Sep 13, 2025
Hydrogen Cyanide (HCN) Detection Assay
- Md Sahadat Ali1,
- Jonathan D. Eisenback1,
- Fatima Tuz Zohora Mony1
- 1Virginia Tech

Protocol Citation: Md Sahadat Ali, Jonathan D. Eisenback, Fatima Tuz Zohora Mony 2025. Hydrogen Cyanide (HCN) Detection Assay. protocols.io https://dx.doi.org/10.17504/protocols.io.dm6gpmy98gzp/v1
Manuscript citation:
Ali MS, Mony FTZ, Evans M, Rideout S, Haak D, Eisenback JD (2025)
Unveiling the antagonistic activity of ‘Candidatus Pseudomonas auctus’ JDE115 against Agroathelia rolfsii: a soybean nodule endophyte with biocontrol potential. (In preparation)
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: September 12, 2025
Last Modified: September 13, 2025
Protocol Integer ID: 227137
Keywords: Hydrogen cyanide, HCN, picrate paper, Pseudomonas, volatile metabolites, biocontrol, rhizosphere, protocol detects hydrogen cyanide, hcn biosynthesi, hydrogen cyanide, candidatus pseudomonas auctus, hcn, picrate paper assay, bacteria, cultured on nutrient, assay
Disclaimer
This protocol is provided for research and educational purposes only. While validated in our laboratory, results may vary with strains, media, and conditions. Users are responsible for complying with biosafety and chemical safety regulations. The authors and Virginia Tech assume no liability for misuse or misapplication.
Abstract
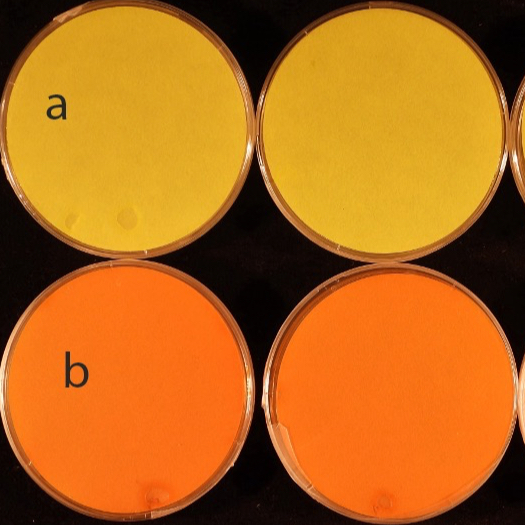
This protocol detects hydrogen cyanide (HCN) production by ‘Candidatus Pseudomonas auctus’ JDE115 using a picrate paper assay. Bacteria are cultured on nutrient sucrose agar supplemented with glycine (4.4 g L⁻¹) to enhance HCN biosynthesis. Plates are sealed with a picrate-impregnated filter paper on the lid and incubated at 28 °C for 7 days. A positive reaction is indicated by a visible yellow-to-orange/reddish-brown color change of the paper. Five biological replicates are recommended, with the experiment repeated independently at least three times for reproducibility.
Attachments
Image Attribution
All assay images (e.g., plate setups and end-point color changes) were generated by the authors at Virginia Tech. Images are original and may be used for academic and research purposes with appropriate citation of this protocol.
Guidelines
- Prepare agar and picrate papers as needed.
- Keep plates sealed during incubation to prevent volatile loss.
- Include appropriate controls (medium-only; optional HCN-negative strain).
- Standardize plate volume and incubation conditions across replicates.
Materials
- ‘Candidatus P. auctus’ JDE115 or any Plant growth-promoting rhizobacteria (PGPR) bacteria
- Nutrient sucrose agar components (per L): Sucrose 5.0 g; Yeast extract 4.0 g; Peptone 4.0 g; Beef extract 2.0 g; Agar 15.0 g; dH₂O to 1 L
- Glycine (add to 4.4 g L⁻¹ final concentration)
- Picrate solution: 0.5% (w/v) picric acid in 1% (w/v) sodium carbonate (Na₂CO₃), freshly prepared
- Sterile filter paper
- LB medium (negative control)
- Sterile deionized water
- Optional: known HCN-negative strain (reference control)
Equipment:
- Autoclave
- Petri dishes
- Whatman No. 1 filter paper
- Forceps
- Parafilm‱/sealing tape
- Incubator (25 °C)
- Magnetic stir plate and stir bars
- 1 L borosilicate bottles
- Fume Hood/Bio-Safety Cabinet
- Camera or scanner for documentation
Troubleshooting
Safety warnings
- Picric acid is an energetic compound when dry. Keep picrate solutions and papers moist; store and dispose per institutional policy.
