Oct 01, 2021
Histological quantification of thickness of the mucosal and muscle layers of the rat stomach
- Madeleine R. Di Natale1,2,
- Lauren Patten2,
- Martin Stebbing1,2,
- John Furness1,2
- 1The University of Melbourne;
- 2The Florey Institute of Neuroscience and Mental Health

Protocol Citation: Madeleine R. Di Natale, Lauren Patten, Martin Stebbing, John Furness 2021. Histological quantification of thickness of the mucosal and muscle layers of the rat stomach. protocols.io https://dx.doi.org/10.17504/protocols.io.bybtpsnn
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it’s working
Created: September 17, 2021
Last Modified: October 01, 2021
Protocol Integer ID: 53331
Keywords: Masson's trichrome, rat, stomach, smooth muscle, mucosa, H&E, muscle layers of the rat stomach, rat stomach, rat stomach assist, relationships between the muscle layer, histological quantification of thickness, muscle layer, histological quantification, mucosal, microscopy technique, microscopy, thickness,
Abstract
The thickness, organization and relationships between the muscle layers of the rat stomach assist in understanding their function. Here we describe protocols for identifying, measuring and quantifying the muscle layers of the rat stomach using histological and microscopy techniques.
We would like to acknowledge Phenomics Australia Histopathology and Slide Scanning Service, University of Melbourne.
Guidelines
No specific guidelines for this protocol.
Materials
RA Lamb Dry Chemical Stains, Haematoxylin (Weigert A)Thermo FisherCatalog #LAMB-250-C RA Lamb Dry Chemical Stains, Haematoxylin (Weigert B)Thermo FisherCatalog #LAMB-260-C
HistoleneTrajan Scientific and MedicalCatalog #11031 Absolute Ethanol Mayers hematoxylin solution 70% Ethanol Magnesium SulfateP212121 Sodium bicarbonateMerck MilliporeSigma (Sigma-Aldrich)Catalog #S6014 Eosin Solution DPXMerck Millipore (EMD Millipore)Catalog #1.00579.0500 Fuchsin acidBio Basic Inc.Catalog #FB0469.SIZE.100g Ponceau XylidineMerck MilliporeSigma (Sigma-Aldrich)Catalog #22308 Phosphomolybdic Acid solutionMerck MilliporeSigma (Sigma-Aldrich)Catalog #HT153 Remel™ Light Green CounterstainThermo FisherCatalog #R40123 Acetic acid, glacialMerck MilliporeSigma (Sigma-Aldrich)Catalog #537020 XyleneBio Basic Inc.Catalog #XC9800.SIZE.1L
Protocol materials
Phosphomolybdic Acid solutionMerck MilliporeSigma (Sigma-Aldrich)Catalog #HT153
Acetic acid, glacialMerck MilliporeSigma (Sigma-Aldrich)Catalog #537020
XyleneBio Basic Inc.Catalog #XC9800.SIZE.1L
Eosin Solution
Ponceau XylidineMerck MilliporeSigma (Sigma-Aldrich)Catalog #22308
Remel™ Light Green CounterstainThermo FisherCatalog #R40123
HistoleneTrajan Scientific and MedicalCatalog #11031
Mayers hematoxylin solution
Magnesium SulfateP212121
Fuchsin acidBio Basic Inc.Catalog #FB0469.SIZE.100g
DPXMerck Millipore (EMD Millipore)Catalog #1.00579.0500
RA Lamb Dry Chemical Stains, Haematoxylin (Weigert A)Thermo FisherCatalog #LAMB-250-C
RA Lamb Dry Chemical Stains, Haematoxylin (Weigert B)Thermo FisherCatalog #LAMB-260-C
Absolute Ethanol
70% Ethanol
Sodium bicarbonateMerck MilliporeSigma (Sigma-Aldrich)Catalog #S6014
Acid Alcohol
Safety warnings
Before completing any steps in this protocol please refer to the each SDS/MSDS and follow local safety guidelines set by your institute and manufacturers.
Tissue Collection
Stomach tissue is collected from adult Sprague-Dawley male and female rats. Rats are supplied with food and water ad libitum prior to any tissue collection. All procedures were approved by The Florey Institute of Neuroscience and Mental Health Animal Ethics Committee.
Animals are deeply anesthetized with either an intraperitoneal injection of pentobarbital sodium (100mg/kg) or an intraperitoneal injection of a mixture of ketamine (50 mg/kg) and xylazine (10 mg/kg) prior to being perfused transcardially with phosphate buffered saline (PBS: 0.15 M NaCl, 0.01 M sodium phosphate buffer, pH 7.2) followed by fixative.
