Jan 12, 2026
Immune Adherence Hemagglutination Assay
- Brian Andrich L. Pollo1
- 1University of the Philippines Manila

Protocol Citation: Brian Andrich L. Pollo 2026. Immune Adherence Hemagglutination Assay. protocols.io https://dx.doi.org/10.17504/protocols.io.q26g77ye3gwz/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: January 10, 2026
Last Modified: January 12, 2026
Protocol Integer ID: 238379
Keywords: Immune Adherence Hemagglutination (IAHA), Complement Activation, Immune Complexes, SARS-CoV-2, Pathogenic Coronaviruses, Human Erythrocytes, N-Acetylcysteine (Nac), Serological Assay, Hemagglutination Scoring, Viral Immunology, Complement Receptor 1 (CR1), HBS++, Antipeptide Antibodies, Immune Response Modulation, Diagnostic Protocol, Bioassay Validation, immune adherence hemagglutination assay this protocol, immune adherence hemagglutination assay, immune adherence hemagglutination, immunogenicity of pathogenic coronavirus peptide, monitoring humoral immune response, circulating immune complex, pathogenic coronavirus peptide, humoral immune response, successful complement activation, immunogenicity, capacity of antipeptide antibody, immune complex, antipeptide antibody, addition of normal human plasma, visual scoring of hemagglutination pattern, viral pathogenesi, hemagglutination pattern, normal human plasma, human erythrocyte, red blood cell, peptide, complement, assay
Funders Acknowledgements:
DOST-PCHRD
Grant ID: MD-PhD Dissertation grant
Disclaimer
DISCLAIMER – FOR INFORMATIONAL PURPOSES ONLY; USE AT YOUR OWN RISK
The protocol content here is for informational purposes only and does not constitute legal, medical, clinical, or safety advice, or otherwise; content added to protocols.io is not peer reviewed and may not have undergone a formal approval of any kind. Information presented in this protocol should not substitute for independent professional judgment, advice, diagnosis, or treatment. Any action you take or refrain from taking using or relying upon the information presented here is strictly at your own risk. You agree that neither the Company nor any of the authors, contributors, administrators, or anyone else associated with protocols.io, can be held responsible for your use of the information contained in or linked to this protocol or any of our Sites/Apps and Services.
Abstract
This protocol describes an Immune Adherence Hemagglutination (IAHA) assay used to detect and semi-quantitatively analyze circulating immune complexes and complement-activating sequences. The method is particularly effective for evaluating the immunogenicity of pathogenic coronavirus peptides and the neutralizing capacity of antipeptide antibodies.
The procedure involves a multi-step incubation process: forming or introducing an immune complex (such as heat-aggregated plasma, tetanus toxoid mixtures, or COVID-19 peptide-sera complexes) in HBS++, followed by the addition of normal human plasma (NHP) as a complement source. A critical modification in this protocol is the use of N-Acetylcysteine (Nac) in lieu of traditional dithiothreitol (DTT) to stabilize the reaction before the addition of human erythrocytes. Results are interpreted via visual scoring of hemagglutination patterns on a U-bottom plate, where positive adherence indicates successful complement activation and receptor binding on the red blood cells. This protocol offers a scalable, non-radioactive alternative for monitoring humoral immune responses and viral pathogenesis.
