Dec 12, 2025
Hypoglossal motor neuron stain of mouse brainstem
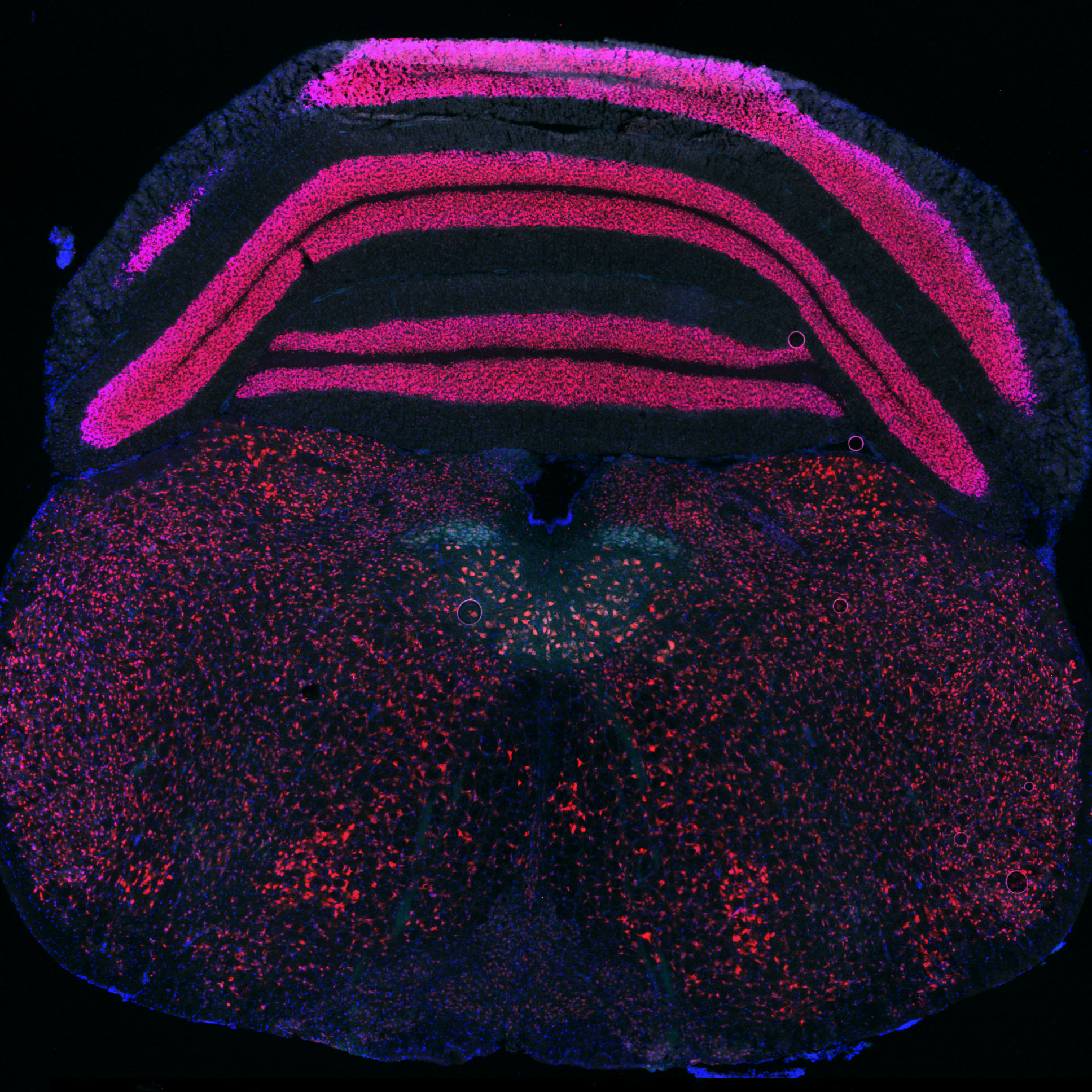
- Lucille Vue1,
- Miranda Cullins1,
- Tiffany J Glass1
- 1Division of Otolaryngology, Department of Surgery, University of Wisconsin-Madison, WI, USA

Protocol Citation: Lucille Vue, Miranda Cullins, Tiffany J Glass 2025. Hypoglossal motor neuron stain of mouse brainstem. protocols.io https://dx.doi.org/10.17504/protocols.io.dm6gpzb6dlzp/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: May 10, 2024
Last Modified: December 12, 2025
Protocol Integer ID: 99513
Keywords: Hypoglossal Nucleus, 12N, staining, NeuN, ChAT, neuron, brainstem, mouse, immunofluorescence, brain, hypoglossal motor neuron stain of mouse brainstem, hypoglossal motor neuron stain, hypoglossal nucleus motoneurons in mouse brainstem, hypoglossal nucleus motoneuron, hypoglossal neuron, area measurements on the hypoglossal nucleus, hypoglossal nucleus, mouse brainstem, neuronal nuclei, immunofluorescence, method for immunofluorescence, using antibody
Funders Acknowledgements:
NIH NICHD
Grant ID: R01HD104640-01A1
NIH NIDCD
Grant ID: R01DC019735-01A1
Abstract
This protocol details the method for immunofluorescence staining of the hypoglossal nucleus motoneurons in mouse brainstem using antibodies targeted against NeuN (Neuronal nuclei) and ChAT (Choline-Acetyltransferase). The generated images can be used to study the hypoglossal neurons, that appear NeuN+/ChAT+, and therefore to perform cell counts and area measurements on the hypoglossal nucleus.
