Mar 30, 2026
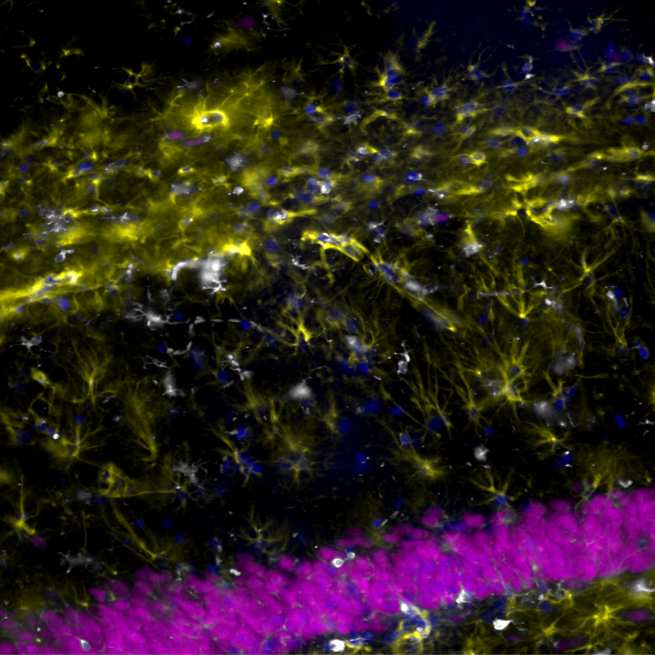
IHF AEGING GFAP + Iba1 + NeuN
- Aroa S. Maroto1,
- Silvia De Santis1
- 1Instituto de Neurociencias (Consejo Superior de Investigaciones Científicas - Universidad Miguel Hernández), San Juan de Alicante 03550, Spain

Protocol Citation: Aroa S. Maroto, Silvia De Santis 2026. IHF AEGING GFAP + Iba1 + NeuN. protocols.io https://dx.doi.org/10.17504/protocols.io.kxygx83j4v8j/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: January 21, 2026
Last Modified: March 30, 2026
Protocol Integer ID: 239008
Keywords: Iba1, GFAP, NeuN, Glia, Neuron, Rat brain, neun in rat brain section, neun immunofluorescence for the detection, neun immunofluorescence, rat brain section, gfap, using cryostat sectioning, fixed rat brain, cryostat sectioning, neuronal cell, iba1, rat brain
Abstract
Immunofluorescence for the detection of GFAP, Iba1, and NeuN in rat brain sections, with the aim of characterising the organisation and status of glial and neuronal cells in fixed rat brains, from which 30 µm thick coronal sections were obtained using cryostat sectioning.
Guidelines
This protocol is optimised for cryosections of 30 µm-thick adult rat brains that have been fixed in PFA and cryoprotected in 30% sucrose prior to embedding in OCT.
Materials
**EQUIPMENT**
- Multiwell 24 plate
- ThermoMixer
- 2ml microtubes
- Black tray
- Aluminum foil
- Tweezers
**CHEMICALS**
- Tri-sodium citrate Ref S4641
- PBS 1x
- Tween20 Ref: P7949
- Triton X-100 Ref: X100
- Goat Serum Ref: S26-M Sigma (GS)
- DAPI Ref: D9542
- VECTASHIELD® HardSet™ Ref H-1400
**BUFFERS**
- Sodium Citrate Buffer (10mM Sodium Citrate + 0.05% Tween20 pH6)
- Wash buffer (Wb) PBS-Tx (PBS 1x + 0.5% Triton)
- Blocking Buffer (Wb + 5% GS)
- Antibody Diluent (Wb + 2.5% GS)
**ANTIBODIES**
- GFAP Abcam ab46474 1/1000
- NeuN Merck MAB377 1/1000
- Iba1 Ref. 019-19741/WAKO 1/5000
- Anti-Chicken 488 ThermoFisher Alexa Fluor™ 488 A-11039 1/500
- Anti-Mouse 555 ThermoFisher Alexa Fluor™ 555 A-21424 1/500
- Anti-Rabbit 647 ThermoFisher Alexa Fluor™ 647 A-21245 1/500
**PROTECTIVE GEAR**
- Laboratory Coat
- Gloves
Troubleshooting
Safety warnings
DAPI is potentially mutagenic
Use laboratory coat and globes.
It is important that the tissue is not folded during the process, since this improves the antigen-antibody bond.
Ethics statement
All experimental procedures were approved by the Animal Care and Use Committee of the Instituto
de Neurociencias de Alicante (Spain) and were conducted in compliance with Spanish (Law 32/2007) and European regulations (EU Directive 86/609, EU Decree 2001-486, and EU Recommendation 2007/526/EC).
Before start
To succeed in this protocol, you need to have prior knowledge of the techniques involved in free flotation and immunohistochemistry, as well as knowing how to proceed. If you do not yet have the necessary experience, seek expert advice to learn the technical details.
Good luck!
Antigen Retrieval
50m
Select the slices to be performed and place one slice and citrate into 2 ml tubes. Place one slice in each tube and ensure they are completely submerged.
20m
Place the tubes in the thermoblock at 80ºC for 20 minutes. Shaking gently is not compulsory.
20m
Place around 3 slices/well on multi-well plate and wash with 500 ul wash buffer (Wb) 3 times x 5 min each. Kindly ensure that all the tissue in the well is thoroughly covered.
10m
Blocking and Primary antibody
19h 25m
Add 500 µl/well of blocking Buffer and incubate at RT
2h
Remove the blocking buffer
10m
Prepare Primary antibody dilution GFAP 1/1000 , NeuN 1/1000 and Iba1 1/5000 in antibody diluent
15m
Add 500 µl/well of the primary antibody to each well and incubate at 4 °C with gentle shaking overnight
17h
Secondary antibody
2h 45m
Remove primary antibody
10m
Add 500 µl/well Wb and shaking incubation for 5 min and remove buffer (Repeat this step minimum 3 times).
25m
Prepare Secondary antibody dilution
Anti-Chicken 488 1/500 for GFAP
Anti- Mouse 555 1/500 for NeuN
Anti-Rabbit 647 1/500 for Iba1
10m
Add 500 µl/well of the Secondary antibody to each well and incubate at RT with gentle shaking
2h
DAPI
1h 20m
Remove Secondary antibody
10m
Add 500 µl/well Wb and shaking incubation for 5 min and remove buffer (Repeat this step minimum 3 times)
25m
Add 500 µl/well of the DAPI to each well and incubate at RT with gentle shaking for 5 minutes
10m
Remove DAPI
10m
Add 500 µl/well Wb and shaking incubation for 5 min and remove buffer (Repeat this step minimum 3 times)
25m
Montage
25m
Bring the VECTASHIELD® HardSet™ to room temperature.
Mount the slices on the slides. Cover with approximately 50 µl of VECTASHIELD® HardSet™ per slice
15m
Put the cover slide in a dark box and leave it to dry
10m
You have your slices ready for the microscope
Protocol references
Acknowledgements
The authors gratefully acknowledge the professional staff of the Imaging Facility at the Instituto de Neurociencias for their technical support and assistance in optimizing image acquisition and processing procedures.
The authors gratefully acknowledge the professional staff of the Animal Facility (SEA) of the Universidad Miguel Hernández (UMH) for their excellent animal care.
