Dec 23, 2023
Human iPSCs culture and cardiomyocyte subtype differentiation in fully chemically defined conditions
This protocol is a draft, published without a DOI.
- Joanna Delimata-Raczek1,
- Natalia Koralewska1,
- Marek Figlerowicz1,
- Ireneusz Stolarek1
- 1Institute of Bioorganic Chemistry Polish Academy of Sciences
- MOSAIC

Protocol Citation: Joanna Delimata-Raczek, Natalia Koralewska, Marek Figlerowicz, Ireneusz Stolarek 2023. Human iPSCs culture and cardiomyocyte subtype differentiation in fully chemically defined conditions . protocols.io https://protocols.io/view/human-ipscs-culture-and-cardiomyocyte-subtype-diff-c5ymy7u6
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: December 06, 2023
Last Modified: December 23, 2023
Protocol Integer ID: 91885
Keywords: cardiomyocyte, differentiation, in vitro, cell culture, iPSC, inducing cardiomyogenesi, nuanced stages of cardiomyocyte differentiation, cardiomyocyte subtype differentiation, cardiomyocyte differentiation, differentiation into distinct cardiomyocyte subtype, distinct cardiomyocyte subtype, unprecedented insights into cardiac development, cardiomyocyte, cardiac development, induced pluripotent stem cell, pluripotent stem cell, pivotal avenue in stem cell research, stem cell research, cultivating ips cell, advent of induced pluripotent stem, induced pluripotent stem, regenerative medicine, ips cell, cell technology
Abstract
Cardiomyocyte in vitro differentiation represents a pivotal avenue in stem cell research, offering unprecedented insights into cardiac development and disease understanding. Over the years, protocols for inducing cardiomyogenesis have evolved substantially, particularly with the advent of induced pluripotent stem (iPS) cell technology. The transition from intricate and uncontrolled systems to more refined and robust protocols has been paramount, driven by a cumulative understanding of the molecular and cellular cues governing the process.
This protocol addresses the intricacies of cultivating iPS cells and orchestrating their differentiation into distinct cardiomyocyte subtypes. The protocol encompasses not only the fundamental aspects of human induced pluripotent stem cell (hiPSC) culture but also delves into the nuanced stages of cardiomyocyte differentiation, sub-type specification, and the critical maturation phase. The refined methodologies outlined in this protocol contribute to advancing capabilities in disease modeling, drug discovery, developmental biology, and regenerative medicine.
Protocol materials
ROCK inhibitor (Ri) Y-27632STEMCELL Technologies Inc.Catalog #72308
Essential 8™ Flex Medium KitThermo FisherCatalog #A2858501
Penicillin/StreptomycinThermo Fisher ScientificCatalog #Invitrogen 15140-122
Pluripotent Stem Cell 4-Marker Immunocytochemistry KitThermo FisherCatalog #A24881
Lookout® One-Step Mycoplasma PCR Detection KitMerck MilliporeSigma (Sigma-Aldrich)Catalog #MP0050-100TST
dimethylsulfoxide (DMSO)Merck MilliporeSigma (Sigma-Aldrich)
B-27™ Supplement, minus insulinGibco - Thermo Fisher ScientificCatalog #A1895601
Penicillin-Streptomycin (5,000 U/mL)Thermo FisherCatalog #15070063
RPMI 1640 Medium, GlutaMAX™ Supplement, HEPESThermo FisherCatalog #72400021
B-27™ Supplement (50X), serum freeGibco - Thermo Fisher ScientificCatalog #17504044
IWP-2 1 mg
STEMCELL Technologies Inc.Catalog #72122
StemMACS™ CHIR99021Miltenyi BiotecCatalog #130-103-926
Retinoic acidMerck MilliporeSigma (Sigma-Aldrich)Catalog #R2625-50MG
Human Cardiomyocyte Immunocytochemistry KitThermo FisherCatalog #A25973
Fetal Bovine Serum, qualified, One Shot™ format, United StatesThermo FisherCatalog #A3160501
DMEM/F-12Thermo FisherCatalog #11320033
Trypan Blue Solution 0.4% Sterile-filtered Merck MilliporeSigma (Sigma-Aldrich)Catalog #T8154
PSC Cardiomyocyte Differentiation KitThermo FisherCatalog #A2921201
StemPro™ Accutase™ Cell Dissociation ReagentThermo Fisher ScientificCatalog #A1110501
Geltrex LDEV Free hESC Quality 5 mlThermo Fisher ScientificCatalog #A1413302
Troubleshooting
Safety warnings
Appropriate safety procedures are recommended to be used when handling all cell lines, working with iPSCs and induced cardiomyocytes handle them under at least BSL-1 containment.
