Dec 15, 2021
Version 1
Immunolabelling and clearing of intact spinal cord for visualization of lower urinary tract afferents V.1
- Janet Keast1,
- Peregrine Osborne1,
- John-Paul Fuller-Jackson1
- 1University of Melbourne
- SPARCTech. support email: [email protected]

Protocol Citation: Janet Keast, Peregrine Osborne, John-Paul Fuller-Jackson 2021. Immunolabelling and clearing of intact spinal cord for visualization of lower urinary tract afferents. protocols.io https://dx.doi.org/10.17504/protocols.io.byqdpvs6
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it’s working
Created: October 01, 2021
Last Modified: November 27, 2023
Protocol Integer ID: 53733
Keywords: neural tracing, retrograde tracing, clearing, idisco, light sheet microscopy, visualization of lower urinary tract afferent, labelled lower urinary tract afferent, lower urinary tract afferent, imaging of spinal cord, lower urinary tract, lumbosacral spinal cord, motoneurons within the spinal cord, clearing of intact spinal cord, spinal cord, intact spinal cord, rostrocaudal location of afferent, preganglionic autonomic neuron, mount immunolabeling, autonomic neuron, motoneuron, choline acetyltransferase
Funders Acknowledgements:
NIH-SPARC
Grant ID: OT2OD023872
Abstract
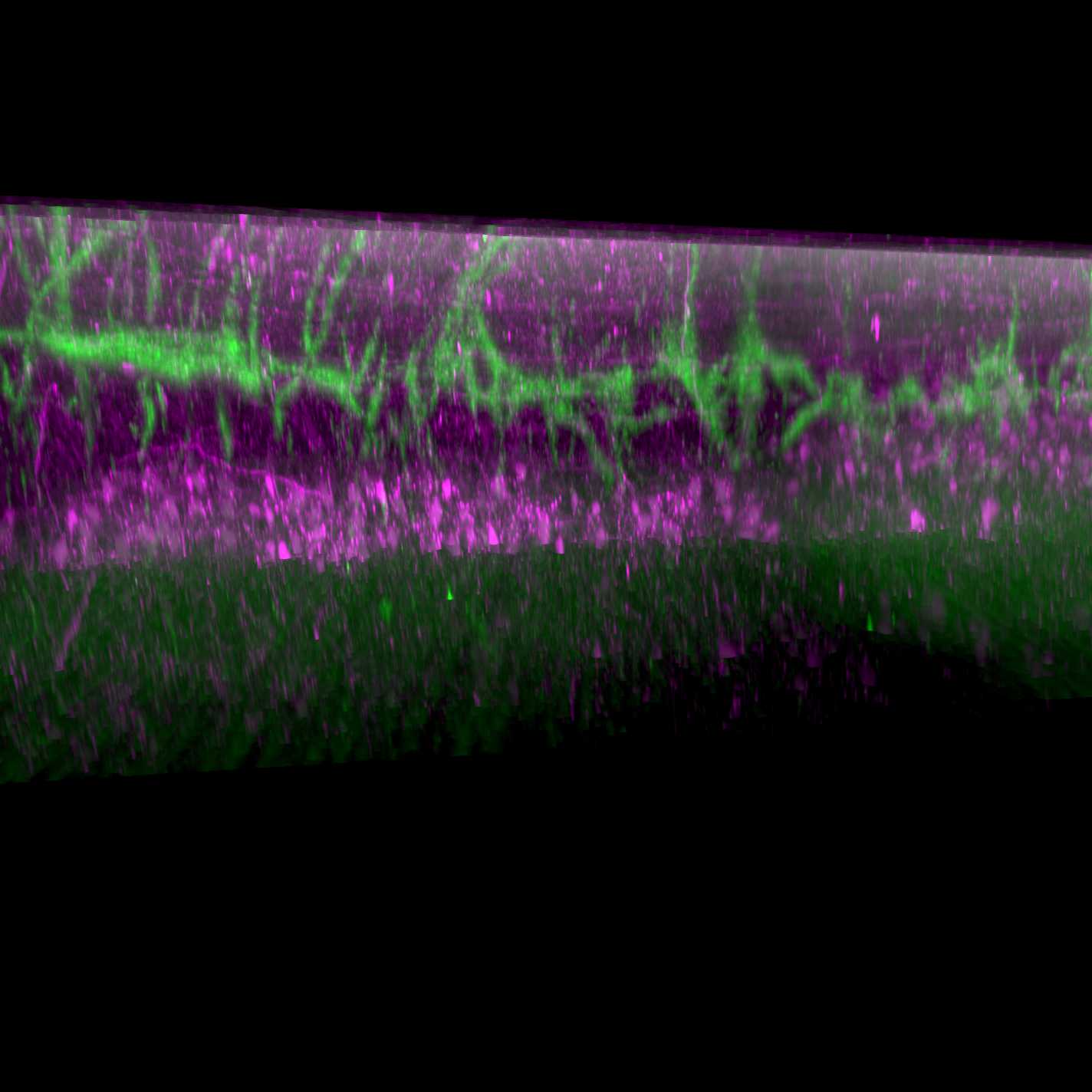
The whole-mount immunolabeling and clearing method (iDISCO) was used to visualize cholera toxin subunit B-labelled lower urinary tract afferents in the lumbosacral spinal cord of the rat. Imaging of spinal cord was performed on a light sheet microscope with a 12x lens. Concurrently, choline acetyltransferase identified preganglionic autonomic neurons and motoneurons within the spinal cord, which were used to confirm the rostrocaudal location of afferents.
Materials
Materials
MethanolMerck MilliporeSigma (Sigma-Aldrich)Catalog #M3641
DichloromethaneMerck MilliporeSigma (Sigma-Aldrich)Catalog #320269
