Aug 26, 2025
Impact of Mechanical Therapy on Lumbar Disc Health and Nerve Function in L5/S1 Herniation
- Mahmudul Haque Udoy1,
- K M Amran Hossain1
- 1Department of Physiotherapy & Rehabilitation, Jashore University of Science & Technology

Protocol Citation: Mahmudul Haque Udoy, K M Amran Hossain 2025. Impact of Mechanical Therapy on Lumbar Disc Health and Nerve Function in L5/S1 Herniation. protocols.io https://dx.doi.org/10.17504/protocols.io.ewov119o2vr2/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: August 25, 2025
Last Modified: August 26, 2025
Protocol Integer ID: 225345
Keywords: Lumbar Disc Herniation, McKenzie Therapy, Pain Reduction, Disc Morphology, Neurophysiological Recovery, Randomized Controlled Trial, mechanical therapy on lumbar disc health, s1 lumbar disc herniation, lumbar disc health, mckenzie therapy alongside conventional treatment, effectiveness of mckenzie therapy, impact on pain relief, valuable evidence on mckenzie therapy, receiving mckenzie therapy, s1 herniation, mckenzie therapy, functional outcomes for patient, pain relief, mechanical therapy, nerve function in l5, pain intensity, receiving conventional treatment, functional recovery, conventional treatment, effective treatment protocol
Funders Acknowledgements:
Department of Physiotherapy & Rehabilitation
Grant ID: Jashore University of Science & Technology
Abstract
This randomized controlled trial aims to evaluate the effectiveness of McKenzie therapy compared with conventional treatments in improving both structural and functional outcomes for patients with L5/S1 lumbar disc herniation (LDH). A total of 60 patients diagnosed with L5/S1 LDH will be randomized into two groups: one receiving McKenzie therapy alongside conventional treatment, and the other receiving conventional treatment only. Primary outcomes include pain intensity, neurological function, and disc morphology, while secondary outcomes assess functional recovery. The trial will provide valuable evidence on McKenzie therapy’s impact on pain relief and structural improvements, particularly in low-resource settings like Bangladesh. Results are expected to support the development of more cost-effective treatment protocols for managing LDH in such settings.
Guidelines
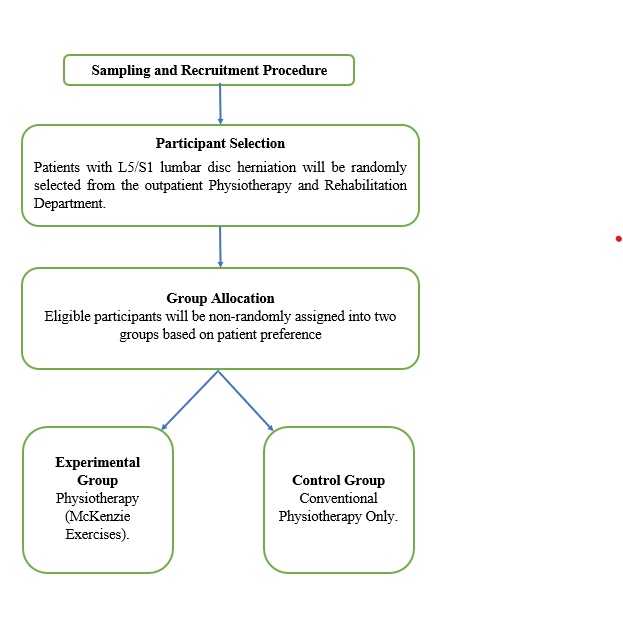
1. Study Design and Oversight
- Study Type: Randomized Controlled Trial (RCT).
- Study Site: The study will be conducted at Jashore University of Science and Technology.
- Randomization: Participants will be randomly assigned to either the McKenzie group (experimental) or the conventional physiotherapy treatment group (control) using a computer-generated sequence and allocation concealment via sealed envelopes.
2. Blinding
- Assessors: Outcome assessors will be blinded to group allocation to prevent bias in assessments.
- Participants and Clinicians: Complete blinding is not feasible due to the nature of the interventions (exercises).
3. Training and Certification
- Therapists: Only certified McKenzie therapists will administer the McKenzie exercise program.
- Conventional Physiotherapy Therapists: Certified therapists will deliver the conventional physiotherapy treatment, including manual therapy and stretching.