Attachments

IAHA procedure.pdf
897KB
Image Attribution
CC BY Brian Pollo
Guidelines
1. Place 30μL of the immune complex^^note 1 in HBS++^^note 2 on a U-bottom plate
2. Add 30μL of normal human plasma (NHP)^^note 3^^
3. Incubate the plate at 37°C for 30min
4. Add 30μL 35mM N-Acetylcysteine (Nac) in PBS^^note 4^^
5. Add 30μL 1.5% human erythrocyte in PBS^^note 5^^
6. Incubate the plate at room temperature (RT) for 60min
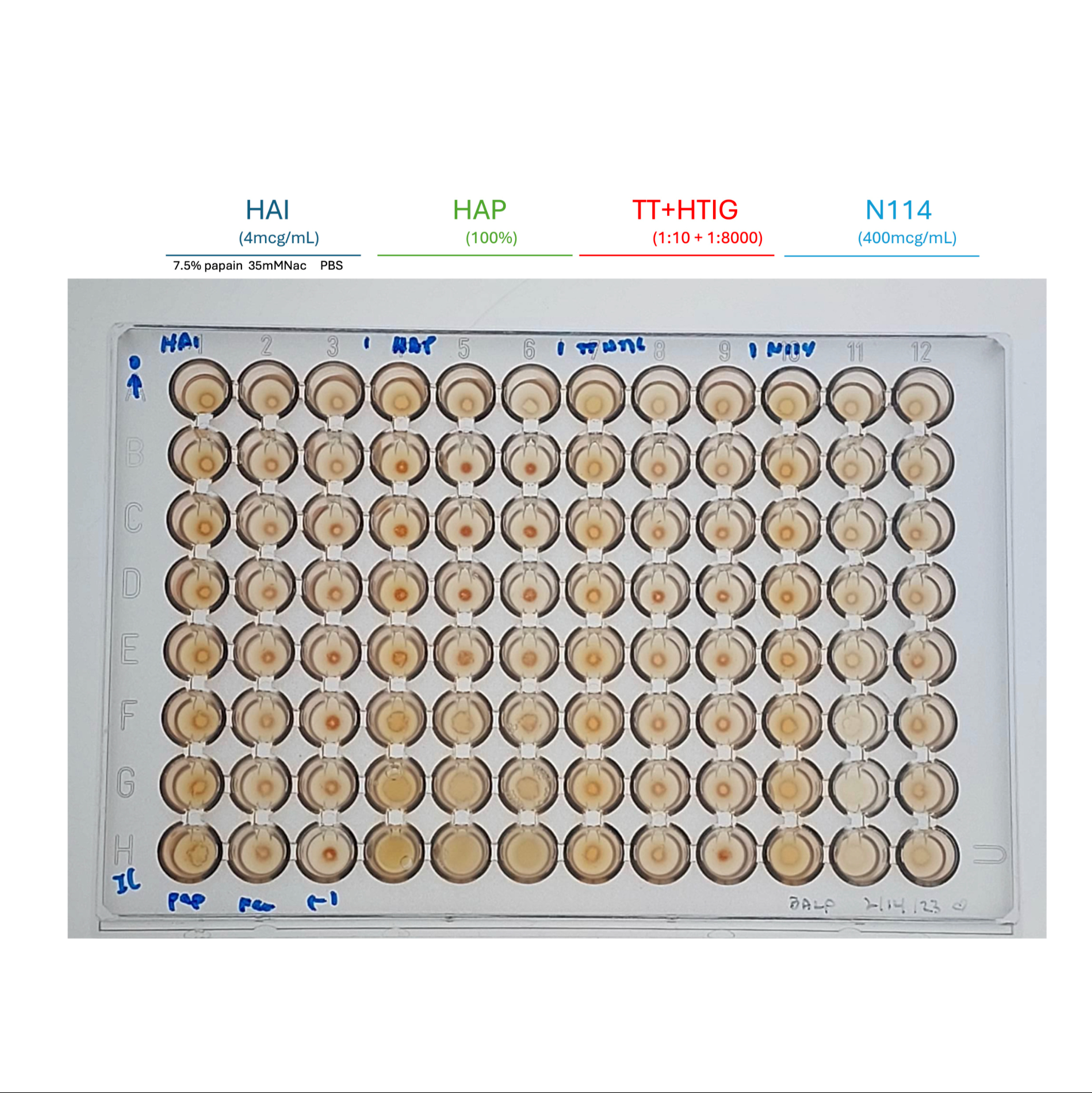
7. Interpret the results visually (scoring from 0 – 4+)
Materials
Materials
- Immune Complexes (Select one based on application):
- Heat-Aggregated Plasma (HAP): Normal Human Plasma (NHP) heated at $63^{\circ}C$ for 30 minutes.
- Heat-Aggregated Immunoglobulin (HAI): $10mg/mL$ solution heated at $63^{\circ}C$ for 30 minutes.
- TT-HTIG Mixture: 1:10 Tetanus Toxoid (TT) mixed with 1:8000 Human Tetanus Immunoglobulin (HTIG), incubated at $37^{\circ}C$ for 30 minutes.
- COVID Peptide + Sera Mixture: Equal volumes of $400mcg/mL$ COVID peptide and COVID patient sera.
- Normal Human Plasma (NHP): Used as the primary complement source.
- HBS++ Buffer: HEPES-buffered saline supplemented with calcium and magnesium, used for initial complex formation.
