Sep 10, 2024
Indirect Co-Culture Assay using Boyden Chambers
- Bianca Cruz Pachane1,
- Heloisa Sobreiro Selistre de Araujo1
- 1Universidade Federal de São Carlos - UFSCar

External link: https://doi.org/10.1002/jex2.70075
Protocol Citation: Bianca Cruz Pachane, Heloisa Sobreiro Selistre de Araujo 2024. Indirect Co-Culture Assay using Boyden Chambers. protocols.io https://dx.doi.org/10.17504/protocols.io.5jyl82om7l2w/v1
Manuscript citation:
Pachane BC, Bottaro PHT, Machado AM, Castro CAd, Guerra G, Gozzer LT, Grigoli MM, Zutião AD, Fuzer AM, Cominetti MR, Altei WF, Selistre‐de‐Araujo HS (2025) Tumoural Hypoxic Extracellular Vesicles Foster a Protective Microenvironment in Triple‐Negative Breast Cancer. Journal of Extracellular Biology 4(9). doi: 10.1002/jex2.70075
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: September 10, 2024
Last Modified: September 10, 2024
Protocol Integer ID: 107204
Keywords: Boyden chamber, Zymography, Co-culture, Invasion assay, tumor microenvironment, tumor microenvironment without direct contact, transwell assay, tumoral invasion, tumoral evs in cell, culture assay, intercellular adhesion, using boyden chamber, cell line, tumoral ev, effect of hypoxic, assay, cell, hypoxic, boyden chamber, porous membrane
Funders Acknowledgements:
São Paulo Research Foundation
Grant ID: 2021/01983-4
Disclaimer
DISCLAIMER – FOR INFORMATIONAL PURPOSES ONLY; USE AT YOUR OWN RISK
The protocol content here is for informational purposes only and does not constitute legal, medical, clinical, or safety advice, or otherwise; content added to protocols.io is not peer reviewed and may not have undergone a formal approval of any kind. Information presented in this protocol should not substitute for independent professional judgment, advice, diagnosis, or treatment. Any action you take or refrain from taking using or relying upon the information presented here is strictly at your own risk. You agree that neither the Company nor any of the authors, contributors, administrators, or anyone else associated with protocols.io, can be held responsible for your use of the information contained in or linked to this protocol or any of our Sites/Apps and Services.