- Assessors: Individuals conducting MSK-USG (Musculoskeletal Ultrasound), NCS (Nerve Conduction Studies), and EMG (Electromyography) must be properly trained and certified.
4. Participant Eligibility Criteria
Inclusion Criteria:
- MRI-confirmed L5/S1 Lumbar Disc Herniation (LDH).
- Chronic Symptoms lasting more than 6 weeks with VAS ≥3.
Exclusion Criteria:
- History of lumbar surgery.
- Any endocrine disorders, osteopenia, infections, or carcinomas.
- Fractures in the spine, ribs, or upper limb within the past year.
- Phobia towards physiotherapy or specific treatments.
5. Intervention Delivery
McKenzie Group (Experimental Group):
- McKenzie Exercise Program: A 4-week program consisting of 12 clinic sessions (5–7 minutes per session, 10 repetitions per exercise). Participants are also expected to perform home exercises 5-7 times daily.
Control Group (Conventional Physiotherapy Treatment):
- Conventional Physiotherapy Program: Includes manual passive stretching exercises for lumbo-pelvic muscles (5-7 repetitions, 10-15 second hold), Maitland mobilization for lumbar spine segments, and stabilization exercises for the lower back.
- Frequency: 2-3 times per week for 4 weeks, with at-home exercises performed daily.
6. Outcome Assessments
Primary Outcome: Pain levels measured by VAS (Visual Analog Scale) and PPTs (Pressure Pain Thresholds).
Secondary Outcomes:
- Disc Morphology assessed via MSK-USG (musculoskeletal ultrasound).
Neurophysiology: Measured using SLR (Straight Leg Raise), NCS (Nerve Conduction Studies), and EMG (Electromyography).
- Disability: Measured using ODI (Oswestry Disability Index).
- Time Points: Assessments will occur at baseline, 1 month, 3 months, and 6 months.
7. Data Management
- Data Collection: Data will be recorded in structured forms designed for each participant. All data will be anonymized.
- Storage: Data will be stored in a password-protected database to ensure privacy and security.
- Audits: Weekly audits will be conducted to monitor data accuracy and integrity throughout the study.
Materials
1. McKenzie Exercises
- Equipment: Treatment plinths, floor mats, pillows/bolsters.
- Instructional: Printed exercise guides (English/Bengali), home exercise diaries.
- Monitoring: Timer, attendance log sheets.
2. Conservative Treatment:
- Weeks 1–2 (Acute Phase): Focus on pain reduction, inflammation control, posture correction, and gentle exercises.
- Weeks 3–4 (Subacute Phase): Increase exercise intensity, continue therapy, and reduce the frequency of pain modalities as the patient improves.
- Post-4 Weeks (Recovery/Follow-up Phase): Emphasize strengthening and mobility, reduce dependency on pain management modalities, and reassess progress
3. Outcome Assessments
- Pain: Visual Analog Scale (VAS) forms, pressure algometer.
- Disc Morphology: Musculoskeletal ultrasound, ultrasound gel, and cleaning wipes.
- Neurophysiology: NCS/EMG system, surface electrodes, conductive gel, goniometer (for SLR test).
- Disability: Oswestry Disability Index (ODI) questionnaires (English/Bengali).
4. Data Management
- Forms: Data collection forms (VAS, PPTs, MSK-USG, NCS, EMG, SLR, ODI), adverse event forms.
- Storage: Computer with database software, external hard drive/secure cloud, secure filing cabinet.
5. Safety/Emergency
- Sharps container, first aid kit, resuscitation kit, hand sanitizers, disinfectants, and emergency contact list.
6. Participant Materials
- Consent forms and study information sheets (English/Bengali).
Troubleshooting
Safety warnings
- Monitor for Adverse Events: Participants should be monitored for any adverse effects or exacerbation of symptoms during both McKenzie exercises and conventional physiotherapy sessions.
- Exercise Caution with McKenzie Exercises: McKenzie exercises should be tailored to the individual’s condition to avoid overexertion or injury. Participants should stop exercises if they experience pain beyond their normal tolerance.
- Manual Handling: Ensure proper technique during manual mobilizations to prevent any injuries.
Ethics statement
- This study will be conducted in accordance with the ethical principles outlined in the Declaration of Helsinki.
- Informed Consent: All participants will provide written informed consent before enrollment, understanding the procedures and any potential risks involved.