Before start
Keep in mind that while working with cells everything should be kept sterile, every medium or any reagent should be prepared under the sterile hood, autoclaved, or filtered through 0,2um filter.
iPSC culture and maintenance
Coating culture plates with ECM proteins for enhanced attachment of iPSCs.
ThawGeltrex LDEV Free hESC Quality 5 mlThermo Fisher ScientificCatalog #A1413302 on ice 4 °C Overnight . It is prohibited to thaw Geltrex at room temperature or in a water bath. Remember to keep it on ice during the whole aliquoting procedure and use only ice-cold pipette tips and tubes.
Note
Making small aliquot portions of Geltrex is recommended to limit the freeze-thaw cycles. *Storage of aliquots in -20 °C freezer for up to 1 year.
12h
Resuspend single Geltrex aliquot in cold DMEM/F-12Thermo FisherCatalog #11320033 medium to obtain desired dilution. Store diluted coating medium in the fridge.
Note
1:60-1:100 dilution usually works fine but it should be first tested on a particular cell line.
Coat 6-well culture plate by adding 1 mL of coating medium to each well and cover the entire surface of each well. Incubate the plate in the incubator for at least 00:30:00 before proceeding further.
Note
Coated plates can be used immediately after removal of the coating medium or stored in the fridge for up to 2 weeks with added PBS and wrapped in parafilm to prevent from drying.
30m
iPSC thawing
Prepare culture medium using the following kit:Essential 8™ Flex Medium KitThermo FisherCatalog #A2858501 : add 48.5 mL of Essential 8 flex medium to the 50 mL centrifuge tube and 1 mL of 10x supplement. It is not necessary, but it is advised to also add 500 µL of Penicillin/StreptomycinThermo Fisher ScientificCatalog #Invitrogen 15140-122 to prevent cell culture from contamination.
Prepare the previously coated 6-well plate by adding 2 mL of warm culture medium to each well and 2 µL from 10 millimolar (mM) stock solution ofROCK inhibitor (Ri) Y-27632STEMCELL Technologies Inc.Catalog #72308 to minimize the cell death.
Note
Preparing 10mM stock solution of rock inhibitor Y-27632
1. Dissolve 5 mg of rock inhibitor in 1.56 mL DMSO.
2. Mix well until it dissolves completely.
3. Prepare small aliquot portions, for example 10 µL in PCR tubes and keep them in the -20 °C freezer .
Take out the desired cryovial containing frozen cells from the liquid nitrogen and quickly transfer it to 37 °C water bath. Be careful not to submerge the cap of the cryovial. You can swirl the tube a little to help it thaw quicker but pay attention to save a little ice cube inside the cryovial before taking it out. Disinfect the cryovial with 70% ethanol and put it under the hood.
Transfer 1 mL of thawed cell suspension to the 15 mL centrifuge tube and add 9 mL of warm culture medium very gently drop by drop, swirling the tube to minimize osmotic shock to the cells.
Centrifuge the cell suspension 1400 rpm, Room temperature, 00:05:00 . Carefully remove and discard the supernatant. Gently add 1 mL of medium to the remaining cell pellet. Gently resuspend cells in medium and seed into 2 wells of a previously prepared 6-well plate slowly drop by drop.