Abstract
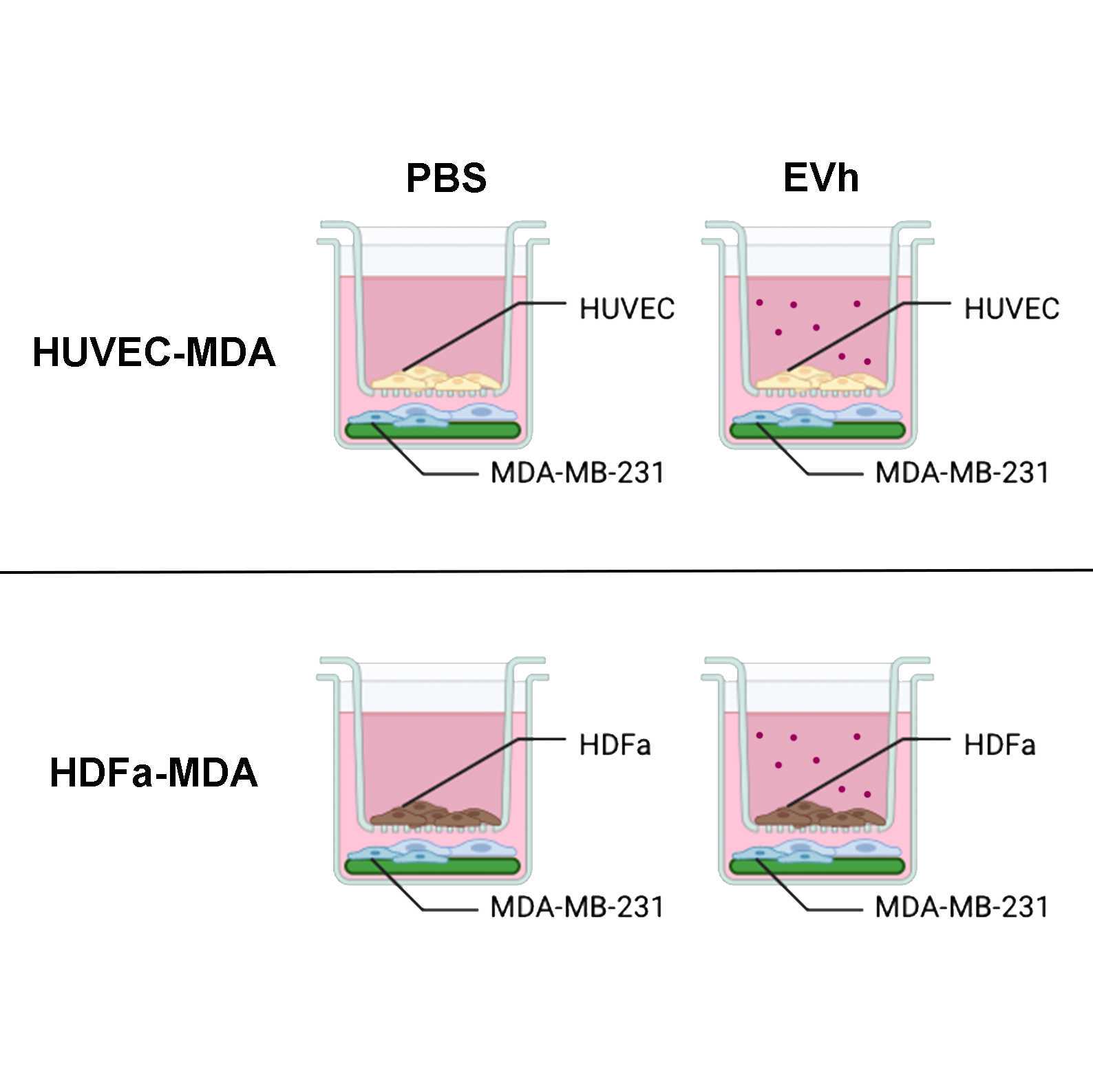
Here, we describe a transwell assay using Boyden chambers as an indirect co-culture method, where two cell lines are grown together but separated by a porous membrane. This assay was used to determine the effect of hypoxic, tumoral EVs in cells from the tumor microenvironment without direct contact between them, focusing on intercellular adhesion and tumoral invasion.
Image Attribution
Experimental diagram created using BioRender.com and Adobe Photoshop.
Materials
Materials and reagents
- Sterile microtubes and pipettes
- 24-well plates, Corning - Catalog #3337
- Round glass coverslips 13mm ø
- Falcon® Permeable Support for 24-well Plate with 3.0 µm Transparent PET Membrane, Corning - Catalog #353096
- Falcon® Permeable Support for 24-well Plate with 8.0 µm Transparent PET Membrane, Corning - Catalog #353097
- Poly-L-Lysine (PLL 0.1 % (w/v) in H2O), Sigma-Aldrich - Catalog #P8920
- Glutaraldehyde, 50% in H2O, Sigma-Aldrich - Catalog #G6403
- Gelatin From Pig Skin, Fluorescein Conjugate, Thermo Fisher - Catalog #G13187
- Sterile PBS
- OptiMEM I Reduced Serum Media, Gibco - Catalog #31985070
- Trypan Blue solution 0.4%, Merck Millipore (Sigma-Aldrich) - Catalog #T8154-100 ml
- Paraformaldehyde solution (PFA 4% in deionized water, pH 7.6 - Sterile)
- Triton X-100 0.1% (v/v) in deionized water
- Phalloidin + DAPI (1 µl Phalloidin-iFluor 647, Abcam - Catalog #ab176759 + 0.76 µL 4,6-Diamidino-2-Phenylindole, Dihydrochloride (DAPI), Thermo Fisher Scientific - Catalog #D1306 in 5 mL PBS)
- Scalpel
- Fluoromount mounting media, Sigma-Aldrich - Catalog #F4680
- Histological slides, Exacta.
