Jun 08, 2025
Grimelius’ argyrophil silver stain (diffuse endocrinocytes)
- Egor Kuzmin1
- 1Sechenov University

Protocol Citation: Egor Kuzmin 2025. Grimelius’ argyrophil silver stain (diffuse endocrinocytes). protocols.io https://dx.doi.org/10.17504/protocols.io.j8nlkrqb1v5r/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: May 25, 2025
Last Modified: June 08, 2025
Protocol Integer ID: 218897
Keywords: Silver, Grimelius, Argyrophilic, Diffuse endocrinocytes, Endocrine cell, Enteroendocrinocytes, Argyrophil , argyrophilic grimelius silver stain, granules of diffuse endocrine cell, silver stain, high silver concentrations without additional reduction, low silver concentration, use of silver salt, diffuse endocrine cell, utilizing high silver concentration, endocrine cell, employing low silver concentration, low silver nitrate concentration, silver salt, diffuse endocrinocyte, use of aqueous silver, argyrophilic reaction, aqueous silver, classical histochemical approach, principal types of silver, staining methodology, cell, sodium sulfite mixture, insulin, chemical reduction, silver, argyrophil silver stain, endocrine
Abstract
The classical histochemical approach for visualizing granules of diffuse endocrine cells involves the use of silver salts at varying concentrations. This distinguishes two principal types of silver-based reactions: argyrophilic reactions (employing low silver concentrations followed by chemical reduction) and argentaffin reactions (utilizing high silver concentrations without additional reduction). The gold standard for identifying these endocrine cells is the argyrophilic Grimelius silver stain, which enables the identification of the broadest spectrum of endocrine cells except for cholecystokinin-producing (I-cells), insulin-producing (β-cells), somatostatin-producing (D-cells), and PYY-cells. The Grimelius staining methodology entails the use of aqueous silver nitrate solutions followed by reduction with a hydroquinone-sodium sulfite mixture. Staining is performed at elevated temperature using either a water bath or microwave oven. This method is characterized by high reproducibility when the protocol is strictly followed, reagent economy through the use of low silver nitrate concentrations and a long-lasting reducing agent, and superior result quality yielding uniform, photostable staining without precipitate. It may require multiple staining cycles to controllably intensify the reaction and performs robustly even on very old specimens, facilitating studies with archival paraffin blocks.
Guidelines
- Use only scrupulously clean glassware. All contact with metal must be excluded.
- Use distilled or deionized water.
- The 2% silver nitrate solution may be stored refrigerated in amber glassware and remains usable for several weeks.
- Perform the staining procedure under safelight conditions. Store slides in light-proof storage boxes.
Materials
- Silver nitrate; AgNO₃
- Hydroquinone; 1,4-Dihydroxybenzene; C6H4(OH)2
- Anhydrous sodium sulfate; Na2SO4
- Sodium thiosulfate; Na2S2O3
- Ortho-xylene; C6H4(CH3)2
- Ethanol 100% or isopropanol 100%
- Distilled water
Troubleshooting
Safety warnings
- Formalin is a volatile compound classified as carcinogenic.
- Xylene, isopropyl alcohol (IPA), and ethyl alcohol are volatile substances hazardous to human health, especially following long-term exposure. Procedures utilizing these solvents require a ventilated fume hood and the use of medical gloves (e.g., nitrile).
- Silver nitrate is a powerful oxidizing agent that leaves black stains on the skin and is a suspected carcinogen. Both crystalline dust and aqueous solutions can cause damage to mucous membranes and skin. When handling silver compounds, personal protective equipment must be worn, and work must be conducted in a certified chemical fume hood.
Before start
Autopsy specimens were fixed by immersion in neutral buffered formalin for 48 hours. Processing involved a standard protocol utilizing a graded series of isopropyl alcohols, followed by paraffin embedding. Histological blocks were cut on a microtome to yield 3 µm sections. Sections were placed on silane-coated adhesive glass slides for secure adhesion. All staining reagents were freshly prepared immediately before staining commenced.