Image Attribution
Lucille M Vue, University of Wisconsin-Madison
"Immunofluorescence stain of the mouse brainstem labeling cholinergic neurons"
Materials
Animals :
Ts65Dn mice (JAX, RRID:IMSR_JAX:005252)
Reagents :
Phosphate Buffer Saline 0.01M, pH 7.4 at 25°C (Sigma-Aldrich, P3813)
16% Paraformaldehyde 10mL vials (Ted Pella, 18505)
Tri-Sodium Citrate (dihydrate) (Sigma-Aldrich, C8532)
Tween‱-20 (Sigma-Aldrich, P1379)
Normal Donkey Serum (Sigma-Aldrich, D9663)
Triton‱ X-100 (Sigma-Aldrich, T8787)
Prolong‱ Gold Antifade Reagent with DAPI (Invitrogen, P36931)
Antibodies :
Chicken anti-NeuN (Neuronal Nuclei) (Millipore-Sigma, Cat#ABN91, RRID:AB_11205760)
Goat anti-ChAT (Choline AcetylTransferase) (Sigma-Aldrich, Cat#AB144P, RRID:AB_2079751)
Cy3 Donkey anti-chicken (Jackson Immunoresearch, Cat#703-165-155, RRID:AB_2340363)
Cy5 Donkey anti-goat (Jackson Immunoresearch, Cat#705-175-147, RRID:AB_2340415)
Equipment :
pH-meter
Programmable Microwave 700W, 2450 MHz : Danby DMBW0720ASD
Orbital Shaker (Rotating Platform)
Vortex Mixer
Epifluorescence microscope (Olympus, BX53) with Blue (Semrock, DAPI-1160B-OFF-ZERO), Red (Semrock, TRITC-B-OFF-ZERO) and Far Red (Semrock, Cy5-4040C-OFF-ZERO) channels.
Others :
Wet Ice
Distilled Water
Microwave-Safe Coplin Jars
Slide Case
Troubleshooting
Safety warnings
Paraformaldehyde (PFA) should only be handled with appropriate personal protective equipment such as gloves and protective clothing (lab coat) under a chemical hood. Workers should review the material safety data sheet (MSDS) available online and their laboratory's chemical safety guidelines before working with PFA.
DAPI should only be handled with appropriate personal protective equipment such as gloves and protective clothing (lab coat). Workers should review the material safety data sheet (MSDS) available online and their laboratory's chemical safety guidelines before working with PFA.
Ethics statement
This protocol describes methods used to analyze tissues of mice that have been euthanized in accordance with a protocol approved by the University of Wisconsin School of Medicine and Public Health institutional animal care and use committee (IACUC).
Before start
This protocol is optimized for 25 μm sections fresh unfixed mouse brainstem sections on slides. The objective of this protocol is to generate immunofluorescence imaging of the mouse hypoglossal nucleus to perform cell counts and area measurements.
