details see
Lee, W.-S., et al. (2015). "Virus induced gene silencing (VIGS) for functional analysis of wheat genes involved in Zymoseptoria tritici susceptibility and resistance." Fungal Genetics and Biology 79: 84-88. http://dx.doi.org/10.1016/j.fgb.2015.04.006
Mellado-Sánchez, M., et al. (2020). "Virus-mediated transient expression techniques enable gene function studies in black-grass." Plant Physiology 183(2): 455-459. Accession Number: 32238443 PMCID: PMC7271810 DOI: DOI:10.1104/pp.20.00205
Mellado-Sánchez, M., et al. (2020). "Virus-mediated transient expression techniques enable genetic modification of Alopecurus myosuroides." bioRxiv: 2020.2001.2028.923466 DOI: 10.1101/2020.01.28.923466
Bouton, C., et al. (2018). "Foxtail mosaic virus: A Viral Vector for Protein Expression in Cereals." Plant Physiology 177(4): 1352-1367. http://dx.doi.org/10.1104/pp.17.01679
Image Attribution
Image drawn by Macarena Mellado-Sanchez
Troubleshooting
Procedure
1d 1h 20m
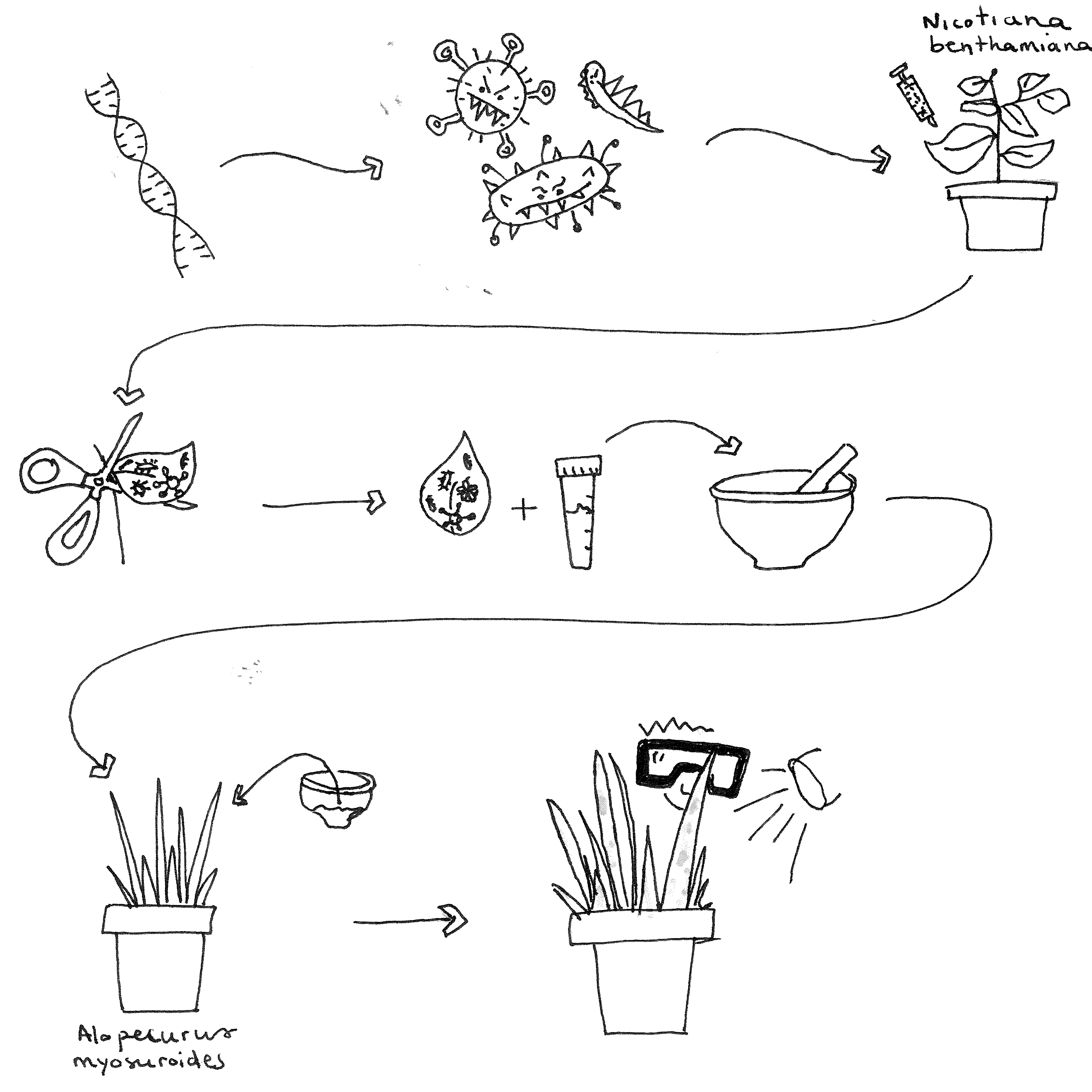
Pictorial representation of protocol - Drawn by Macarena Mellado-Sanchez
Grow at least three Nicotiana benthamiana plants for each construct in Levington® Advance F2+S Seed & Modular Compost + Sand Compost, in a controlled environment room with 16:00:00 photoperiod, 23 °C – 20 °C (day – night), 130 μmol m−2 s−1 light intensity and 60% relative humidity.