- Pierce™ BCA Protein Assay Kit, Thermo Fisher - Catalog #23227
- Precision Plus Protein™ Dual Color, Bio-Rad - Catalog #1610374
Cell lines and growth media
- MDA-MB-231 (ATCC‱ CRM-HTB-26‱) - Leibovitz L-15 10% FBS
- HDFa (ATCC‱ PCS-201-012‱) - DMEM 10% FBS 1% pen/strep
- HUVEC (ATCC‱ CRL-1730‱) - DMEM 10% FBS 1% pen/strep
Equipments:
- Biological cabinet
- Cell incubator (37 ºC, 5% CO2)
- Cell counter - TC20 Cell Counter, Bio-Rad - Catalog #1450011
- Epifluorescence microscope - ImageXpress Micro XLS, Molecular Devices - Catalog #500496
- Orbital shaker
- Mini-PROTEAN Tetra Vertical Electrophoresis System, Bio-Rad - Catalog #1658033FC
- ChemiDOC XRS+ Gel Imaging System, Bio-Rad - Catalog #1708265EDU
Protocol materials
Parafilm™ M Laboratory Wrapping Film, 4 in. W x 125 ft. L; (10cm x 38m)Thermo FisherCatalog #1337410
Troubleshooting
Safety warnings
Light-sensitive assay. Work under sterile conditions.
Before start
Fluorescent gelatin preparation: Under sterile conditions, solubilize the fluorescent gelatin stock at 37 °C with warmed PBS following the manufacturer's instructions for a concentration of 5 mg/mL . Aliquot in microtubes and maintain at -20 ºC until time of use.
Before use, thaw gelatin at 37 °C for 00:30:00 . Dilute stock to a 0.2 mg/mL working solution with warmed PBS and maintain at 37 °C until use.
Cell culture: Maintain cells in culture during at least two passages after thawing.
Preparation of Gelatin-Coated Coverslips
20m
In preparation:
- Clean round glass coverslips (13 mm ø) with 70% ethanol wipes before use. Maintain slips in a clean container.
- Prepare a 0.5 % (v/v) glutaraldehyde solution in H2O and keep at 4 °C until use, protected from light.
Under sterile conditions, apply 20 µL of PLL solution (0.1 mg/ml) in spaced droplets to a surface covered in Parafilm™ M Laboratory Wrapping Film, 4 in. W x 125 ft. L; (10cm x 38m)Thermo FisherCatalog #1337410
Drop coverslips atop the droplets and incubate at Room temperature for 00:20:00 minimum .
20m
Using forceps, transfer the coverslip to a 24-well plate with the coating facing upwards.
Wash coverslips twice with 500 µL PBS.
Cross-link coating with 500 µL of cold 0.5 % (v/v) glutaraldehyde for00:15:00 at Room temperature
15m
Prepare a Petri dish with the bottom covered in Parafilm™ M Laboratory Wrapping Film, 4 in. W x 125 ft. L; (10cm x 38m)Thermo FisherCatalog #1337410
Apply spaced 20 µL droplets of the fluorescent gelatin working solution at 0.2 mg/mL to the Parafilm-covered surface.
Remove the coverslips from the 24-well plate and drop them atop the droplets, with the coating facing down. Incubate at 4 °C Overnight , protected from light.
20m
The next day, remove the slips from the Petri dish using a forceps and transfer them, with the coating facing up, to a fresh 24-well plate.
Wash coverslips thrice with 500 µL PBS.
Slips can be stored at 4 ºC for up to a week, wrapped in aluminium foil.
Pre-condition gelatin coating with 500 µL of Leibovitz L-15 10% FBS for 00:30:00 at 37 °C without CO2.
30m
MDA-MB-231 Seeding in Gelatin-Covered Coverslips
Subculture cells as usual. Resuspend cell pellets in Leibovitz L-15 10% FBS and count cells using the trypan blue exclusion method.
Remove the pre-conditioning medium from the 24-well plate.
