Jul 25, 2025
High throughput wild type measles quantification in settled solids using digital RT-PCR

- Alessandro Zulli1,
- Abby Paulos2,
- Bridgette Hughes1,3,
- Dorothea Duong1,3,
- Amanda Bidwell1,
- Peter Thana1,3,
- marlene.wolfe 2,
- Alexandria Boehm1
- 1Stanford University;
- 2Emory University;
- 3Verily Life Sciences

Protocol Citation: Alessandro Zulli, Abby Paulos, Bridgette Hughes, Dorothea Duong, Amanda Bidwell, Peter Thana, marlene.wolfe , Alexandria Boehm 2025. High throughput wild type measles quantification in settled solids using digital RT-PCR. protocols.io https://dx.doi.org/10.17504/protocols.io.j8nlky425g5r/v1
Manuscript citation:
Measles RNA detection in wastewater solids
Abigail P. Paulos, Alessandro Zulli, Bridgette Shelden, Dorothea Duong, Alexandria B. Boehm, Marlene K. Wolfe
medRxiv 2025.07.18.25331801; doi: https://doi.org/10.1101/2025.07.18.25331801
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
This protocol is in use by WWSCAN and public health departments across the United States
Created: July 25, 2025
Last Modified: July 25, 2025
Protocol Integer ID: 223249
Keywords: rna samples from solid wastewater sample, nucleic acid from sar, wastewater surveillance of sar, pcr inhibitor removal of settled solid, high throughput rna extraction, rna extraction, targeting bovine coronavirus vaccine, bovine coronavirus vaccine, extracted rna sample, pcr inhibitor removal, nucleic acid extraction, direct absolute quantification of the target rna molecule, samples before nucleic acid extraction, separate pcr reaction, pcr inhibition, target rna molecule, rna sample, solid wastewater sample, pmmov rna, triplex reverse transcriptase, wastewater surveillance, quantitative analysis of nucleic acid, bcov quantification, pcr, wastewater sample, rna, pepper mild mottle virus, enveloped virus, pcr workflow, pcr v3, targeting bcov, indicator of pcr inhibition, nucleic acid, ddpcr, droplet digital polymerase chain reaction, wastewater, high throughput sar, solids for sar, type measles virus rna in wastewater, high throughput wild type measles quantification, wild type measles quantification, type m
Disclaimer
DISCLAIMER – FOR INFORMATIONAL PURPOSES ONLY; USE AT YOUR OWN RISK
The protocol content here is for informational purposes only and does not constitute legal, medical, clinical, or safety advice, or otherwise; content added to protocols.io is not peer reviewed and may not have undergone a formal approval of any kind. Information presented in this protocol should not substitute for independent professional judgment, advice, diagnosis, or treatment. Any action you take or refrain from taking using or relying upon the information presented here is strictly at your own risk. You agree that neither the Company nor any of the authors, contributors, administrators, or anyone else associated with protocols.io, can be held responsible for your use of the information contained in or linked to this protocol or any of our Sites/Apps and Services.
Abstract
We developed and validated a hydrolysis probe-based RT-qPCR assay for specific detection of wild-type measles virus RNA in wastewater that eliminates cross-reactivity with vaccine strains. The wild type measles assay targets the 3′ region of the measles N gene with redesigned probe and reverse primer sequences optimized for circulating genotypes B3 and D8. In vitro validation demonstrated no cross-reactivity against 23 common respiratory pathogens or measles vaccine strains.
