Dec 11, 2025
GeoMx RNA slide prep for fresh frozen human post-mortem brain tissue
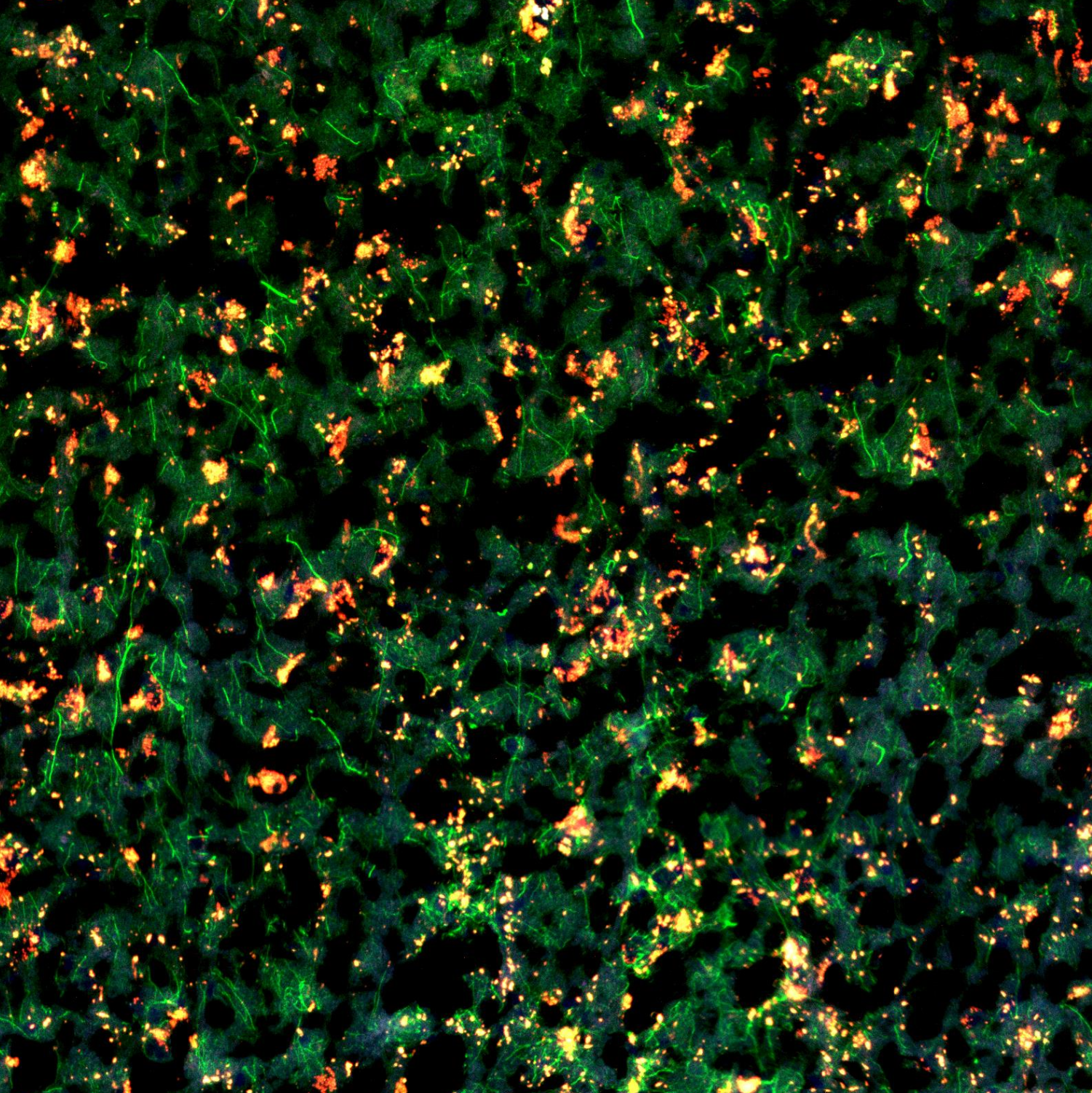
- Toby Curless1,2,
- Hemanth Ramesh Nelvagal1,2,
- Zane Jaunmuktane1,2
- 1Department of Clinical and Movement Neurosciences, UCL Institute of Neurology, Queen Square, London WC1N 3BG, UK;
- 2Aligning Science Across Parkinson’s (ASAP) Collaborative Research Network, Chevy Chase, MD, 20 815, USA

Protocol Citation: Toby Curless, Hemanth Ramesh Nelvagal, Zane Jaunmuktane 2025. GeoMx RNA slide prep for fresh frozen human post-mortem brain tissue. protocols.io https://dx.doi.org/10.17504/protocols.io.6qpvr8q1blmk/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: August 20, 2024
Last Modified: December 12, 2025
Protocol Integer ID: 105982
Keywords: ASAPCRN, spatial transcriptomics, RNA, fresh frozen, human brain , geomx rna slide prep, geomx manual slide preparation manual for rna, fresh frozen post mortem human brain tissue sample, geomx, human brain tissue sample, rna, fresh frozen post mortem, manual slide preparation manual
Funders Acknowledgements:
Aligning Science Across Parkinson’s through the Michael J. Fox Foundation for Parkinson’s Research (MJFF)
Grant ID: ASAP-000478
Abstract
This protocol largely follows the GeoMx manual slide preparation manual for RNA, with a few minor changes. This protocol also includes concentrations of antibodies, optimised on 10 um fresh frozen post mortem human brain tissue samples.