Dibenzyl etherMerck MilliporeSigma (Sigma-Aldrich)Catalog #108014
Ethyl cinnamateMerck MilliporeSigma (Sigma-Aldrich)Catalog #112372
1X Dulbecco’s Phosphate Buffered Saline (DPBS) Thermo Fisher ScientificCatalog #14190094
Gelatin from porcine skinMerck MilliporeSigma (Sigma-Aldrich)Catalog #G1890
Hydrogen peroxide 30%Merck Millipore (EMD Millipore)Catalog #822287.1000
SaponinMerck MilliporeSigma (Sigma-Aldrich)Catalog #S4521
ThimerosalMerck MilliporeSigma (Sigma-Aldrich)Catalog #T5125
Triton X-100Merck MilliporeSigma (Sigma-Aldrich)Catalog #T8787-50ML
Rabbit anti-cholera toxin antibodyMerck MilliporeSigma (Sigma-Aldrich)Catalog #C3062
Goat anti-choline acetyltransferase antibodyMerck Millipore (EMD Millipore)Catalog #AB144P
Cy3 Donkey anti-rabbit IgGJackson ImmunoResearch Laboratories, Inc.Catalog #711-165-152
AF647 Donkey anti-sheep IgGThermo Fisher ScientificCatalog #A21448
Equipment
Equipment
Ultramicroscope II
NAME
Light sheet microscope
TYPE
Miltenyi Biotec
BRAND
NA
SKU
LINK
Solutions
PBS: phosphate-buffered saline, 0.1 M, pH 7.2
DPBS: 1x Dulbecco's phosphate-buffered saline
DPBS-T: 1x Dulbecco's phosphate-buffered saline containing 0.5% Triton X100
DPBSG-T: 1x Dulbecco's phosphate-buffered saline containing 0.2% gelatin, 0.5% Triton X-100 and 0.01% thimerosal
Primary antibodies
| A | B | C | D | E | |
| Abbreviation | Synonym | RRID | Host species | Dilution | |
| ChAT | Choline acetyltransferase | AB_11214092 | Goat | 1:500 | |
| CTB | Cholera toxin subunit B | AB_258833 | Rabbit | 1:3000 |
Secondary antibodies
| A | B | C | |
| Tag-antibody | Host species | Dilution | |
| AF647 anti-sheep | Donkey | 1:500 | |
| Cy3 anti-rabbit | Donkey | 1:2000 |
Troubleshooting
Spinal cord preparation
30m
While immersed in phosphate buffered-saline (PBS), pH 7.2, trim nerve roots of fixed spinal cord to within approximately 2 mm of the spinal cord surface to facilitate the identification of segments later following imaging.