Attachments
Materials
Equipment
●Bio-Rad QX200 AutoDG
●Bio-Rad QX200 Droplet Reader
●Bio-Rad PX1 PCR Plate Sealer (Bio-Rad, Catalog #:1814000)
●Agilent Bravo
●Eppendorf Master Cycler ProThermal Cycler
●Bio-Rad C1000 Touch Thermal Cycler
●Rainin Single-Channel Pipettes L1000XLS+, L200XLS+, L20XLS+, L10XLS+, , L2XLS+
●Rainin Multi-Channel Pipettes L12-1000XLS+, L12-200XLS+, L12-20XLS+, L12-10XLS+, L8-1000XLS+, L8-200XLS+, L8-20XLS+, L8-10XLS+
●Axygen Plate Centrifuge
●Benchtop vortex
●96-well plate cold block for AutoDG droplet generator
●96-well plate cold block for ddPCR plates
●Zebra GX430t label printer
Reagents
●ddPCR™ One-Step RT supermix for Probes
●Reverse Transcriptase
●300mM DTT
●Automated Droplet Generation Oil for Probes
●Ambion Nuclease Free Water, 50mL
●ddPCR Droplet Reader Oil
Consumables
●Bio-Rad ddPCR™ 96-Well Plates
●Eppendorf twin.tec PCR Plate
●DG32™ Automated Droplet Generator Cartridges
●PCR PX1 Plate Sealer, foil, pierceable
●Pipet Tips for the AutoDG™
●Rainin low retention, pre-sterilized, filter tips 1000 µL, 200 µL, 20 µL
●Axygen Automation Tips, 20 µl, filtered, sterile tips (Axygen 20 µL tips)
●Reagent Reservoirs
●Automation Reservoirs
●Eppendorf Tubes, 1.5mL, 2mL and 5mL.
●2.5” x 0.5” Cryogenic Labels for the Zebra GX430t label printer
Samples and Test Materials
●Purified RNA from wastewater samples.

Protocol materials
One-Step RT-ddPCR Advanced Kit for ProbesBio-Rad LaboratoriesCatalog #186-4021
Troubleshooting
Background and assay design
The purpose of this document is to describe the usage of a hydrolysis probe-based assay that is specific and sensitive to wild type (WT) measles virus RNA in wastewater, that does not cross-react with the vaccine strain of the measles virus.
Roy et al. previously reported a measles RT-QPCR assay targeting the 3′ region of the measles N gene. We downloaded 97 (43 genotype B3 and 54 genotype D8) measles sequences from NCBI in March 2025 and obtained the Edmonston-Enders and Edmonston-Zagreb vaccine sequences. We then aligned the sequences of the Roy et al. primers and probe with the sequences. The in silico results suggested that the Roy et al. assay could be made specific to circulating WT measles genotypes B3 and D8, and not specific to the vaccine sequences, by changing the sequence of the probe and the reverse primer. The newly designed assay is shown in Table 1. Note that although the original Roy et al. assay used a locked nucleic-acid probe, the modified assay in Table 1 does not.
| A | B | |
| Forward Primer | AGGATGAGGCGGACCARTACTT | |
| Reverse Primer | CRATATCTGAGATTTCCTTGTTCTC | |
| Probe | CATGATGATCCAAGTAGTAGTGA |
Table 1. Wild type measles assay.
Preparation
Retrieve all kit components from the One-Step RT-ddPCR advanced kit for probes from the -20 °C freezer and thaw the components On ice .
One-Step RT-ddPCR Advanced Kit for ProbesBio-Rad LaboratoriesCatalog #186-4021
Preparation
For re-running frozen plates only:
Thaw the RNA storage plate On ice . Before analysis of thawed frozen samples, ensure that the plate is adequately sealed and briefly vortex and centrifuge the plate to ensure the samples are well mixed.
Keep extracted RNA samples On ice or in a cold block from the freezer at all times.
ddPCR master mix preparation (Wild type measles)
45m
Prepare Wild-type Measles Master Mix
Prepare a working stock of fresh nuclease free water by pouring from a 50 mL Ambion Nuclease Free water into a 5mL eppendorf tube.
Store Master Mix components On ice as much as possible during the preparation process.
Briefly vortex One-Step RT supermix for Probes (Yellow Tube), Reverse Transcriptase (Orange Tube), and DTT (Grey Tube) to mix contents then briefly spin with the benchtop centrifuge.