5m
Gently rock plate side to side, and back and forth to spread the cells evenly across the well. Transfer the plate to the incubator. The next day in the morning change the medium to fresh culture medium without rock inhibitor.
iPSC culture
Incubate the cells at 37 °C/ 5% CO2 incubator and observe their growth and confluence under the microscope daily. Perfect undifferentiated iPSC colonies should look compact and present distinct borders. Cells present large nuclei and smaller cytoplasm region. It is essential to check iPSCs for the presence of spontaneous differentiation. If any signs of differentiation occur, colonies should be discarded immediately.
Expected result
Morphological differences between correct and incorrect iPSC colonies
white arrows present differentiated regions
Medium should be changed every day. When using Essential 8™ Flex Medium KitThermo FisherCatalog #A2858501 it is possible to skip weekend medium change by adding 2x more medium on Friday afternoon, but it is only recommended when cells are at low confluency.
Pluripotency characterization
To guarantee the reliability of the research iPSC lines should be verified for pluripotency markers either by qPCR or immunocytochemistry. The most common pluripotency markers are OCT4, SOX2, SSEA4, TRA-1-60, and alkaline phosphatase.
Expected result
iPSC colonies present positive staining for pluripotency markers OCT4 and SSEA4 usingPluripotent Stem Cell 4-Marker Immunocytochemistry KitThermo FisherCatalog #A24881
Mycoplasma testing
It is very important to maintain contamination-free cultures. A commonly known good laboratory practice is to regularly check cell cultures for mycoplasma. Mycoplasma contamination is very hard to notice without special tests. This contamination usually does not cause cell death, but it negatively affects the cell metabolism and growth making the research not reliable. The best way to assess mycoplasma contamination in culture is to use available qPCR or immunocytochemistry ready-to-use kits, for example:Lookout® One-Step Mycoplasma PCR Detection KitMerck MilliporeSigma (Sigma-Aldrich)Catalog #MP0050-100TST
iPSC passage
The optimal time for harvest or passage for iPSCs is typically when cells reach approximately 70-80% confluency.
To start with prepare mild cell detachment solution, for example PBS with 0.5 millimolar (mM) EDTA. Warm it to Room temperature before adding it to the cells.
Prepare a new Geltrex-coated plate as in steps and #2.2
Wash cells with warm PBS first, then add 1 mL of cell detachment solution to each well in a 6-well plate format. Transfer the plate to the incubator 37 °C . After 00:03:00 take out the plate and observe the cells under the microscope. iPSCs colonies should look loose and be ready to detach, if not please transfer the plate back to the incubator for one minute or two more, but do not exceed the incubation time and do not let the iPSC colonies detach completely and float in the solution.
3m
Remove the cell detachment solution and rinse the cells with 1 mL of warm culture medium. Pipette the cells a couple of times, but be careful not to over-pipette the cells. The cells should stay in clumps and easily detach from the well. Transfer the appropriate amount to new wells.
You can split the cells using ratio for example 1:6 to 1:30 or take a small amount for counting first to have an exact number of cells in a new well.
Note
Cell counting
1. Take 10 µL of cell suspension from the well and transfer it to a small PCR tube.
2. Pipette cell suspension a couple of times to make asingle-cell suspension.
3. Add 10 µL of Trypan Blue Solution 0.4% Sterile-filtered Merck MilliporeSigma (Sigma-Aldrich)Catalog #T8154
and mix well by pipetting
4. Take 10 µL for counting using a cell counter or Neubauer chamber.
Agitate the plate gently from side to side and forwards and backwards to ensure that the cell aggregates are evenly distributed across the surface of the well containing culture medium with theaddition of rock inhibitor.
The next day in the morning, exchange the medium to fresh culture medium without rock inhibitor. Continue to change the medium daily until iPSCs reach confluency again.
iPSC cryopresevation
Prepare cryopreservation medium: culture medium without antibiotics with 5%-10% dimethylsulfoxide (DMSO)Merck MilliporeSigma (Sigma-Aldrich) . The cryopreservation medium should be stored in the fridge and keep cold during the procedure.