- N-Acetylcysteine (Nac) Solution: 35mM Nac in Phosphate Buffered Saline (PBS).
- Human Erythrocyte Suspension (1.5%): Prepared from whole blood containing CPDA-1 anticoagulant, diluted in PBS.
- EDTA-PBS (Optional): Concentrations ranging from 4mM to 40mM, used specifically to abrogate complement effects for negative controls.
- Microtiter Plate: U-bottom 96-well plate for visual interpretation of hemagglutination patterns.
Troubleshooting
Before start
**Note 1**
- A variety of immune complexes may be used including:
- Heat-aggregated plasma (HAP)
- NHP heated at 63°C on a heat block for 30min
- Heat-aggregated immunoglobulin (HAI)
- Prepared 10mg/mL, then heated at 63°C on a heat block for 30min
- TT-HTIG mixture
- Equal volumes of 1:10 tetanus toxoid (TT) and 1:8000 human tetanus immunoglobulin (HTIG) are mixed, then incubated at 37°C for 30 min
- COVID peptide + COVID sera mixture
- Equal volumes of 400mcg/mL COVID peptide and COVID sera are mixed, then incubated at 37°C for 30 min
- Inada et al (1981) used the following immune complexes as standards
- Heat aggregated human IgG (HAG) – prepared 10mg/mL, 63°C, 30min (when testing, start @ 500mcg/mL)
- 25uL 1:800 rabbit antihuman IgG serum + 25uL serially diluted human IgG (starting 64mcg/mL), 37°C, 60min
**Note 2**
- HBS++ is HEPES buffered saline (pH 7.4) supplemented with magnesium and calcium. Gelatin was removed to promote buffer longevity
- This method works with gelatin-HEPES buffer supplemented with magnesium and calcium (GHB++) as well
- In the original procedure by Inada et al. (1981), gelatin-veronal buffer supplemented with magnesium and calcium (GVB++) was used
**Note 3**
- This step adds the complement source
- This method works with normal human sera (NHS) as well
- In the original procedure by Inada et al. (1981), guinea pig sera was used
**Note 4**
- The purpose of this step is to stabilize C3b on the erythrocyte surface
- In my experiments, I dilute 171μL Nac to 3mL with PBS
- Notice the shift from HBS++ to PBS. Complement activation ends at this step because of dilution of Mg^^2+ 26 Ca^^2+^^.
- PBS is cheaper than HBS. Theoretically at least, HBS may be used in place of PBS. No problems with the use of PBS have been encountered. Potential problems include the precipitation of calcium phosphate, although this occurs at more basic pH (with an optimum of 10–12)
- I use 100mg/mL N-acetylcysteine solution for injection (IM/IV) ampules
- This method works with N-acetylcysteine 600mg effervescent tablets as well, but note that it contains various excipients that may interfere, such as starches that could potentially activate complement
- In the original procedure by Inada et al. (1981), 20mM dithiothreitol (DTT) was used. I replaced it with Nac using a 0.98:1.6 ratio roughly based on this paper: https://www.ncbi.nlm.nih.gov/articles/PMC1475835
**Note 5**
- For my experiments, I dilute 165μL of human E in 11mL PBS (for use with 2 plates) or 60 μL human E in 4mL.
- I use human E from whole blood containing CPDA-1 as anticoagulant
**Note 4 26 5**
- To abrogate the effects of complement, EDTA-PBS* may be used in lieu of PBS for steps 4 �26 5. It works via chelation of divalent cations.
- In the original procedure by Inada et al. (1981), 10mM EDTA-GVB was used.
*I have used a variety of EDTA concentrations, ranging from 4mM to 40mM
Procedure
Place 30μL of the immune complex in HBS++ on a U-bottom plate
Add 30μL of normal human plasma (NHP)
Incubate the plate at 37°C for 30min
Add 30μL 35mM N-Acetylcysteine (Nac) in PBS
Add 30μL 1.5% human erythrocyte in PBS
Incubate the plate at room temperature (RT) for 60min
Interpret the results visually (scoring from 0 – 4+)
Protocol references
https://sci-hub.ru/https://doi.org/10.1016/0769-2625(81)90026-X
Detection of circulating immune complexes: A new application of immune adherence haemagglutination
Y. Inada, M. Kamiyama, T. Kanemitsu, W.S. Clark
Annales de l'Institut Pasteur / Immunologie
Volume 132, Issue 2, March–April 1981, Pages 181-190