One-Step RT-ddPCR Advanced Kit for ProbesBio-Rad LaboratoriesCatalog #186-4021
In a 2mL eppendorf tube, prepare the master mix according to the table for measles quantification. Store prepared master mix on ice.
Note
The 20x wild type measles Primer/Probe Mix contains primers and probes in the following concentrations (suspended in molecular grade water):
| A | B | C | |
| 20x Concentration | Final Concentration/Rxn | ||
| Primers (each) | 18 uM | 900 nM | |
| Probe | 5 uM | 250 nM |
Reactions assume 22uL volume.
| A | B | C | |
| Reagents | Volume per Well | Volume Per Plate | |
| ddPCR™ One-Step RT supermix for Probes (Yellow Tube) | 5.5 µL | 580.8 µL | |
| 20x Measles Primer/Probe Mix | 1.1 µL | 348.48 µL | |
| Reverse Transcriptase (Orange Tube) | 2.2 µL | 232.32 µL | |
| 300mM DTT (Gray Tube) | 1.1 µL | 116.16 µL | |
| Nuclease Free Water | 6.6 µL | 464.64 µL | |
| Total Volume | 16.5 µL | 1742.4 µL |
Measles ddPCR Master Mix. Volume per plate assumes 96 well plate (with 10% excess).
Transfer Master Mix and Samples to ddPCR plate (both assays)
For each assay, remove a white cold block from the freezer and place a new Bio-Rad ddPCR 96-well Plate in it.
Note
Note that these steps may be completed with a liquid handling robot. We use the BRAVO system.
Using a new reagent reservoir, manually pipette 16.5 µL of the appropriate Master Mix to each well of each ddPCR Plate.
Transfer samples to the following wells on the plate either manually or using a liquid handling robot such as the Agilent Bravo system:
- Transfer 5.5 µL of samples from columns 1-10 of the RNA plate into columns 1-10 of the ddPCR plate.
- Transfer 5.5 µL of the extraction controls from column 12 of the RNA plate into column 11 of the ddPCR plate.
- Transfer 5.5 µL of NTC (water) into A-G of column 12.
- Add 5.5 µL of ddPCR positive control to well H12 of the ddPCR plate (add manually even if using a liquid handler)
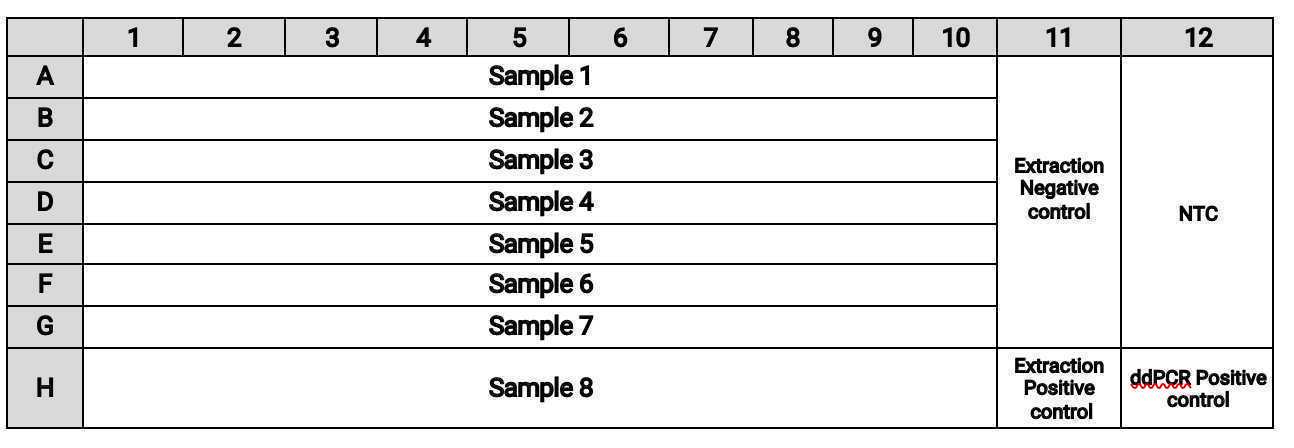
The plate layout should be:
Note
Note: The set up of this plate is slightly different from the set up of the 96 well extraction plate used in our companion protocol. Column 11 of this plate uses column 12 of the extraction plate. The lower right hand corner was purposely chosen for the placement of the positive controls.