Guidelines
Cross-contamination of RNA should be avoided at all points. Regularly clean surfaces with RNase AWAY
Materials
REAGENTS
Leica tissue freezing medium
ChemCruz – Paraformaldehyde solution 4% in PBS sc-281692 – Santa Cruz Biotechnology
Gibco – PBS pH 7.4 – 70011-036
VWR – Formamide, deionized ULTRA PURE – CAS 75-12-7
Invitrogen Alexa FluorTM 488 goat anti-chicken IgY (H+L) – REF – A11039
Invitrogen Alexa FluorTM 647 goat anti-rabbit IgG (H+L) – REF – A21244
ATLAS ANTIBODIES – HPA014518 Anti-P2RY12 produced in Rabbit
Novus NBP1-05198 – Chicken anti-GFAP pAb
Invitrogen SYTOTM 83 orange fluorescent nucleic acid stain – REF – S11364
Invitrogen IHC Antigen Retrieval Solution 10X High pH – REF – 00-4956-58
ThermoFisher Scientific RNase AWAY – catalog # 7002
SIGMA – Gycine – G7126-100G
Promega – Tris base, molecular biology grade – REF – H5133
Sigma-Aldrich SSC Buffer 20x Concentrate – AM9763
Sigma-Aldrich Formalin solution, neutral buffered, 10% HT501128-4L
Invitrogen Proteinase K – REF – AM2548
EQUIPMENT
Morphy Richards – compact intellisteam 6L food steamer
ACD – HybEZTM II Oven
Labnet international, inc. – MINI INCUBATOR
KIMTECH – 7558 2 ply wipes
BIO-RAD C1000 TouchTM Thermal Cycler
SciSpin Mini – centrifuge
Eppendorf Easypet 3
Cleaver scientific Stirring water bath
Grace Bio-Labs HybriSlips
Coplin jars – any manufacturer
Pipettes – any manufacturer
Superfrost charged slides –
Leica CM1950 cryostat
Tweezers - any manufacturer
Paintbrushes – any manufacturer
Serological pipettes - any manufacturer
GeoMx DSP - NanoString
Troubleshooting
Safety warnings
Some chemicals used in this protocol are potentially harmful. Ensure that you read all safety instructions
Ethics statement
The protocols.io team notes that research involving animals and humans must be conducted according to internationally-accepted standards and should always have prior approval from an Institutional Ethics Committee or Board.
Prepare reagents
Prepare reagents for GeoMx pipeline prior to tissue preparation
| A | B | C | |
| Reagent | Dilution | Storage | |
| 70 % EtOH | Prepare 100 mL of 70 % ethanol by adding 30 mL of DEPC-treated water to 70 mL of 100 % ethanol. Change at least weekly | ||
| 50 % EtOH | Prepare 100 mL of 50 % ethanol by adding 50 mL of DEPC- treated water to 50 mL of 100% ethanol. Change at least weekly. | RT | |
| 1X PBS pH 7.4 | Prepare 1 L of 1X PBS by combining 100 mL of 10X PBS and 900 mL of DEPC-treated water. Don't reuse. | 4°C | |
| 10% neutral buffered formalin (NBF) | Caution: NBF is hazardous, handle with care and minimize inhalation risks. Work with 10% NBF in the fume hood. | RT | |
| Proteinase K | Prepare 1 µg/mL by adding 10 μL of 20 mg/mL Proteinase K to 200 mL of 1X PBS made with DEPC-treated water Note: Should be prepared fresh for each run and not reused. Inaccurate concentration of Proteinase K will affect assay performance. | RT | |
| NBF stop buffer | Prepare NBF stop buffer by adding 12.12 g Tris base and 7.5 g Glycine to 1 L DEPC- treated water. Do not reuse. | RT | |
| 2X SSC | Prepare 1 L of 2X SSC by combining 100 mL of 20X SSC and 900 mL of DEPC-treated water. Do not reuse. | RT | |
| 4X SSC | Prepare 1 L of 4X SSC by combining 200 mL of 20X SSC and 800 mL of DEPC-treated water. Do not reuse. | RT |
Prepare Tissue Sections
1d
Remove frozen human brain tissue from -80 °C freezer
In a biosafety cabinet, on dry ice, chip a 1 cm2 piece of tissue from the middle frontal gyrus, extending from the white matter to the superficial cortex using a scalpel and tweezers
Decontaminate Leica CM1950 cryostat (and tweezers, paintbrushes (any accessories used to manipulate tissue sections)) using 70 % ethanol, RNase away and a UV decontamination cycle
Set chamber and specimen head temperatures on cryostat to -20 °C
Transport tissue chips on 0 °C to cryostat.
