Aug 26, 2025
Immunohistochemical detection of porcine reproductive and respiratory syndrome virus (PRRSV) in formalin-fixed, paraffin-embedded (FFPE) pig tissues
Forked from a private protocol
- Jayne Wiarda1
- 1National Animal Disease Center, ARS, USDA
- Jayne Wiarda

Protocol Citation: Jayne Wiarda 2025. Immunohistochemical detection of porcine reproductive and respiratory syndrome virus (PRRSV) in formalin-fixed, paraffin-embedded (FFPE) pig tissues. protocols.io https://dx.doi.org/10.17504/protocols.io.j8nlky831g5r/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: August 26, 2025
Last Modified: August 26, 2025
Protocol Integer ID: 225473
Keywords: NP103, immunohistochemical detection of porcine, pig tissues an immunohistochemistry, respiratory syndrome virus, pig tissue, prrsv, porcine, immunohistochemistry, immunohistochemical detection, tissue
Funders Acknowledgements:
USDA-ARS
Grant ID: CRIS #5030-32000-230-000-D
Disclaimer
All opinions expressed in this paper are the authors’ and do not necessarily reflect the policies and views of USDA or ARS. Mention of trade names or products is for information purposes only and does not imply endorsement by the USDA. USDA is an equal opportunity employer and provider.
Abstract
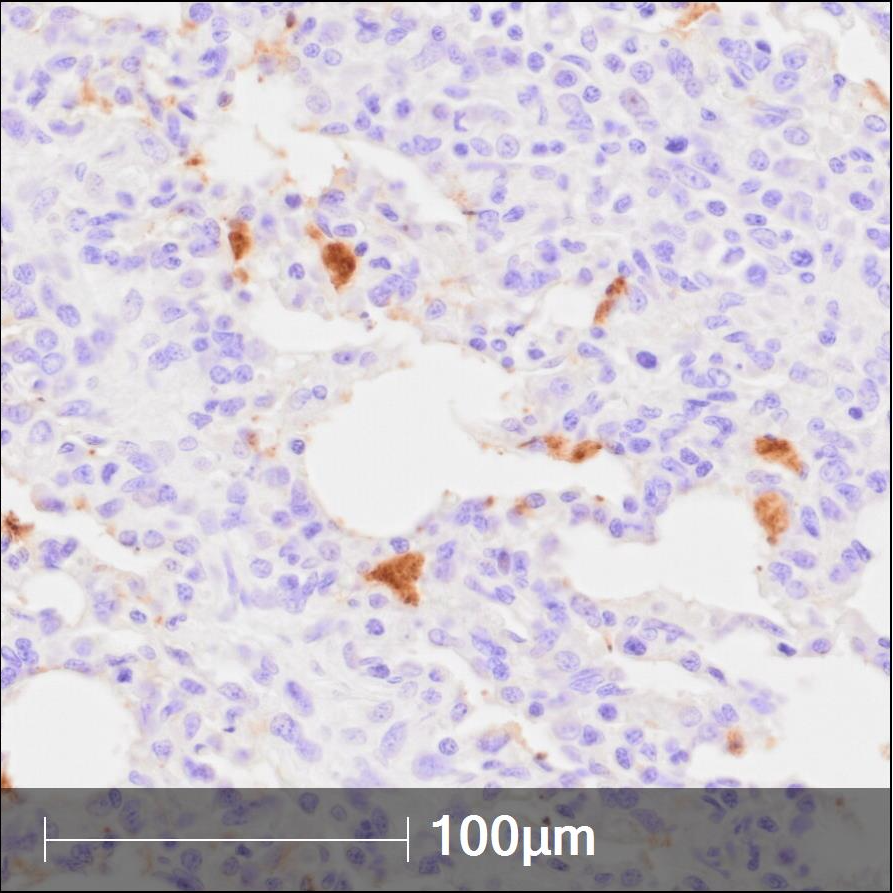
An immunohistochemistry (IHC) staining protocol for in situ identification of porcine reproductive and respiratory syndrome virus (PRRSV) in formalin-fixed, paraffin-embedded (FFPE) tissue.