Bleaching
1d
Wash samples in 1x Dulbecco’s PBS (DPBS)(6 x 15 mins).
Dehydrate samples in a series of methanol in DPBS dilutions while on rotation at 12 rpm:
- 50% methanol in DPBS (1.5 h)
- 80% methanol in DPBS (1.5 h)
- 100% methanol (1.5 h)
Bleach samples overnight in 6% hydrogen peroxide in methanol at 4°C, protected from light.
Blocking
2d
Rehydrate samples in a series of methanol in DPBS dilutions while on rotation at 12 rpm:
- 100% methanol (2 x 1.5 h)
- 80% methanol in DPBS (1.5 h)
- 50% methanol in DPBS (1.5 h)
- DPBS (1.5 h)
Incubate samples in DPBS containing 0.2% gelatin, 0.5% Triton X-100 and 0.01% thimerosal (DPBSG-T) for 36 h while on rotation at 12 rpm
Primary antibody incubation
1w 3d
Incubate samples in primary antibody solution containing DPBSG-T with 0.1% saponin for 10 days at 37°C with agitation. Volume of solution need only be sufficient to cover the sample.
Secondary antibody incubation
5d
Wash spinal cords in 1x DPBS with 0.5% Triton X-100 (DPBST) (6 x 15 mins).
Incubate samples in secondary antibody solution containing DPBSG-T with 0.1% Saponin for 4 days at 37°C with agitation. Volume of solution need only be sufficient to cover the sample.
Dehydration and delipidation
1d 8h
Wash spinal cords in DPBST (6 x 15 mins).
Dehydrate spinal cords in a series of methanol in DPBS dilutions while on rotation at 12 rpm:
- 20% methanol in DPBS (1 h)
- 40% methanol in DPBS (1 h)
- 60% methanol in DPBS (1 h)
- 80% methanol in DPBS (1h)
- 100% methanol (2 x 1 h)
Incubate samples in a solution of 2/3 dichloromethane and 1/3 methanol overnight on rotation. Ensure that samples sink to the bottom of the vial at the end of this step, otherwise continue incubation in freshly made solution.
Incubate samples in 100% dichloromethane for 30 mins while on rotation at 12 rpm. Repeat this step until samples sink.
Clearing
2h
Incubate samples in dibenzyl ether until the samples have become clear. Ensure each vial is completely filled with dibenzyl ether to minimize sample oxidation as a result of large amounts of air in the vial. This process should not take longer than 2 h.
Storage
Store cleared samples in fresh dibenzyl ether. Keep away from light (wrap in foil and store in an opaque container).
Light sheet microscopy
Prior to visualization on a light sheet microscope, samples should be transferred into ethyl cinnamate, at least 3 h prior.
Remove sample from ethyl cinnamate and gently dry on a tissue. Affix sample to plastic mount using the minimum amount of super glue required. Tips:
- Avoid adhering the sample to the base via any region of the sample that is of interest; both the super glue and proximity to the plastic will reduce imaging quality in that area.
- Mount the sample perfectly in the middle; the light sheet microscope stage has limited mobility, particularly at higher magnifications.
- Orientate the sample such that the thinnest plane of the sample is perpendicular to the light sheet beams. The further light has to travel through a sample, the poorer the image quality.
- Orientate the sample so that the region of interest is facing as close to the lens as possible.
In this protocol, the spinal cord was mounted perpendicular to the light sheet beams, with the dorsal horn facing the lens, and the ventral side of L5 being the point of adherence to the mounting platform.
In order to visualize the lower urinary tract afferents in the dorsal horn of the spinal cord, use the highest magnification lens available on the light sheet microscope.
For example, a 12x lens is adequate for afferent visualization.
Look for cholera toxin subunit B labelled projections in the lateral and medial boundaries of the dorsal horn. One should also find dense afferent innervation of the sacral preganglionic nucleus of L6 and S1 spinal cord .