Introduction
Fig. 1. A light micrograph of a raccoon duodenum specimen stained with silver nitrate via the Grimelius method. The mucocytes of the large Brunner’s glands are visible, with their excretory ducts opening into the base of the intestinal crypt. At the transition zone between the crypt and the Brunner’s gland duct, endocrine cells exhibiting argyrophilic cytoplasm are observed.
Preparation of solutions
Stable silver solution
2% silver nitrate (AgNO₃ 2g/100ml) in distilled water or buffer.
Filter. Store in a dark refrigerator.
Working staining solution
2 mL stable silver solution per 100 mL distilled water.
Prepare fresh before use. Pre-warm.
Reducing Solution
Mix
1% hydroquinone
5% anhydrous sodium sulfate in distilled water.
Prepare fresh before use. Pre-warm.
Differentiating Solution (booking of the silver reaction)
5% sodium thiosulfate in distilled water
Protocol
1h 34m
STARTING POINT:
Dewaxing, rehydration, rinsing in distilled water. Rinse in pre-warmed distilled water
- ortho-xylene 5 min
- ortho-xylene 5 min
- absolute ethanol or isopropanol 5 min
- 50% ethanol or isopropanol 5 min
- distilled water 5 min
25m
FIRST CYCLE OF SILVER STAINING:
Incubation in AgNO₃ working staining solution for 2 minutes at 70 °C
The solution requires pre-warming followed by maintenance at a constant temperature. During slide transfer, contact with metallic instruments must be precluded. To achieve this, the tips (branches) of the forceps can be pre-treated with a paraffin coating; this creates a protective barrier against reduction of the silver.
2m
Rapid rinse in 70 °C distilled water
Wash with vigorous agitation but briefly using a single portion of pure distilled water.
30s
Incubation in hydroquinone-sodium sulfate reducing solution for 1 minute at 70 °C
Avoid excessive incubation time at this step to preclude background staining.
1m
Thorough rinsing in multiple changes of 70 °C distilled water
Rinse thoroughly in several aliquots of water to arrest silver reduction and prevent background development. The water becomes turbid rapidly at this stage; therefore, when staining large batches of sections, monitor water clarity and replace it at regular intervals.
1m
SECOND CYCLE OF SILVER STAINING:
Incubation in AgNO₃ working staining solution for minutes 1 at 70 °C
The solution requires pre-warming followed by maintenance at a constant temperature. During slide transfer, contact with metallic instruments must be precluded. To achieve this, the tips (branches) of the forceps can be pre-treated with a paraffin coating; this creates a protective barrier against reduction of the silver.
1m
Rapid rinse in70 °C distilled water
Wash with vigorous agitation but briefly using a single portion of pure distilled water.
30s
Incubation in hydroquinone-sodium sulfate reducing solution for 1 minute at 70 °C
Avoid excessive incubation time at this step to preclude background staining.
1m
Thorough rinsing in multiple changes of 70 °C distilled water
Rinse thoroughly in several aliquots of water to arrest silver reduction and prevent background development. The water becomes turbid rapidly at this stage; therefore, when staining large batches of sections, monitor water clarity and replace it at regular intervals.
There are only two cycles in the original protocol.
In special cases, the number of cycles can be increased.
1m
BOOKING OF THE SILVER REACTION:
Cool to room temperature.
Rinse using room-temperature distilled water.
30m
Rinse in 5% sodium thiosulfate differentiating solution.
This step arrests silver reduction, thus enhancing the long-term preservation of the specimen and preventing fading of the reaction products.
1m
FINAL STEP:
Counterstain with nuclear dyes (e.g., Carazzi’s hematoxylin for 5 minutes, differentiate in acidified water, blue in ammonia water).
A wide range of counterstains is suitable, including Fast Green, Neutral Red, Toluidine Blue, and hematoxylin. When using hematoxylin, differentiation steps in both acidic and alkaline media may be employed to achieve optimal results.
5m
Rinse, dehydrate, mount in resin medium.