Different fixation methods are completed to allow for comparisons, these fixation methods are the following; 4% paraformaldehyde (Sigma Aldrich, USA), 10% neutral buffered formalin (Trajan, Melbourne, Australia) or Zamboni’s fixative (2 % formaldehyde plus 0.2 % picric acid in 0.1 M sodium phosphate buffer, pH 7.0). Some samples are placed into PBS containing nicardipine (1 µm) for 30-40 mins at room temperature before fixation.
Safety information
Be careful with fixatives. Refer to the each SDS/MSDS and follow local safety guidelines set by your institute and manufacturers.
Histological Staining
Place tissue into histology cassettes in desired orientation and dehydrate through graded ethanol to histolene before embedding samples in paraffin.
Cut sections (5 μm) on microtome, dry sections at 37.7 ºC and stain with haematoxylin and eosin (H&E).
This is completed using a Leica Autostainer XL and Leica CV5030 coverslipper
Haematoxylin and Eosin staining steps are as follows:
1. Histolene - 5 minutesHistoleneTrajan Scientific and MedicalCatalog #11031
2. Histolene - 1 minute HistoleneTrajan Scientific and MedicalCatalog #11031
3. Absolute Alcohol - 1 minute Absolute Ethanol
4. Absolute Alcohol - 1 minute Absolute Ethanol
5. 70% Alcohol - 1 minute 70% Ethanol
6. Wash in running tap water
7. Stain in Mayer’s Haematoxylin - 6 minutes Mayers hematoxylin solution
8. Wash in running tap water - 1 minute
9. Differentiate in 0.3% Acid alcohol - 1-2 dips Acid Alcohol
10. Blue in Scott’s tap water (magnesium sulfate buffered with sodium bicarbonate) - 1 minute Magnesium SulfateP212121 Sodium bicarbonateMerck MilliporeSigma (Sigma-Aldrich)Catalog #S6014
11. Wash in running tap water - 3 minute
12. Check microscopically
13. Counter stain with Eosin - 3 minute Eosin Solution
14. Rinse in running tap water - 1-2 dips
15. 70% Alcohol - 2 dips 70% Ethanol
16. Absolute Alcohol - 4 dips Absolute Ethanol
17. Absolute Alcohol - 1 minute Absolute Ethanol
18. Absolute Alcohol - 1 minute Absolute Ethanol
19. Histolene - 1-2 minutes HistoleneTrajan Scientific and MedicalCatalog #11031
20. Histolene - 1-2 minutes HistoleneTrajan Scientific and MedicalCatalog #11031
21. Histolene - 1-2 minutes HistoleneTrajan Scientific and MedicalCatalog #11031
22. Mount in DPX DPXMerck Millipore (EMD Millipore)Catalog #1.00579.0500
Results:
Nuclei-blue
Cytoplasm, muscle, collagen, erythrocytes-pink/red
Masson’s trichrome staining is conducted manually by submerging slides into the solutions described in the steps below.
Masson's trichrome staining steps are as follows:
1. Bring sections to water.
2. Place sections in Bouin's fixative - 60 minutes at 60°C
3. Wash in running water - 10 minutes
4. Iron Haematoxylin
4.1 Weigert's A (I part) - 2 minutesRA Lamb Dry Chemical Stains, Haematoxylin (Weigert A)Thermo FisherCatalog #LAMB-250-C
4.2 Weigert's B (I part) - 2 minutes RA Lamb Dry Chemical Stains, Haematoxylin (Weigert B)Thermo FisherCatalog #LAMB-260-C
5. Wash in water.
6. 1% Ponceau 2 R- (2 parts) + 1 % Acid Fuchsin acidBio Basic Inc.Catalog #FB0469.SIZE.100g - (1 Part) - 5 minutes Ponceau XylidineMerck MilliporeSigma (Sigma-Aldrich)Catalog #22308