Attachments
Guidelines
Assay Controls
• Controls will be required to ensure there is minimal/no cross-reactivity with off-target epitopes in the tissue and to ensure stains are specific to the intended antibody targets
• IHC controls:
o Secondary only control
• This slide receives only diluent in place of primary antibody. All other reagents are still applied to the slide.
Assay Variations
· Parameters may need to be further optimized for different experiments, tissues, targets, or species.
Materials
Equipment
• Pipettes/pipette tips
• Drying oven (able to reach & hold 60℃)
• Fume hood
• Slide staining tray (e.g. Simport M920-2)
• EZ-Batch Slide Holder (Advanced Cell Diagnostics 321716)
• EZ-Batch Wash Tray (Advanced Cell Diagnostics 321717)
• Tissue-Tek Vertical 24 slide rack (American Master Tech Scientific LWS2124)
• Tissue-Tek Staining Dishes (American Master Tech Scientific LWS20WH)
• Tissue-Tek Clearing Agent Dishes, xylene resistant (American Master Tech Scientific LWS20GR)
• Brightfield microscope and/or slide imaging platform
Reagents/Supplies
• Distilled water (obtained in-house)
• Phosphate-buffered saline (PBS), pH 7.2-7.4 (made in-house)
• 0.05% PBS-Tween (PBS-T), pH 7.35 (made in-house)
• Xylenes (Macron Fine Chemicals 8668-16)
• 100% ethanol (Pharmco 111000200)
o Dilute with distilled water to make 95%, 85%, and 70% concentrations
• Pro-Par Clearant (Anatech 510)
• Fixative
o 10% NBF (Cancer Diagnostics, Inc. 111) or 4% PFA (Electron Microscopy Sciences 15713)
• ImmEdge Hydrophobic Barrier Pen (Vector H-4000)
• Ready to use Proteinase-K (Dako S302080-2/S302030-2)
• BLOXALL Endogenous Blocking Solution (Vector SP-6000)
• ImmPRESS-VR Horse Anti-Mouse IgG Polymer Detection Kit, Peroxidase (Vector MP-6402-15)
o Normal Horse Serum, 2.5%
o ImmPRESS-VR Horse Anti-Mouse IgG Polymer Reagent
• Mouse anti-PRRSV monoclonal antibody clone SR30; stock concentration 0.2 g/L (RTI Lab SR30-A)
• 1% bovine serum albumin (BSA) in PBS (made in-house)
• ImmPACT DAB EqV Substrate Kit, Peroxidase (Vector SK-4103)
o ImmPACT DAB EqV Reagent 1 (Chromogen)
o ImmPACT DAB EqV Reagent 2 (Diluent)
• Gill’s Hematoxylin I (American Master Tech Scientific HXGHE1LT)
• Refrax Mounting Medium (Anatech 711)
• #1 thickness cover glass (e.g. Fisherbrand 12-545-F)
Troubleshooting
Safety warnings
***For all reagents, refer to MSDS to determine appropriate precautions, personal protective equipment (PPE), and disposal methods before use***
Before start
Starting specimens
Starting samples = FFPE tissues cut to 4 micron thickness and adhered to positively-charged microscopy slides (e.g. SuperFrost Plus Slides; Fisher Scientific 12-550-15). It is crucial that tissues are adequately fixed to prevent tissue degradation. Tissues no thicker than 0.5 centimeters should be freshly harvested and placed into 10% neutral-buffered formalin (NBF) or 4% paraformaldehyde (PFA) at a ratio of at least 20 volumes fixative per one volume tissue. Fix tissues between 16-30 hours at room temperature (RT), followed by immediate transfer to 70% ethanol and processing into FFPE tissue blocks. Fixation times should be optimized for individual tissues and experiments.