Firstly perform cell detachment as described in steps and #6.6 but instead of adding the cell suspension to the new well transfer it to 15 mL centrifuge tube.
Centrifuge the tube 1400 rpm, Room temperature, 00:05:00
5m
Remove the supernatant with care not to move the cell debris. Add an appropriate amount of cold cryopreservation medium to the tube, and resuspend the cells. Transfer cryotubes to a freezing container and put them in -80 °C freezer Overnight . The next day, you can transfer cryotubes to the liquid nitrogen for longer storage.
Note
For example, it is recommended to use 1 mL of cell suspension for 1 cryotube and freeze around 1x10^6 live cells per vial.
Equipment
Mr. Frosty Freezing Container
NAME
freezing container
TYPE
ThermoFisher Scientific
BRAND
5100-0001
SKU
LINK
Differentiation of hiPSCs into cardiomyocytes
Standard iPSC culture before differentiation.
When cells become confluent passage them to prepared geltrex-coated 12-well plate.
View cells under microscope every day to estimate their confluence. When cells reach desired confluence between 60%-80% they are ready for differentiation.
Differentiation of hiPSCs into cardiomyocytes using commercially available kitPSC Cardiomyocyte Differentiation KitThermo FisherCatalog #A2921201
On day 0 of the differentiation remove iPSC culture medium from well and replace it with Cardiomyocyte Differentiation Medium A from the kit.
On day 2 replace the medium with Cardiomyocyte Differentiation Medium B from the kit.
Note
On this step of protocol cells may start to look different, they become more opaque and many dead cells are floating in the medium.
On day 4 replace the medium with Cardiomyocyte Maintenance Medium. Change the medium every 2-3 days with Cardiomyocyte Maintenance Medium. Starting from day 8 cells should begin to contract.
Subtype differentiation of hiPSCs into cardiomyocytes
25m
Standard iPSC culture before differentiation.
When cells become confluent passage them to prepared geltrex-coated 12-well plate.
Differentiation to cardiomyocyte subtypes
Note
When iPSCs reach 70-90% confluence they are ready to start differentiation
In the beginning, prepare two types of medium:
1. Cardiomyocytes induction medium
2. Cardiomyocytes culture medium
Note
1. Cardiomyocytes induction medium: RPMI 1640 Medium, GlutaMAX™ Supplement, HEPESThermo FisherCatalog #72400021 with 1x B-27™ Supplement, minus insulinGibco - Thermo Fisher ScientificCatalog #A1895601 from 50x stock and 1% of Penicillin-Streptomycin (5,000 U/mL)Thermo FisherCatalog #15070063
2. Cardiomyocytes culture medium: RPMI 1640 Medium, GlutaMAX™ Supplement, HEPESThermo FisherCatalog #72400021 with 1x B-27™ Supplement (50X), serum freeGibco - Thermo Fisher ScientificCatalog #17504044 from 50x stock and 1% of Penicillin-Streptomycin (5,000 U/mL)Thermo FisherCatalog #15070063
Prepare small molecules working solutions of CHIR99021, IWP2 and retinoic acid (RA)
Note
1. Dissolve StemMACS™ CHIR99021Miltenyi BiotecCatalog #130-103-926 4.66 mg/mL in DMSO 37 °C 00:05:00 to obtain 10 millimolar (mM) working solution, aliquot small portions, and store in the freezer-20 °C
2. Dissolve IWP-2 1 mg
STEMCELL Technologies Inc.Catalog #72122 2.34 mg/mL in DMSO 37 °C 00:20:00 to obtain 5 millimolar (mM) working solution, aliquot small portions, and store in the freezer-20 °C
3. Dissolve Retinoic acidMerck MilliporeSigma (Sigma-Aldrich)Catalog #R2625-50MG 29.94 mg/mL in DMSO to obtain 100 millimolar (mM) master stock, aliquot 10 µL portions, and store in the freezer -80 °C . To obtain 1 millimolar (mM) working solution thaw and dilute RA aliquot in 990 µL EtOH and store in the fridge for 2 weeks, protecting from light.