Bring the BioRad ddPCR plate with Master Mix and samples to the Bio-Rad PX1 PCR Plate Sealer and place it in the metal carrier inside the plate sealer.
Note
Do not store the metal carrier inside the plate sealer, as it will become warm and warm the samples during sealing.
Align a PCR Plate Heat Seal Foil on top of the ddPCR plate with the red line facing up.
Seal the plate by pressing the green “seal” button.
Briefly vortex the plate using a bench top plate vortexer.
Briefly spin down the plate in the bench top Axygen Plate Centrifuge.
Store the ddPCR plate on a white cold block or On ice until droplet generation.
ddPCR droplet generation
45m
Generate Droplets:
On the AutoDG droplet generator: select “configure plate” and then press the blue arrow in the upper left corner to highlight all columns.
Load 3 DG32 cartridge plates and 2 tip boxes with the lid removed onto the AutoDG deck. The icons on the AutoDG display will change from yellow to red if the plates and tips are oriented correctly and to green if placed correctly.
Place the sealed, vortexed, and centrifuged ddPCR plate in the Sample Plate position on the AutoDG deck.
Remove the AutoDG cooling block from the freezer and place it in the Droplet Plate slot on the AutoDG deck.
For the Droplet Plate: label a new Bio-Rad ddPCR™ 96-Well Plate and place it in the black cooling block.
Click “Generate Droplets” to begin droplet generation. Droplet generation will take approximately 00:45:00 .
45m
When droplet generation is done, open the lid and remove the droplet plate from the black cold block and place it in the metal carrier inside the plate sealer.
Align a PCR Plate Heat Seal Foil on top of the droplet plate with the red line facing up.

Photograph showing proper setup of samples and consumables in the AutoDG. After droplet generation, samples have been emulsified and deposited into the "Droplet Plate," which is ready for thermocycling.
Seal the plate by pressing the green “seal” button.
Note
NOTE: Do NOT vortex plate at this point!
Thermocycling (Wild type measles)
45m
Place the plate in the thermocycler. Verify and run the thermocycler program according to the table for wild type measles ddPCR.
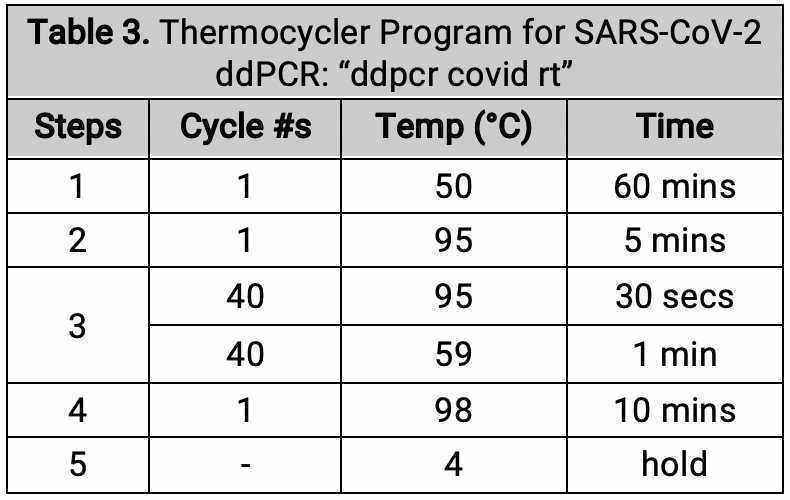
Cycling conditions for the wild type measles RNA target assay
After the PCR program is done, store the ddPCR plate at 4 °C (in the thermal cycler, on ice or in the fridge) until droplet reading.