Baking
20m
Before starting the assay:
• Preheat a dry oven to 60℃
• Load slides for assay into vertical slide rack
• Bake slides 20 min 60℃ 00:20:00
20m
While slides bake:
o Prepare 0.05% PBS-T
Immediately before deparaffinizing:
o Add ~200 mL xylenes to each of three clearing agent dishes in a fume hood
o Add ~200 mL 100% ethanol to each of two staining dishes in a fume hood
o Add ~200 mL 95% ethanol to a staining dish in a fume hood
o Add ~200 mL 85% ethanol to a staining dish in a fume hood
o Add ~200 mL 70% ethanol to a staining dish in a fume hood
o Add ~200 mL distilled water to a staining dish in a fume hood
o Add ~200 mL PBS-T to a staining dish
Deparaffinizing & Rehydrating
20m
• Submerge slide rack in fresh xylenes 5 min RT 00:20:00
• Submerge slide rack in fresh xylenes 5 min RT
• Submerge slide rack in fresh xylenes 5 min RT
• Submerge slides rack in fresh 100% ethanol 1 min RT
• Submerge slides rack in fresh 100% ethanol 1 min RT
• Submerge slides rack in fresh 95% ethanol 1 min RT
• Submerge slides rack in fresh 85% ethanol 1 min RT
• Submerge slides rack in fresh 70% ethanol 1 min RT
• Submerge slides rack in fresh distilled water 3 min RT
• Submerge slides rack in fresh PBS-T for transport
20m
While slides deparaffinize/rehydrate:
o Turn off dry oven
o Prepare humidified slide staining tray by adding water to bottom & placing lid on top
o Add ~200 mL PBS-T to each of two wash trays
Hydrophobic Barrier
20m
• Apply hydrophobic barrier around each tissue 00:20:00
o One by one, unload slides from vertical rack submerged in PBS-T. Dry off only the area around the tissue where a barrier will be drawn with a hydrophobic barrier pen. Keep tissue area wet the whole time. Draw barrier and place slides into the EZ-Batch slide holder placed inside the slide staining tray. Using a pipette, apply a small amount of PBS-T within the barrier (just enough to keep the tissue wet while drawing barriers on remaining slides). Slides will remain locked in the EZ-Batch slide holder throughout the protocol until being transferred back to a vertical rack for counterstaining.
• Leave slide holder in slide staining tray
20m
Proteinase Digestion
10m
• Decant slide holder and again place flat in slide staining tray 00:10:00
• Incubate with Dako Ready to use Proteinase-K 3 min RT
o Apply drops to completely cover tissues
o Let incubate in slide staining tray with lid closed
• Decant slide holder and transfer to wash trays for PBS-T washes
• Submerge slide holder in fresh PBS-T 2 min RT
• Submerge slide holder in fresh PBS-T 2 min RT
10m
While slides incubate with Proteinase:
o Discard deparaffinizing & rehydrating reagents
o Add ~200 mL PBS-T to each of two wash trays
Tissue Quenching
15m
• Decant slide holder and again place flat in slide staining tray00:15:00
• Incubate with BLOXALL Endogenous Blocking Solution 10 min RT
o Apply to completely cover tissues; let incubate in slide staining tray with lid closed
• Decant slide holder and transfer to wash trays for PBS-T washes
• Submerge slide holder in fresh PBS-T 2 min RT
• Submerge slide holder in fresh PBS-T 2 min RT
15m
While slides incubate with enzyme block:
o Discard epitope retrieval reagents
o Add ~200 mL PBS-T to each of two wash trays
Protein Blocking
30m
• Decant slide holder and again place flat in slide staining tray 00:30:00
• Incubate with Normal Horse Serum 2.5% 30 min RT
o Serum is a component of the secondary antibody reagent kit
o Apply to completely cover tissues; let incubate in slide staining tray with lid closed
30m
While slides incubate with serum:
o Discard tissue quenching reagents
o Prepare diluted primary antibody by adding antibody stock to 1% BSA in PBS at a dilution of 1:10,000. Total volume to use is dependent on tissue sizes. Make sure to mix reagents before pipetting. Mix diluted antibody well before use.