- Wear lab coat, nitrile gloves, and eye protection. Follow local chemical and biosafety regulations at all times.
- Remove picrate papers in a fume hood and collect as energetic/oxidizer chemical waste per institutional policy.
- After removing picrate papers, autoclave plates and dispose of biological material as regulated medical waste.
- Decontaminate work surfaces and tools after use.
Before start
- Calibrate pipettes and verify incubator temperature (25 °C).
- Pre-label all plates (sample IDs, date, replicate number).
- Set up a fume hood/BSC for plate sealing and end-point inspection.
- Prepare dedicated waste containers for picrate papers and biological materials.
Picrate paper preparation
Prepare 0.5% (w/v) picric acid in 1% (w/v) Na₂CO₃.
Soak filter paper for ~5 min.
Drain excess; place on sterile surface until uniformly moist (do not allow to dry).
Store short-term in a sealed, labeled container to prevent drying.
Nutrient sucrose agar + glycine (per 1 L)
Sucrose 5.0 g; Yeast extract 4.0 g; Peptone 4.0 g; Beef extract 2.0 g; Agar 15.0 g; Glycine 4.4 g; dH₂O to 1,000 mL.
Plate preparation (Nutrient sucrose agar + glycine)
- Label a 1 L bottle; add a sterile stir bar and ~800 mL dH₂O.
- Add sucrose, yeast extract, peptone, beef extract, and glycine (4.4 g L⁻¹); stir to dissolve.
- Add agar gradually while stirring to avoid clumps; bring to 1,000 mL with dH₂O.
- Cap loosely; autoclave at 121 °C for 15 min (liquid cycle).
- Cool to ~50–55 °C; in a sterile area, pour ~20–25 mL per 90 mm plate. Allow to solidify.
Note: Pouring should be done inside the BSC.
Inoculation and setup
- From a fresh colony (grown on LB agar for 48 h at 28°C), streak or spot-inoculate the center of each glycine-supplemented plate.
- Prepare controls:
Medium-only (no bacteria) to verify picrate color stability.
Optional HCN-negative strain as procedural control.
- Using sterile forceps, affix one moist picrate paper to the underside of the plate lid (ensure it does not touch agar).
- Close and seal plates with Parafilm‱ to retain volatiles.
- Keep picrate papers moist at all times; drying reduces sensitivity and increases hazard.
- If condensation forms on lids, allow plates to equilibrate at room temperature before sealing to minimize drip.
Incubation
Incubate sealed plates at 25 °C for 7 days. Do not open during incubation.
Readout and documentation
- In a fume hood, examine picrate papers:
Positive: yellow → orange/reddish-brown.
Negative: no change (remains yellow).
- Photograph or scan each plate/lid for records.
- For semi-quantitative comparison across strains, standardize lighting and use image analysis (hue/saturation) against control references.
- Perform ≥5 biological replicates and repeat the assay ≥3 times independently.
Protocol references
Lorck H. (1948). Production of hydrocyanic acid by bacteria.Physiol. Plant1142–146. 10.1111/j.1399-3054.1948.tb07118.x
Blumer C, Haas D (2000) Mechanism, regulation, and ecological role of bacterial cyanide biosynthesis. Arch Microbiol 173:170–177. https://doi.org/10.1007/s002039900127
Lo Cantore P, Giorgio A and Iacobellis NS (2015) Bioactivity of volatile organic compounds produced by Pseudomonas tolaasii. Front. Microbiol. 6:1082. doi: 10.3389/fmicb.2015.01082
Acknowledgements
We acknowledge the Nematode Diagnostic Lab at Virginia Tech for providing facilities and resources essential for conducting this work.