- Ethical Approval: The study protocol will be reviewed and approved by an ethical review board (IRB) to ensure participant safety and adherence to research standards
STEP 1
- A Randomized Controlled Trial (RCT) will be conducted to evaluate the effectiveness of McKenzie exercises in treating L5/S1 lumbar disc herniation (LDH).
- Participants will be randomly assigned to one of two groups: a) Experimental Group (McKenzie): Receives a 4-week McKenzie exercise program consisting of 12 clinic sessions and daily home exercises. b) Control Group (Conventional Physiotherapy): Receives manual stretching, Maitland mobilization, and stabilization exercises for 4 weeks.
STEP 2
- Data Collection and Outcome Measures
a) Primary Outcome: Pain levels will be measured using VAS (Visual Analog Scale) and PPTs (Pressure Pain Thresholds), Disc morphology will be assessed via MSK-USG (musculoskeletal ultrasound), neurophysiology (SLR, NCS, EMG)
b) Secondary Outcomes: Disability using the ODI (Oswestry Disability Index).
c) Assessments will be made at baseline, 1 month, 3 months, and 6 months.
STEP 3
- Data Management and Ethics a) Data will be anonymized, stored in a password-protected database, and undergo weekly audits for accuracy.
b) Informed consent will be obtained from all participants, and ethical approval will be sought from the IRB. Safety measures will be implemented throughout the study, with close monitoring of any adverse events
Protocol references
1. Robin McKenzie, S. M. (2003). The Lumbar Spine Mechanical Diagnosis & Therapy (2nd ed., Vol. 2). Spinal Publications New Zealand Ltd.
2. Hossain, M. A., Jahid, I. K., Hossain, M. F., Uddin, Z., Kabir, M. F., Hossain, K. M. A., Hassan, M. N., & Walton, L. M. (2020). Efficacy of McKenzie Manipulative therapy on pain, functional activity and disability for lumbar disc herniation. medRxiv (Cold Spring Harbor Laboratory). https://doi.org/10.1101/2020.07.13.20152843
3. Ahsan, M. K., Hossain, M. R., Khan, M. S. I., Zaman, N., Ahmed, N., Montemurro, N., & Chaurasia, B. (2020). Lumbar revision microdiscectomy in patients with recurrent lumbar disc herniation: A single-center prospective series. Surgical Neurology International, 11, 404. https://doi.org/10.25259/sni_540_2020
4. Li, W., Liu, Y., Zheng, C., Miao, J., Chen, H., Quan, H., ... & Zhang, K. (2018). Diagnosis of compressed nerve root in lumbar disc herniation patients by surface electromyography. Orthopaedic Surgery, 10(1), 47–55. https://doi.org/10.1111/os.12362
5. McNally, D. S., Naish, C., & Halliwell, M. (2000). Intervertebral disc structure: Observation by a novel use of ultrasound imaging. Ultrasound in Medicine & Biology, 26(5), 751–758. https://doi.org/10.1016/S03015629(00)00209-X
6. Kilpikoski, S., Häkkinen, A. H., Repo, J. P., Kyrölä, K., Multanen, J., Kankaanpää, M., ... & Ylinen, J. (2024). The McKenzie Method versus guideline-based advice in the treatment of sciatica: 24-month outcomes of a randomised clinical trial. Clinical Rehabilitation, 38(1), 72–84. https://doi.org/10.1177/02692155231196393
7. Dunsford, A., Kumar, S., & Clarke, S. (2011). Integrating evidence into practice: Use of McKenzie-based treatment for mechanical low back pain. Journal of Multidisciplinary Healthcare, 4, 393–402. https://doi.org/10.2147/JMDH.S24733
8. Hirayama, J., Yamagata, M., Ogata, S., Shimizu, K., Ikeda, Y., & Takahashi, K. (2006). Relationship between low-back pain, muscle spasm and pressure pain thresholds in patients with lumbar disc herniation. European Spine Journal, 15(1), 41–47. https://doi.org/10.1007/s00586-004-0813-2
Acknowledgements
We express gratitude to the Department of Physiotherapy & Rehabilitation (PTR) at Jashore University of Science and Technology for partial funding and provision of facilities for this study. We also acknowledge the anticipated contributions of Certified McKenzie Therapists, Certified Physiotherapists, and the research team for their expertise in executing this future protocol.