Seed 5x104 cells in 1000 µL Leibovitz L-15 10% FBS in each well. Seal the plate with Parafilm™ M Laboratory Wrapping Film, 4 in. W x 125 ft. L; (10cm x 38m)Thermo FisherCatalog #1337410
Incubate cells at 37 °C without CO2 Overnight for adhesion.
HUVEC Seeding in Transwell Insert
Subculture cells as usual. Resuspend cell pellets in DMEM 10% FBS 1% pen/strep and count cells using the trypan blue exclusion method.
Add 750 µL DMEM 10% FBS 1% pen-strep to the wells of a 24-well plate (Corning).
Assemble a Boyden chamber using a 24-well plate and 3.0 µm inserts (Falcon).
Seed 5x104 cells in 250 µL DMEM 10% FBS 1% pen/strep to the upper chamber.
Incubate cells at 37 °C 5% CO2 Overnight for adhesion.
HDFa Seeding in Transwell Insert
Subculture cells as usual. Resuspend cell pellets in DMEM 10% FBS 1% pen/strep and count cells using the trypan blue exclusion method.
Add 750 µL DMEM 10% FBS 1% pen-strep to the wells of a 24-well plate (Corning).
Assemble a Boyden chamber using a 24-well plate and 8.0 µm inserts (Falcon).
Seed 2x103 cells in 250 µL DMEM 10% FBS 1% pen/strep to the upper chamber.
Incubate cells at 37 °C 5% CO2 Overnight for adhesion.
Indirect Co-Culture Assembly
1d
Remove the growth media from all wells and Boyden chambers (both the upper and the lower chambers).
Transfer the MDA-MB-231 coverslips to a fresh well.
Add 750 µL OptiMEM to the coverslip-containing wells.
Transfer either the HUVEC or the HDFa inserts to the wells.
Add 250 µL OptiMEM containing EVh (109 particles/ml) or the equivalent treatment volume in PBS to the upper chamber.
Incubate cells at 37 °C 5% CO2 for 24:00:00
1d
Fixation and Cell Staining
10m
Collect both the upper and the lower chamber's supernatant into a 1.5 ml microtube. Keep On ice . This will be processed later for gelatin zymography -
Transfer the inserts to new wells.
Fix cells with 1000 µL 4% PFA for 00:10:00 at Room temperature
10m
Wash wells twice with 500 µL PBS.
Clean the outside part of the insert membrane with a swab to remove any debris.
Permeabilize cells with 100 µL 0.1% Triton X-100 at Room temperature for 00:05:00
5m
Wash wells twice with 500 µL PBS.
Stain cells with the DAPI + Phalloidin-647 mixture. Add 20 µL of staining solution in spaced droplets to a Parafilm-covered surface. Drop coverslips (coating facing down) and insert membranes atop the droplets and incubate at Room temperature , protected from light for 00:20:00
20m
Wash wells twice with 500 µL PBS.
Assemble coverslips to clean histological slides using Fluoromount as mounting media. Allow slide to dry for at least 04:00:00 , protected from light.
4h
Detach membranes from inserts using a scalpel. Assemble the membranes in histological slides using Fluoromount as mounting media. Seal the membrane with a rectangular coverslip and allow slide to dry for at least 04:00:00 , protected from light.
4h
Once dry, seal coverslips using clear nail polish and store at 4 °C until time of analysis.
Cell Imaging by Epifluorescence HTS
Using the microscope ImageXpress Micro XLS+ (Molecular Devices), check the template for the histological slides and the filters for DAPI (nuclei), FITC (gelatin - for MDA-MB-231 only) and Cy5 (phalloidin-647).
Set laser intensity to a minimum of 10 ms and increase gradatively if necessary.
Check the wells using the 4X objective.
Change into the 20x objective and adjust the laser focus. Select 9 sites per well minimally.
Acquire the plate. Export metadata for analysis.
For representative images, change to the 40x objective and adjust the laser focus.
Select the sites of interest and acquire.
Export image channels and combinations.
Gelatin Degradation Quantification on FIJI
On FIJI (ImageJ), import HTD files through BioFormats.
Images should already be scaled. If not, adjust scale based on the objective lens used for acquisition.