- distilled water 5 min
- 50% ethanol or isopropanol 5 min
- absolute ethanol or isopropanol 5 min
- ortho-xylene 5 min.
- ortho-xylene 5 min.
- mount
25m
Result
Аrgyrophilic structures (e.g., endocrine cells of the gastrointestinal tract, pancreas, thyroid, pituitary) except for cholecystokinin-producing (I-cells), insulin-producing (β-cells), somatostatin-producing (D-cells), and PYY-cells.
Expected result
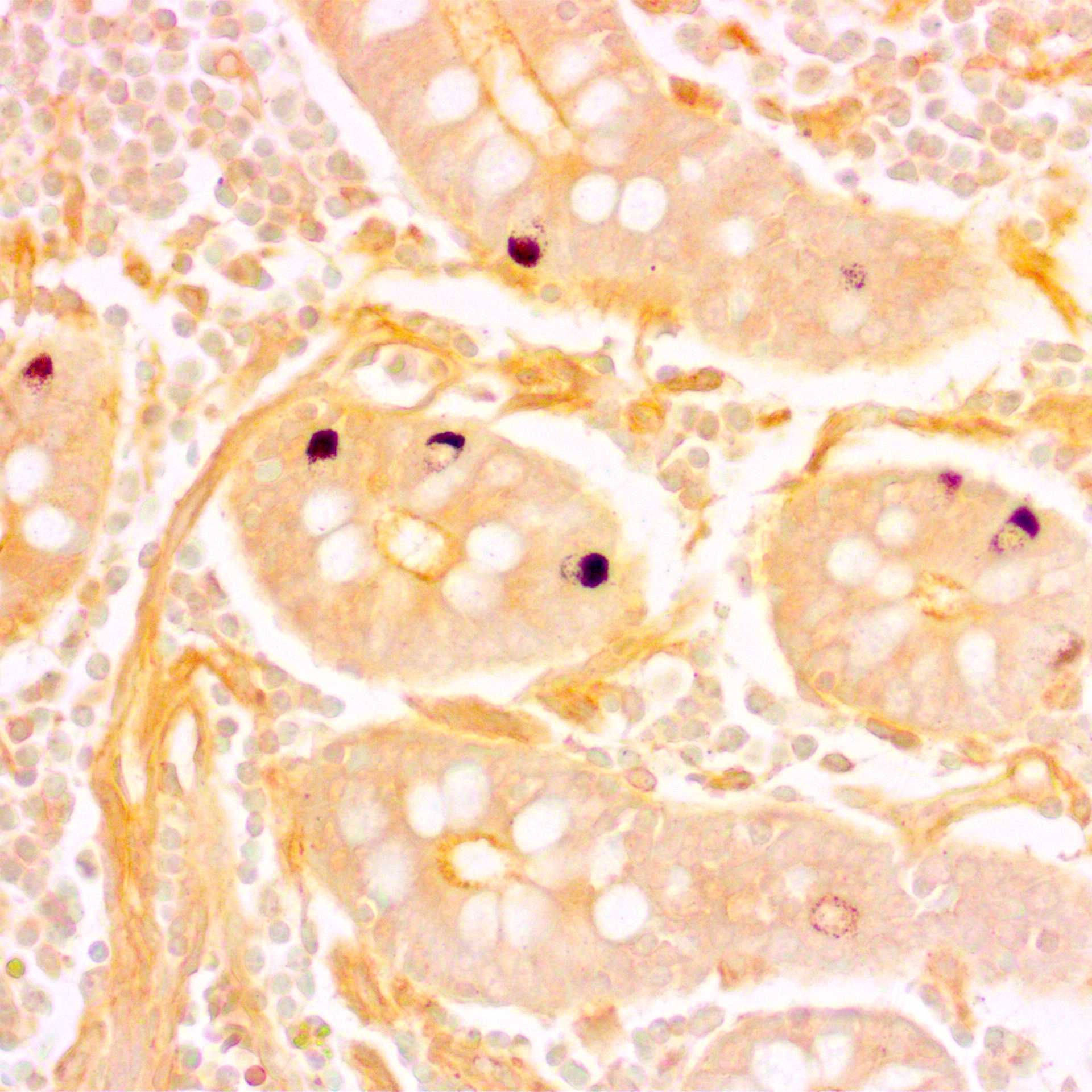
Fig. 2. Microphotographs of bovine duodenal sections stained with the Grimelius silver stain and counterstained with Carazzi's hematoxylin. Enteroendocrine cells are identified at the crypt base; their cytoplasm is stained brown, and their nuclei are stained blue
Fig. 3. Microphotographs of histological preparations of the European souslik (Spermophilus citellus) duodenum stained by the Grimelius silver method and by the combined Grimelius silver–Carazzi's hematoxylin method. Argyrophilic cells, characterized as enteroendocrine cells, are identified at all levels of the mucosa (villi, crypts, and Brunner's glands).