Note
Ensure plants have at least three fully expanded leaves before infiltrating.
4w
Pick a generous amount of Agrobacterium confirmed to contain BSMVα, BSMVβ and BSMVγ from fresh agar plates or glycerol stocks kept at -80 °C and inoculate 5 mL LB broth (Lennox) containing antibiotics kanamycin (50 μg/ml ) and gentamicin sulphate (25 μg/ml ) in universal tubes and incubate for approximately 20:00:00 -22:00:00 at 28 °C on a shaker at 250 rpm .
Note
Remember to include negative and positive control(s) as needed. Empty Vector (EV) and Non-target controls (NC) are recommended.
Note
Ensure you start a BSMVα and BSMVβ for each BSMVγ you are using. It is often useful to use the 5ml overnight to inoculate larger volume LB (e.g. 25 mL or 50 mL ) and incubate an additional 1-2 nights (particularly of the BSMVα and BSMVβ ) to ensure there is sufficient bacterial culture for infiltration.
Note
Alternatively start Agrobacteria confirmed to be carrying modified FoMV and P19.
22h
Spin down cells for 3500 rpm, 00:20:00 at 15 °C -17 °C .
Note
Do not spin down more than 30ml in 50ml Falcon tubes or else it does not form pellet.
20m
Re-suspend agrobacteria in inoculation buffer (10 millimolar (mM) MgCl2, 10 millimolar (mM) MES pH 5.6 supplemented with 150 micromolar (µM) acetosyringone) to a final OD600 of 1.5 for BSMV
| A | B | C | |
| Agroinfiltration buffer | For 50 mL | Working concentrations | |
| 2M MgCl2 | 250 µl | 10 mM | |
| 0.5 M MES | 1 ml | 10 mM | |
| 100 mM acetosyringone* | 75µl | 150 µM | |
| RO water | 48.67 ml | ||
| *Acetosyringone – 0.0196 g/ml. Dissolve in DSMO. Store at -20°C in small aliquots. | |||
Note
Acetosyringone must be added right before used.
Note
Bacterially contaminated LB broth waste has to be disposed as GMO waste.
Note
If using FoMV constructs the final concentration is OD600 of 0.6, and 0.3 for P19.
Incubate at Room temperature without shaking for at least 03:00:00 .
3h
Mix equal volumes of BSMVα, BSMVβ and BSMVγ strains at OD600 1.5 together.
Note
If using FoMV - mix equal volumes of FoMV construct at OD600 0.6 and P19 at OD600 0.3.
Infiltrate the Infiltration Mix into leaves (abaxial side) of ~ 4-week-old Nicotiana benthamiana.
- Press a 1ml syringe tip without a needle flat against the leaf with enough pressure to seal but without damaging the leaf.
- Infiltrate the bacterial mix into the leaf slowly trying to fill the leaf completely.
- Infiltration can be easier starting from the part of the leaf where the cross is where no big plant veins are.
Recommended region to start infiltration
Note
If using FoMV, infiltrate FoMV and P19 mixture into leaves.