The mouse model used for this experiment is the Ts65Dn model from Jackson Laboratories, stock number 005252 (RRID:IMSR_JAX:005252). Animals should be euthanized using carbon dioxide at different age points, namely post-natal day 7 (p7), p21, p35, 5 months and 20 months. For p7 animals, a secondary euthanasia method such as quick decapitation should be applied to ensure proper euthanasia.
Brains should be frozen in dry ice powdered using a pestle and mortar and stored at -80ºC until sectioning. Every other section should be collected onto slides, then a hydrophobic barrier should be drawn along the perimeter of the slide with a hydrophobic pen. After sectioning, slides can be stored at -80ºC for up to two weeks prior to staining.
Each experimental iteration should include at least one experimental slide and one negative control slide of similar tissue from which the primary antibody will be omitted.
Staining - Day One
2h 5m
Fixation.
34m
Dilute a 10mL vial of 16% Paraformaldehyde (PFA) in 30mL of Phosphate Buffer Saline 0.01M (PBS) to create a 4% PFA solution.
Take slides out from storage and allow for a brief thaw. Place slides into a slide staining chamber containing a thin layer of water on the bottom.
4m
Apply 4% PFA to all slides. Incubate for 15 minutes at room temperature.
15m
PBS Wash : Remove the solution from the slides and apply PBS. Allow PBS to sit for 5 minutes.
5m
Repeat step 1.4 for a total of three times.
10m
Antigen Retrieval.
31m
Prepare Sodium Citrate Buffer following the directions below :
| A | B | |
| Sodium Citrate Buffer (10mM Sodium citrate, 0.05% Tween 20, pH 6.0) | ||
| Tri-Sodium Citrate (dihydrate) | 2.94g | |
| Distilled water | 1L | |
| Adjust pH to 6.0 with Hydrogen Chloride (HCl) | ||
| Tween® 20 | 0.5mL | |
| Mix well | ||
Fill a microwave-safe Coplin Jar with Sodium Citrate Buffer and insert the slides. Make sure the slides are completely covered with solution.
Microwave 1 minute at maximum power (700W).
1m
Microwave 10 minutes at minimum power (10% of maximum power, or 70W).
10m
Transfer the Coplin Jar on ice to let it cool. When the solution has cooled enough for you to grab the slides, take them out and quickly dry the hydrophobic barrier.
5m
PBS Wash : Remove the solution from the slides and apply PBS. Allow PBS to sit for 5 minutes.
5m
Repeat step 2.6 for a total of three times.
10m
Blocking.
1h
Prepare a solution of Normal Donkey Serum (NDS) at 2% and 0.3% Triton‱ X-100 in PBS. Vortex thoroughly.
Apply blocking solution to all slides. Incubate for 1 hour at room temperature.
1h
Primary Antibody Incubation.
1d
Prepare 250 μL of primary antibody cocktail containing antibodies targeted against Neuronal Nuclei (NeuN) and Choline-Acetyltransferase (ChAT) for each slide. Vortex to mix.
| Catalog Number | RRID | Host | Target | Concentration | Volume | |
| ABN91 | AB_11205760 | Chicken | NeuN | 1:500 | 0.5μL | |
| AB144P | AB_2079751 | Goat | ChAT | 1:50 | 5μL | |
| NDS 2% | 244.5μL | |||||
Apply primary antibodies. Do not add to the negative control slide. Instead, apply NDS 2%.
Close the slide case, place on a rotating platform set on very low speed at room temperature and incubate overnight.
1d
Staining - Day Two
2h 30m
Secondary Antibody Incubation.
2h 30m
Prepare 200 μL of primary antibody cocktail for each slide. Vortex to mix.
| Catalog Number | RRID | Fluorophore | Host | Target | Concentration | Volume | |
| 703-165-155 | AB_2340363 | Cy3 (red) | Donkey | Chicken | 1:200 | 1μL | |
| 705-175-147 | AB_2340415 | Cy5 (far red) | Donkey | Goat | 1:200 | 1μL | |
| NDS 2% | 198µL | ||||||
Take the slide case off the shaker and wipe off any condensation under the slides and inside the lid of the case.
PBS Wash : Remove the solution from the slides and apply PBS. Allow PBS to sit for 5 minutes.
5m
Repeat step 5.3 for a total of three times.
10m
Apply secondary antibodies to all slides.
Close the slide case, place on the shaker at room temperature and incubate 2 hours.
2h
PBS Wash : Remove the solution from the slides and apply PBS. Allow PBS to sit for 5 minutes.