Read Droplets
Before running the plate on the QX200 droplet reader, open the drawer on the left side of the instrument and check that there is adequate droplet reader oil and that the waste bottle is not overly full.
Load template from previous plate of the same assay - ensure that all parameters (dye, absorbance etc.) are correct for the assay used and that sample names are assigned to the appropriate wells. Save as with a new plate name.
Name the file with a name clearly describing which plate you are reading.
Remove the plate from the thermal cycler and secure it in the droplet reader by placing it in the stage, placing the metal brace on top of it and pressing down the black plastic tabs on either side of the brace.
Click “Run” in Quantasoft to commence droplet reading.
Select “Ok” when the next dialog box comes up. Do not change the settings from “Columns” and “FAM/HEX”.
Measles Post-Processing Analysis
Measles assay analysis

Example of measles assay results. Wild-type detection shown on the left, vaccine assay exclusion shown on the right.
Open QuantaSoft™ Analysis Pro Software
Open the .qlp file associated with the run
On the Plate Editor tab, select all the wells then in the dropdown menu for Assay Information select Simplex/Duplex and label Signal Ch1 (FAM) as "Wild-type measles"
At this point, if sample names were not added at the start of the run, fill in the sample name by selecting the wells, typing in sample ID and click “Apply”.
If wells need to be deselected: Go to the Plate Editor tab, select wells to exclude and click “Clear Selected Well”.
.On the 1D Amplitude tab, make sure that all wells are highlighted and select the Threshold Multiple Wells button to automatically set the threshold between the positive and negative populations
Suggested threshold: 1400
Go through every well to ensure that:
- The droplets are designated properly per well
- There are >10,000 droplets per well
If any well does not have the appropriate droplets, go to the Plate Editor tab, select the affected well and click “Clear Selected Well”.
On the 1D Amplitude tab, select Merged Wells on the left side of the page
Click on the Table Menu Button on the right side of the Well Data table and select Export to CSV to export data.
Save the QuantaSoft Analysis
Dimensional Analysis and Quality Control
For dimensional analysis to express the results of each assay in terms of gene copies/dry weight solids:
Begin with the concentration provided by the QuantaSoft software and reported in the CSV, as expressed in gc/uL of reaction.
Note
This concentration is expressed in terms of the total volume of the merged wells, rather than the volume of the template added. Determination of gc/g relies on the ratio of of template in the reaction and mass in the volume eluted from extraction, so when beginning with this value expressed in terms of gc/uL the number of wells merged is not considered in the dimensional analysis.
To determine the concentration in cp/g dry weight:
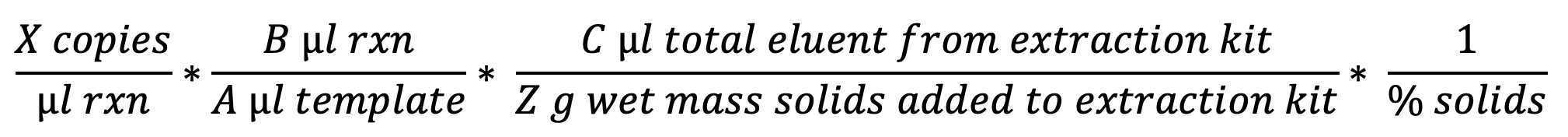
Note
When using the assays described in the three protocols that make up this process as written:
- B = 20 µL total reaction volume
- A = 5 µL volume template in reaction
- C = 60 µL elution volume from each well in extraction kit
- Z = 0.0225 g wet weight of solids added to each extraction well (300 µl of a suspension of 75 mg wet weight solids / ml of solution)
- Percent solids varies for each sample on each day (typical range 0.05 - 0.3, i.e. 5-30%)
Please note that there are many different ways to conceptualize the dimensional analysis. Please be sure to double check your own calculations to make sure they match with the measurement units from your instrument. For example, our instrument reports results as copies per the entire volume of the rxn, but some instruments may report a different concentration.
Links to two companion protocols to this protocol can be found in the description of this protocol.