3h
Allow the infiltrated N. benthamiana plants to grow in the controlled environment room with 16:00:00 photoperiod, 23 °C – 20 °C (day – night), 130 μmol m−2 s−1 light intensity and 60% relative humidity for 3-5 days
- NOTE: If using FoMV, then allow to grow for 5-7 days
5d
Cut the infiltrated leaves from N. benthamiana plants at petiole 3-5 days for BSMV (5-7 days for FoMV) post agroinfiltration. Best practice is to use one leaves from three different plants for one inoculum. Record the weight of the leaves using a fine scale precision balance. Use leaves immediately for rub inoculation or freeze in liquid nitrogen for future experiments. If storing, seal three leaves inside a foil packet and freeze immediately. Record weight, date and construct infiltrated on the outside of the packet and store at -80 °C .
When ready to do rub inoculation use fresh/frozen tissue. Grind leaf material using a chilled mortar and pestle. Add 10 millimolar (mM) potassium phosphate buffer pH 7 at 2 times the weight of N. benthiamiana leaves (ex: 2 mL for 1 g of infiltrated N. benthiamiana leaf). Wait until the material is totally thawed before rubbing onto leaf.
Add a fine layer of abrasive (e.g. Celite or Carborundum) to the 2nd leaf of the target plant.
Note
Abrasive can also be added to the 10mM potassium phosphate buffer and ground leaves directly.
Note
Using a salt shaker or similar is helpful to ensure a fine layer is added.
For rubbing grass leaves, wet your gloved fingers with 10mM phosphate buffer + ground N. benthamiana mix. Holding the top of the leaf with one hand, hold the target leaf between your finger and thumb and gently pull the leaf/buffer mix over the surface of the leaf from base to tip. You should repeat that movement between 5-10 times.
Note
Preferably rub the second leaf of the plant not the first one.
Note
This step needs to be done carefully to ensure sufficient pressure is applied to introduce the buffer+leaf mixture into the leaf without damaging the leaf too much. It will need to be practiced and will be different between different species and developmental stages.
Note
If rubbing dicots, use a circular movements holding the leaf between thumb and finger to ensure good coverage of the leaf without causing too much damage.
When all plants have been inoculated, cover the plants with a propagator lid and wrap in black binbag and leave well-watered in darkness Overnight .
Note
If inoculating multiple constructs, ensure that there is no contact between plants inoculated with different targets.
Note
Efficiency can be increased by keeping the plants in high humidity (e.g. with the propagator lid on) for up to a week. Ensure the black binbags are removed after only one night.
Move plants back onto a bench and grow under standard conditions: 16 hrs day / 8 hrs night, 26.7 °C day and 21.1 °C night with 200 μmolm-2s-1 light.
Note
The temperature at which the plants are subsequently incubated may need to be experimentally derived. Temperatures given here have worked for wheat / blackgrass with either BSMV or FoMV vectors.
First symptoms will appear after 10-15 days post rubbing depending of the cultivar.
Protocol references
Yuan, C., et al. (2011). "A High Throughput Barley Stripe Mosaic Virus Vector for Virus Induced Gene Silencing in Monocots and Dicots." PLOS ONE 6(10): e26468.
Lee, W.-S., et al. (2015). "Virus induced gene silencing (VIGS) for functional analysis of wheat genes involved in Zymoseptoria tritici susceptibility and resistance." Fungal Genetics and Biology 79: 84-88. http://dx.doi.org/10.1016/j.fgb.2015.04.006
Mellado-Sánchez, M., et al. (2020). "Virus-mediated transient expression techniques enable gene function studies in black-grass." Plant Physiology 183(2): 455-459. Accession Number: 32238443 PMCID: PMC7271810 DOI: DOI:10.1104/pp.20.00205
Mellado-Sánchez, M., et al. (2020). "Virus-mediated transient expression techniques enable genetic modification of Alopecurus myosuroides." bioRxiv: 2020.2001.2028.923466 DOI: 10.1101/2020.01.28.923466
Bouton, C., et al. (2018). "Foxtail mosaic virus: A Viral Vector for Protein Expression in Cereals." Plant Physiology 177(4): 1352-1367. http://dx.doi.org/10.1104/pp.17.01679
Acknowledgements
Rothamsted Research receives strategic funding from the Biotechnology and Biological Sciences Research Council of the United Kingdom (BBSRC). We acknowledge support from the Growing Health Institute Strategic Programme [BB/X010953/1; BBS/E/RH/230003A] and Smart Crop Protection Industrial Strategy Challenge Fund (grant no. BBS/OS/CP/000001)