Primary Antibody
5m
• Decant slide holder and again place flat in slide staining tray Overnight
o Do not wash with PBS-T between decanting serum and applying secondary antibody
• Incubate with diluted primary antibody 4C overnight
o Apply to completely cover tissues; let incubate in slide staining tray with lid closed
• • Decant slide holder and transfer to wash trays for PBS-T washes
• Submerge slide holder in fresh PBS-T 2 min RT
• Submerge slide holder in fresh PBS-T 2 min RT
5m
While slides incubate with primary antibody:
o Discard protein blocking reagents
The Next Day:
o Add ~200 mL PBS-T to each of two wash trays
Secondary Antibody
35m
Decant slide holder and again place flat in slide staining tray 00:35:00
• Incubate with Horse anti Mouse IgG (HRP-conjugated polymer) 30 min RT
o Secondary antibody polymer is a component of the secondary antibody reagent kit
o Apply to completely cover tissues; let incubate in slide staining tray with lid closed
• Decant slides and transfer to vertical slide rack for PBS-T washes
• Submerge slide rack in fresh PBS-T 2 min RT
• Submerge slide rack in fresh PBS-T 2 min RT
35m
While slides incubate with secondary antibody:
o Discard primary antibody reagents
o Add ~200 mL PBS-T to each of two wash trays
Immediately before chromogen detection:
o Prepare diluted DAB chromogen by adding equal volumes of DAB Reagent 1 (Chromogen) and DAB Reagent 2 (Diluent) together. Total volume to use is dependent on tissue sizes. Make sure to mix reagents thoroughly. Store in the dark due to light sensitivity.
Chromogenic Detection (DAB)
10m
• Decant slide holder and again place flat in slide staining tray 00:10:00
• Incubate with diluted DAB chromogen 5 min RT
o Apply to completely cover tissues; let incubate in slide staining tray with lid closed
• Decant slide holder and transfer to vertical slide rack for PBS-T washes
• Submerge slide rack in fresh PBS-T 2 min RT
• Submerge slide rack in fresh PBS-T 2 min RT
10m
While slides incubate with chromogen:
o Discard remaining secondary antibody reagents
o Add ~200 mL PBS-T to each of two wash trays
o Add ~200 mL hematoxylin to one staining dish
o Add ~200 mL distilled water to each of three staining dishes
Counterstaining
2m
• Transfer slides to vertical slide rack00:02:00
o Do quickly to avoid drying out slides or alternatively place vertical slide rack in a staining dish containing PBS-T and then transfer slides
• Submerge slide rack in hematoxylin 1 min RT
• Submerge slide rack in fresh distilled water, dunking 3-5 times
• Submerge slide rack in fresh distilled water, dunking 3-5 times
• Submerge slide rack in fresh distilled water, dunking 3-5 times
o Water should no longer appear purple in the third water dish used
2m
Mounting
45m
• Submerge slide rack in fresh 95% ethanol 1 min RT00:45:00
• Submerge slide rack in fresh 100% ethanol 1 min RT
• Submerge slide rack in fresh 100% ethanol 1 min RT
• Submerge slide rack in fresh 100% ethanol 1 min RT
• Submerge slide rack in fresh Pro-Par 5 min RT
• Submerge slide rack in fresh Pro-Par 5 min RT
• Submerge slide rack in fresh Pro-Par 5 min RT
• Mount slides by adding 2-4 drops of mounting media to each slide, followed by application of a cover glass. Remove bubbles from tissue by applying pressure to cover glass.
• Place slides flat in a dry, dark space to air dry at RT overnight
• Assess staining with a brightfield microscope
45m
While slides are air drying:
o Discard chromogen detection, counterstaining, and slide mounting reagents
Protocol references
· Staining protocol was developed by Dr. Jayne Wiarda
· Staining protocol was optimized and executed by Dr. Jayne Wiarda and Colin Stoy
· We thank Adrienne Shircliff for slide sectioning and imaging