Set measurements to contain "Area", "Standard Deviation", "Shape Descriptor", "Mean grey value", "Perimeter" and "Display label".
Concatenate all stacks into a single hyperstack.
Split channels and select the FITC stack for analysis.
Set a threshold to encompass the degradation spots but not the background. Write down the threshold values. Create a new stack with the binary images.
To measure the degraded area, analyze particles with a range of "5-Infinity" and select "Summarize".
Save CSV file. The degraded area (in µm2) per site will be compared between groups in the statistical analysis.
Cell Morphology Analysis on FIJI
and follow until step #54
Split channels and select the Cy5 stack for analysis.
Duplicate Cy5 stack.
Set a threshold to encompass cell cytoplasm. Create a new stack with the binary images.
Using the duplicated stack as a guide, section cells using the "pencil" tool with a 3 px thickness.
To analyze cell morphology, analyze particles with a range of "10-Infinity" and check "Clear Results".
Save CSV file. The cell circularity index of each cell will be compared between groups in the statistical analysis.
Gelatin Zymography of Assay Supernatant
10m
Prepare 10% SDS-PAGE gels containing gelatin (100 mg/ml) using the Mini-PROTEAN system (Bio-Rad) with 0.75 mm spacers. Maintain gels hydrated at 4 °C Overnight after polymerization.
Spin the assay conditioned media at 1200 rpm, 4°C, 00:10:00 . Transfer the supernatant to new tubes and discard pellet.
10m
Store microtubes at -80 °C until the time of analysis.
Thaw microtubes On ice before the next step and avoid freeze/thawing cycles.
Quantify the supernatants using the Pierce™ BCA Protein Assay kit, following the manufacturer's instructions. A plate reader is required for this step.
Aliquot samples in microtubes to contain 15 µg of protein per well.
Mix samples with non-reducing Laemmli sample buffer (10 % (w/v) SDS ; glycerol; 0.1 Molarity (M) EDTA ; 1 Molarity (M) Tris-HCl pH 6.8 ; bromophenol blue) at a 1:2 ratio.
Assemble gels in running buffer and load samples onto gels alongside a protein ladder (Precision Plus Protein™ Dual Color, Bio-Rad).
Run electrophoresis at 80 V at 4 °C for circa 04:00:00 .
4h
Transfer gels onto cases and photograph them using the ChemiDOC Imaging System (Bio-Rad).
Wash gels with 2.5 % (v/v) Triton X-100 for 00:40:00 under orbital agitation.
40m
Incubate gels in buffer (50 millimolar (mM) Tris, 5 millimolar (mM) CaCl2 pH 8.0, 0.02 % (w/v) sodium azide, 10 millimolar (mM) ZnCl2 ) at 37 °C for 20:00:00
20h
Stain gels with Coomassie Brilliant Blue for 24:00:00 under agitation at Room temperature .
1d
Destain gels with acetic acid:methanol:water (1:1:8, v/v) for up to 48:00:00
2d
Photograph gels using the ChemiDOC Imaging System (Bio-Rad).
Perform a band densitometry analysis in each gel.
Protocol references
BOYDEN, Stephen. THE CHEMOTACTIC EFFECT OF MIXTURES OF ANTIBODY AND ANTIGEN ON POLYMORPHONUCLEAR LEUCOCYTES. The Journal of Experimental Medicine, [s. l.], v. 115, n. 3, p. 453–466, 1962.
LEBER, Thomas M.; BALKWILL, Frances R. Zymography: A Single-Step Staining Method for Quantitation of Proteolytic Activity on Substrate Gels. Analytical Biochemistry, [s. l.], v. 249, n. 1, p. 24–28, 1997.
PACHANE, Bianca Cruz et al. Small Extracellular Vesicles from Hypoxic Triple-Negative Breast Cancer Cells Induce Oxygen-Dependent Cell Invasion. International Journal of Molecular Sciences, [s. l.], v. 23, n. 20, p. 12646, 2022.
EVEN-RAM, Sharona; ARTYM, Vira. Extracellular Matrix Protocols: Second Edition. [S. l.]: Humana Press, 2009.
