Oct 22, 2020
Illumina TruSeq Library quantification with qPCR probe method
This protocol is a draft, published without a DOI.
- Kentaro Itokawa1
- 1National Institute of Infectious Diseases, Japan

Protocol Citation: Kentaro Itokawa 2020. Illumina TruSeq Library quantification with qPCR probe method. protocols.io https://protocols.io/view/illumina-truseq-library-quantification-with-qpcr-p-bnpamdie
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: October 21, 2020
Last Modified: October 22, 2020
Protocol Integer ID: 43458
Keywords: truseq type adapter, quantification of illumina library, qpcr probe method, qpcr, illumina library, size distribution of library, probe, library, illumina
Abstract
A homemade solution for quantification of Illumina library.
Because the method uses a quenched-probe (not intercalator), it is expected to be relatively stable for the difference of size distribution of libraries.
Currently, we confirmed this protocol with a probe for TruSeq type adapter.
Materials
MATERIALS
UltraPure™ Salmon Sperm DNA SolutionThermo FisherCatalog #15632011
PhiX Control v3Illumina, Inc.Catalog #FC-110-3001
PrimeTime® Gene Expression Master MixIntegrated DNA Technologies, Inc. (IDT)Catalog #1055770
Troubleshooting
Preparing calibrators and primer/probe
Prepare low-TE buffer (10 mM Tris-HCl, 0.1 mM EDTA, pH 8.0) containing 5 ng/μl salmon sperm DNA.
| 1 M Tris-HCl, pH8.0 | 100 μl | |
| 0.5 M EDTA | 10 μl | |
| UltraPure™DNA Solution 10 mg/mL | 5 μl | |
| milli-Q water | 9889 μl | |
| Total | 10 mL |
Store in -20 °C.
Prepare dilution series of PhiX control with low-TE buffer containing 5 ng/μl salmon sperm DNA in 8-strip PCR tubes.
These are used as calibrators.
| 50 pM | 100 μl | |
| 5 pM | 100 μl | |
| 0.5 pM | 100 μl | |
| 0.05 pM | 100 μl |
Store in -20 °C.
Prepare 20x primers & probe mix solution as below.
| Probe | 5 μM | |
| P5 primer | 5 μM | |
| P7 primer | 5 μM |
Store in -20 °C.
P5 primer: 5'-AATGATACGGCGACCACCGA-3'
P7 primer: 5'-CAAGCAGAAGACGGCATACGA-3’
Prepare the following qPCR master mix per reactions for the number of your samples (+ calibrators).
| Prime Time PCR master mix | 10 μl | |
| 20x primers & probe mix | 1 μl | |
| H2O | 8 μl |
Distribute above to each well.
Add 1 μl of template (diluted library or calibrator) to each well.
Conduct PCR with the following condition.
95 °C for 2 min
30 cycles of
97 °C for 5s
62 °C for 10s
68 °C for 15s (photo with a filter appropriate for SYBR-green or FAM)
Result
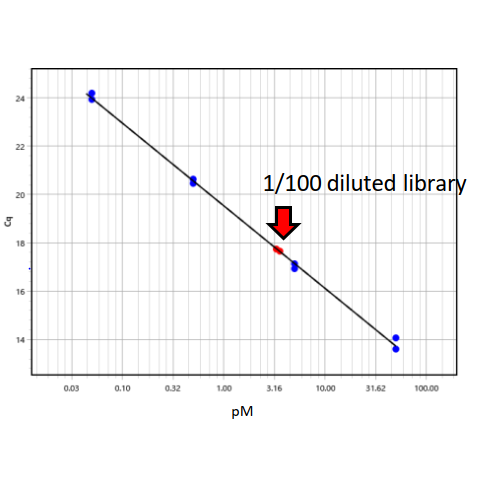
An example of expected result
Calculate the original concentration of your library.