Atrial subtype differentiation
1w 1d
On day 0 change the iPSC culture medium to cardiomyocytes induction medium with an addition of 5 micromolar (µM) CHIR99021 (0.5 µL /ml from 10 millimolar (mM) working stock solution ).
After 48h on day 2 change the medium to fresh cardiomyocytes induction medium with an addition of 5 micromolar (µM) IWP2 (1 µL /ml from 5 millimolar (mM) working stock solution ).
On day 3, without changing the medium add 1 µL /ml RA 1 micromolar (µM) final from 1 millimolar (mM) working stock solution .
On day 4 change the medium to fresh cardiomyocytes induction medium with an addition of1 micromolar (µM) RA.
On day 6 change the medium to fresh cardiomyocytes induction medium.
From day 8 change the medium to cardiomyocytes culture medium and continue media change every 2-3 days.
Ventricular subtype differentiation
1w 1d
On day 0 change the iPSC culture medium to cardiomyocytes induction medium with an addition of 5 micromolar (µM) CHIR99021 (0.5 µL /ml from 10 millimolar (mM) working stock solution ).
After 48h on day 2 change the medium to fresh cardiomyocytes induction medium with an addition of 5 micromolar (µM) IWP2 (1 µL /ml from 5 millimolar (mM) working stock solution ).
On day 4 change the medium to fresh cardiomyocytes induction medium.
On day 6 change the medium to fresh cardiomyocytes induction medium.
From day 8 change the medium to cardiomyocytes culture medium and continue media change every 2-3 days.
Nodal subtype differentiation
1w
On day 0 change the iPSC culture medium to cardiomyocytes induction medium with an addition of 12 micromolar (µM) CHIR99021 (1.2 µL /ml from 10 millimolar (mM) working stock solution ).
After 24h on day 1 change the medium to fresh cardiomyocytes induction medium.
On day 3 change the medium to fresh cardiomyocytes induction medium with an addition of of 5 micromolar (µM) final IWP2 (1 µL /ml from 5 millimolar (mM) working stock solution .)
On day 5 change the medium to fresh cardiomyocytes induction medium with an addition of 3 micromolar (µM) CHIR99021 (0.3 µL /ml from 10 millimolar (mM) working stock solution ).
From day 7 change the medium to cardiomyocytes culture medium and continue media change every 2-3 days.
Cardiomyocytes culture, characterization, passaging and cryopreservation
25m
Longer culture
Cardiomyocytes and their specific subtypes usually start contracting after 8-10 days from the beginning of the differentiation. After 12-15 days they become defined enough to be safely passaged or cryopreserved. Cells can be kept in a culture for at least 1 month or even longer if such experiments are desired.
Note
With time cardiomyocytes become more mature and gain more cardiomyocyte-like characteristics.