5m
Repeat step 8.8 for a total of three times.
10m
Mounting.
Add 2-3 drops of Prolong‱ Gold antifade reagent with DAPI to each slide.
Add a cover slip to cover the slide, by placing the edge of the cover slip next to the sample and lowering the slip slowly to avoid forming bubbles.
Place the slides in a slightly opened container in the dark at 4°C and let the mounting media dry for at least 24 hours.
1d
Microscopy
The microscope used in this experiment is the Olympus BX53 fluorescence microscope with an XYZ motorized stage control. The software used is Olympus CellSens Dimension 4.1. Brainstem images are acquired with the UPlanSapo 4x/0.16 objective (Olympus), and hypoglossal nucleus images are acquired with the UPlanSapo 10x/0.40 objective(Olympus).
Image the slides within 2 weeks. Use the negative control slide to validate the image acquisition settings for each staining run.
Example of a negative control slide acquisition of the hypoglossal nucleus stain of mouse brainstem (post natal day 35) under a 4x objective.
(Blue : DAPI, Red : NeuN, Cyan : ChAT)
Capture a 4x acquisition of the entirety of each slide on three color channels : Blue, Red and Far Red. Make sure the signal is bright enough and that positive areas on each channel can be easily determined.
Example of entire slide acquisition of the hypoglossal nucleus stain of mouse brainstem (post natal day 35) under a 4x objective
(Blue : DAPI, Red : NeuN, Cyan : ChAT)
Determine which sections contain the hypoglossal nucleus from the 4x overviews of each slide. The hypoglossal nuclei are located around the central canal and beginning of the 4th ventricle, both structures appearing on the blue channel. They are characterized by large neuronal cells that are both ChAT (far red channel) and NeuN (red channel) positive. The average length of the hypoglossal nucleus is about 1mm, so there should be an average of 20 sections containing it.
Example of a section containing the hypoglossal nucleus in mouse brainstem (post natal day 35), made visible by NeuN (red) and ChAT (cyan) immunofluorescence under a 4x objective
(Blue : DAPI, Red : NeuN, Cyan : ChAT)
Capture a 10x acquisition of the area of the hypoglossal nucleus on each section containing the hypoglossal nucleus on three color channels : Blue, Red and Far Red. Make sure the signal is bright enough and that positive areas on each channel can be easily determined.
Example of a hypoglossal nucleus stain in mouse brainstem (post natal day 35) made visible by NeuN (red) and ChAT (cyan) immunofluorescence under a 10x objective (Blue : DAPI, Red : NeuN, Cyan : ChAT)
Protocol references
ChAT reactivity of hypoglossal neurons :
Chen, Xin, et al. “Transcriptional Regulation of Mouse Hypoglossal Motor Neuron Somatotopic Map Formation.” Brain Structure & Function, vol. 221, no. 8, Nov. 2016, pp. 4187–202. PubMed, https://doi.org/10.1007/s00429-015-1160-2.
NeuN reactivity of hypoglossal neurons :
Osman, Kate L., et al. “Optimizing the Translational Value of Mouse Models of ALS for Dysphagia Therapeutic Discovery.” Dysphagia, vol. 35, no. 2, Apr. 2020, pp. 343–59. PubMed Central, https://doi.org/10.1007/s00455-019-10034-9.
NeuN and ChAT staining :
Moore, Jeffrey D., et al. “Vibrissa Self-Motion and Touch Are Reliably Encoded along the Same Somatosensory Pathway from Brainstem through Thalamus.” PLOS Biology, vol. 13, no. 9, Sept. 2015, p. e1002253. PLoS Journals, https://doi.org/10.1371/journal.pbio.1002253.
Antigen Retrieval to improve ChAT immunoreactivity :
Aziz, Nadine M., et al. “Spatiotemporal Development of Spinal Neuronal and Glial Populations in the Ts65Dn Mouse Model of Down Syndrome.” Journal of Neurodevelopmental Disorders, vol. 11, Dec. 2019, p. 35. PubMed Central, https://doi.org/10.1186/s11689-019-9294-9.
Antigen Retrieval protocol :
IHC Antigen Retrieval Protocol | Abcam. https://www.abcam.com/protocols/ihc-antigen-retrieval-protocol. Accessed 21 May 2024.