Note
Increasing the number of silver impregnation-reduction cycles or extending incubation times may enhance results. The method is stable, reproducible, and suitable for aged tissue blocks.
Historical note
Historically, the first histochemical methods for detecting diffuse endocrine cells were argyrophil techniques based on the capacity of specific cellular components to reduce silver nitrate to metallic silver. The pioneer was Masson's method (1914), which relied on direct silver reduction in a highly ionized solution. This technique demonstrated specificity only for certain subpopulations (EC cells) rather than all endocrine cells and served as the foundation for modifications by Hamperl (1952), Singh (1964), and Portela-Gomes (1986). A pivotal advancement came with the adoption of argyrophil methods using the reducing agent protargol, initially demonstrated on neural tissue by Bartelmez and Hoerr (1933) and later by Silver (1942). Bodian (1936) subsequently refined and accelerated this protocol.
The most prominent methods specifically developed for detecting argyrophilic endocrine cells were similar techniques based on silver nitrate reduction, described by Hellerström and Hellman (1960), Grimelius (1968), and Pascual (1967). Singh's method (1964) using 1% silver nitrate and Grimelius' method (1968) using 0.03% silver nitrate represent two fundamental modifications of Bodian's (1936) technique. Both replaced Bodian's less stable, shorter-lived protargol (which forms silver proteinate) with a hydroquinone-sodium sulfite mixture as the reducing agent. Pascual's later method (1976) employed 0.5% silver nitrate for extended incubation periods. Churukian's 1979 modification incorporated a pH 4.2 acetate buffer in the 0.5% silver nitrate solutions, reducing background staining and implementing a rapid protocol.
Sevier and Munger's stain (1965) stands apart from the aforementioned methods. It uniquely combines a concentrated 10% silver solution (characteristic of argentaffin techniques) with an external reducing agent (characteristic of argyrophil methods). This hybrid method has demonstrated specificity for EC-like cells (intestine), D1-cells (pancreas), and C-cells (thyroid gland).
Protocol references
1. Grimelius, L. (2004). Silver stains demonstrating neuroendocrine cells. Biotechnic & Histochemistry, 79(1), 37–44. doi:10.1080/10520290410001715264
2. Sabatino, D., Chiaravalli, A.M. (2022). ≠. In: La Rosa, S., Uccella, S. (eds) Endocrine Pathology. Encyclopedia of Pathology. Springer, Cham. doi:10.1007/978-3-030-62345-6_5082
3. Churukian, C. J., & Schenk, E. A. (1979). A Modification of Pascual’s Argyrophil Method. Journal of Histotechnology, 2(3), 102–103. doi:10.1179/his.1979.2.3.102
4. Pascual, J. S. F. (1976). A New Method for Easy Demonstration of Argyrophil Cells. Stain Technology, 51(4), 231–235. doi:10.3109/10520297609116708
5. Singh, I.(1964). A new argyrophile method for the rapid staining of enterochromaffin cells in paraffin sections. Cells tissues organs, 59(3), 290–296. doi:10.1159/000142623