Characterization of cardiomyocytes
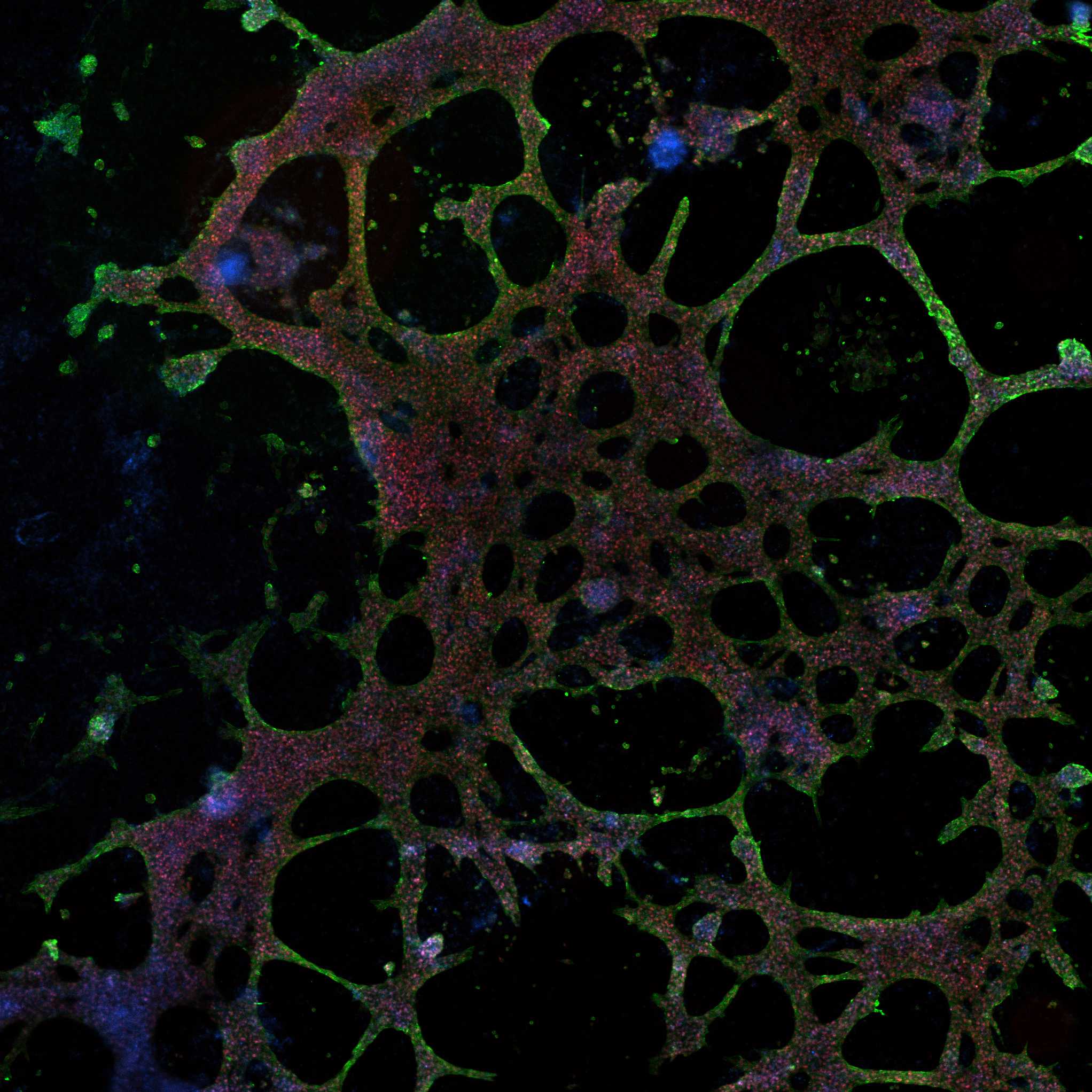
Successfully differentiated cells display completely different morphology, with characteristic net-like structure. The most important feature is their ability to contract. It is also recommended to perform PCR analysis or immunocytochemistry to confirm theexpression of key cardiac markers such as TNNT2 and NKX2.5.
Immunocytochemistry confirmation of key cardiac markers expression in differentiated cardiomyocyte-like cells using commercially available kit Human Cardiomyocyte Immunocytochemistry KitThermo FisherCatalog #A25973
Expected result
Passaging cardiomyocytes
Prepare a new Geltrex-coated plate
For better cell survival post-passaging it is recommended to add 10 micromolar (µM) ROCK inhibitor (Ri) Y-27632STEMCELL Technologies Inc.Catalog #72308 to the culture medium approximately 1 hour before passage.
Aspirate the medium and wash cells with warm PBS. Add warm StemPro™ Accutase™ Cell Dissociation ReagentThermo Fisher ScientificCatalog #A1110501 to the well (1 mL per well in 6-well plate ) and incubate 37 °C 00:05:00 -00:10:00
15m
After 5 minutes observe the cells under the microscope to see if they are ready to detach, if not continue to incubate up to 10 minutes maximum. When cells look ready to be detached pipette the solution a couple of times to detach the cells from the well, be careful not to overpipette the cells, because it may disrupt them.