7. Wash in water.
8. 1% Phosphomolybdic Acid - 3 minutes Phosphomolybdic Acid solutionMerck MilliporeSigma (Sigma-Aldrich)Catalog #HT153
9. Wash in water
10. 1%Remel™ Light Green CounterstainThermo FisherCatalog #R40123 n - 5 minutes
11. Wash and leave in 1% Acetic Acid - 1 minute Acetic acid, glacialMerck MilliporeSigma (Sigma-Aldrich)Catalog #537020
12. 100% ethanol Absolute Ethanol
13. Dehydrate, clear in xylene XyleneBio Basic Inc.Catalog #XC9800.SIZE.1L
14. Coverslip sections using DPX mounting media. DPXMerck Millipore (EMD Millipore)Catalog #1.00579.0500
Results:
Nuclei-blue/black
Cytoplasm, muscle and RBC-red
Collagen-blue/green
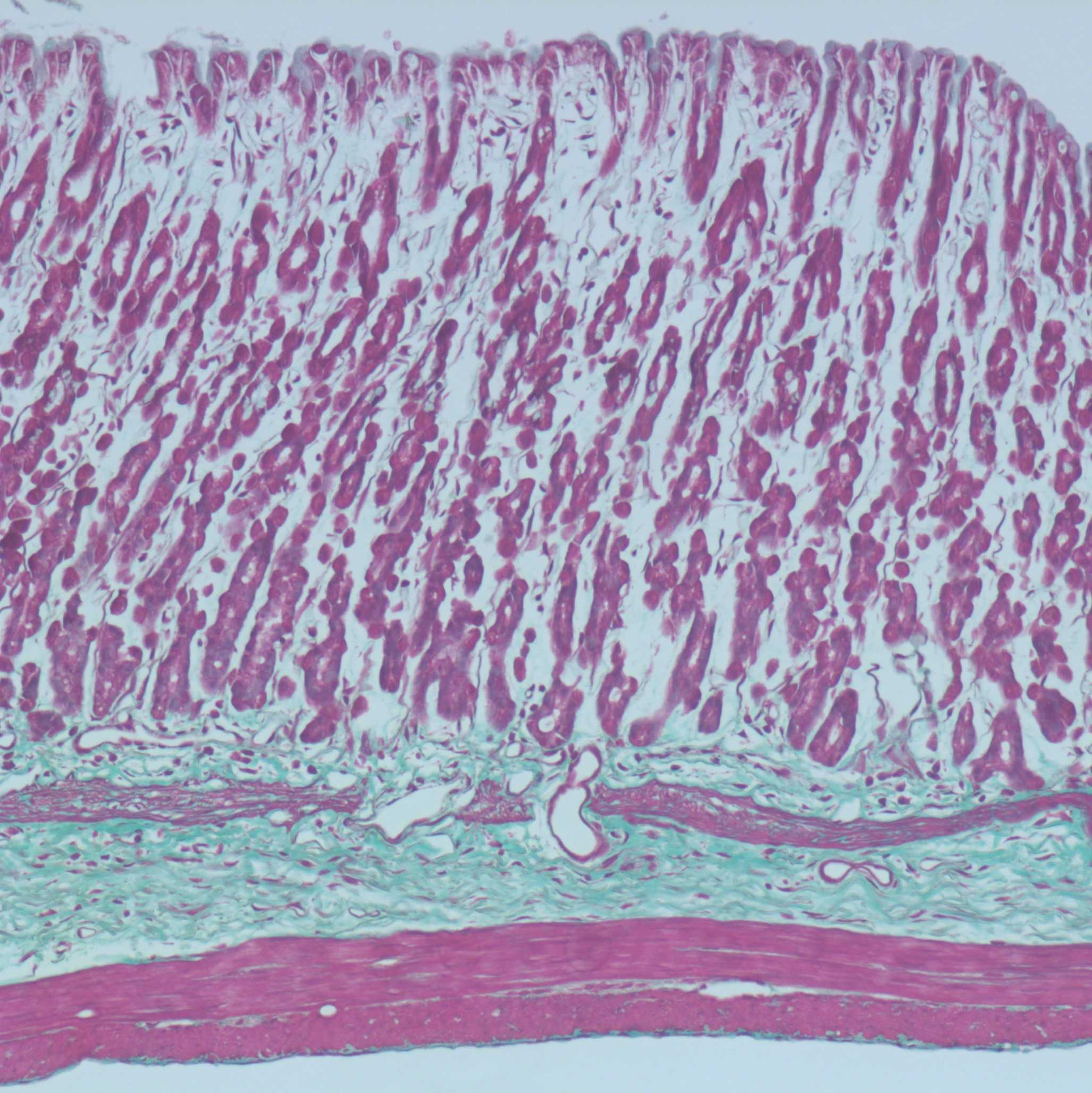
Histological Quantification
Examine and image slides using an Axioplan microscope (Zeiss, Sydney, Australia).
A selection of fiducial points are chosen as sites to measure the thickness of the mucosal and muscle layers in rat stomach.
At each point three measurements of each muscle layer (mucosa, muscularis mucosae, longitudinal muscle, circular muscle, oblique muscle (where applicable)) are made, the average of these measurements is calculated.
Average these measurements for all equivalent fiducial points measured.