Transfer them to 15 mL centrifuge tube, add an appropriate amount of culture medium (1:1), and centrifuge the tube 1400 rpm, Room temperature, 00:05:00
5m
After centrifugation aspirate the supernatant and resuspend the cells in cardiomyocytes culture medium. You can take asmall portion for cell counting and place desired amount of cells on a new geltrex-coated plate with addition of 10 micromolar (µM) rock inhibitor for better cell recovery.
Agitate the plate gently from side to side and forwards and backwards to ensure that the cell aggregates are evenly distributed across the surface of the well. Put the plate in the incubator. The next day in the morning change the medium to fresh culture medium and continue media change every 2-3 days.
Cryopreservation of cardiomyocytes
Note
if cardiomyocytes and their subtypes are not required for any ongoing experiment, they can be cryopreserved from day 12-15 onwards and recovered for culture when needed.
It is recommended to freeze around 2x10^6 cells per cryovial.
After centrifugation from step you can aspirate the supernatant and resuspend cells in cold cryopreservation medium.
Note
Cardiomyocytes cryopreservation medium:
50 % volume Fetal Bovine Serum, qualified, One Shot™ format, United StatesThermo FisherCatalog #A3160501 , 40 % volume RPMI 1640 Medium, GlutaMAX™ Supplement, HEPESThermo FisherCatalog #72400021 ,10 % volume dimethylsulfoxide (DMSO)Merck MilliporeSigma (Sigma-Aldrich)
Add an appropriate amount of cold cryopreservation medium to the tube, and resuspend the cells. Transfer cryotubes to a freezing container and put them in -80 °C freezer Overnight . The next day, you can transfer cryotubes to the liquid nitrogen for longer storage.
Post-thaw recovery
Prepare the previously coated 6-well plate by adding 2 mL of warm culture medium to each well and 2 µL from 10 millimolar (mM) stock solution ROCK inhibitor (Ri) Y-27632STEMCELL Technologies Inc.Catalog #72308 to minimize the cell death.
Take out the desired cryovial from the liquid nitrogen and quickly transfer it to 37 °C water bath. Be careful not to submerge the cap of the cryovial and keep a little ice cube in the cryovial before taking it out. Disinfect the cryovial with 70% ethanol.
Add 9 mL of warm cardiomyocyte culture medium to 15ml centrifuge tube. Centrifuge 1400 rpm, Room temperature, 00:05:00
5m
After centrifugation aspirate the supernatant and resuspend the cells in culture medium. You can take small portion for cell counting.
Note
It is recommended to seed around 1-3x10^6 cells per well in 6-well plate.
Transfer the cells to a new plate, agitate the plate gently from side to side and forwards and backwards to ensure that the cell aggregates are evenly distributed across the surface of the well. Put the plate in the incubator. Next day change the medium without rock inhibitor and continue media change every 2-3 days.
Protocol references
1. Lyra-Leite DM, Gutiérrez-Gutiérrez Ó, Wang M, Zhou Y, Cyganek L, Burridge PW. A review of protocols for human iPSC culture, cardiac differentiation, subtype-specification, maturation, and direct reprogramming. STAR Protoc. 2022 Aug 18;3(3):101560. doi: 10.1016/j.xpro.2022.101560. PMID: 36035804; PMCID: PMC9405110.
2.Kleinsorge M., Cyganek L. Subtype-directed differentiation of human iPSCs into atrial and ventricular cardiomyocytes. STAR Protoc. 2020;1 doi: 10.1016/j.xpro.2020.100026.
3.Ren J., Han P., Ma X., Farah E.N., Bloomekatz J., Zeng X.X.I., Zhang R., Swim M.M., Witty A.D., Knight H.G., et al. Canonical Wnt5b signaling directs Outlying Nkx2.5+ mesoderm into pacemaker cardiomyocytes. Dev. Cell. 2019;50:729–743.e5. doi: 10.1016/j.devcel.2019.07.014.