Allow tissue chips to equilibrate within cryostat chamber for 00:30:00
30m
Insert fresh razor blade into blade holder and secure with the blade clamping lever. Cover blade with safety guard until ready to section
Place cryostat specimen disc on the freeze shelf and cover the disc with tissue freezing medium.
Place tissue chip onto tissue freezing medium in the desired orientation (coronal with all cortical layers and white matter facing upwards)
Move disc to Peltier position on freeze shelf and activate the Peltier element (rapid freezing)
Allow tissue freezing medium to completely freeze before placing on the specimen head
Move the locking lever to orient the specimen so that the face of the block is parallel to the blade
Using a marker pen, mark out the GeoMx scan area on the back of Superfrost Plus charged slides
Set cutting thickness to 10 μm
Trim into the tissue until the whole face of the block is present in each section (trimming in is also necessary for optimal RNA quality)
Using the anti-roll plate, generate a section free of wrinkles/creases. Paintbrushes can be used to gently flatten out tissue
Holding Superfrost Plus slide by the label, align the tissue section within the scan area (if using more than one section per slide, collect sections from the bottom of the slide, up towards the label)
Once aligned with scan area, lower slide onto tissue section, taking care not to press the section into the stage, place a finger on the back of the slide, underneath the section to ensure it adheres fully to the slide
Place slide (tissue facing upwards) onto freeze shelf
Remove specimen from specimen head and return to -80 °C freezer before collecting more sections
Thoroughly clean cryostat chamber and stage with ethanol before mounting a new sample. Repeat the process until all samples have been collected
Expose RNA targets
16m
Remove slides from cryostat freeze shelf and submerge fully in RT 4 % PFA for 00:10:00
10m
Wash slides 3x in 1X PBS (00:02:00 per wash)
2m
Tissue dehydration
58m
Turn on steamer and heat 1 staining jar of DEPC H2O and 1 staining jar of 1X Tris EDTA to 100 °C (use 100 mL staining jars with transferrable slide racks for these steps to minimise time with steamer lid removed)
2m
Transfer slides to a slide rack and bake sections on slides in a 60 °C drying oven for 00:30:00
30m
Spray 70 % ethanol onto a kimwipe and clean the marker pen from the back of the slides
1m
Wash slides in 50 % ethanol for 00:05:00
5m
Wash slides in 70 % ethanol for 00:05:00
5m
Wash slides 2x in 100 % ethanol for 00:05:00 each wash
5m
Let slides air dry for 00:05:00 – during this time, transfer slides to slide rack that can fit inside staining jar
5m
Perform target retrieval
25m
Remove steamer lid and submerge slides in DEPC H2O for 00:00:10 , then immediately transfer to 1X Tris EDTA for 00:20:00
20m 10s
NOTE - Antigen retrieval times may need optimisation – for 10 μm fresh frozen human brain samples, 20 minutes provided optimal results
When timer is finished, remove rack from steamer and immediately wash slides in room temperature 1X PBS for 00:05:00 . Slides can be stored in 1X PBS for 1 hour
5m
Heat water bath to 37 °C
Expose RNA targets
21m
Prepare proteinase K dilution – 0.1 μg/mL by adding 1 μL of 20 mg/mL Proteinase K to 200 mL of 1X PBS – place into a staining jar and add to water bath
1m
NOTE – Proteinase K concentration may need optimisation for different tissue types and thicknesses – for 10 μm fresh frozen human brain samples, 0.1 μg/mL provided optimal results
Once Proteinase K has reached 37 °C , incubate slides in Proteinase K for 00:15:00 (or as optimised for other tissues/thicknesses)
15m
During Proteinase K digest, ensure reagents for next steps are prepared
Wash slides in RT 1X PBS for 00:05:00 , proceed to next steps immediately
5m
Postfix—Preserve tissue morphology for soft tissues
20m
Post-fix the tissue by performing the following washes:
10 % NBF (00:05:00 )
5m
NBF Stop Buffer 2x 00:05:00 each wash
5m
1X PBS (00:05:00 ) – slides can be stored in 1X PBS for 01:00:00 at Room temperature or 06:00:00 at 4 °C
7h 5m
In-situ hybridisation (Overnight)
16h
Prepare hybridisation chamber – clean with RNase AWAY, wipe with Kimwipes. Add fresh Kimwipes to the chamber and dampen with DEPC treated water – make sure that the water does not pool
Prepare buffers for in-situ hybridisation – warm Buffer R to Room temperature before opening and thaw RNA detection probes, mix thoroughly by pipetting
Make the hybridisation solution according to the following table (n = number of slides NOT number of sections)
Remove slides from PBS one at a time, wipe away excess liquid with a fresh Kimwipe and place in hybridisation chamber.
Add 200 µL hybridisation solution to each slide, ensuring no bubbles are introduced. It is preferable to remove some hybridisation solution, than have bubbles present
Gently apply a Grace Bio-Labs HybriSlip by setting one edge of the coverslip down on the slide and gradually laying the rest of the coverslip down to avoid bubbles
Once all HybriSlips have been applied, clamp slides into place, close the lid of the hybridisation chamber and insert into hybridisation oven. Incubate at 37 °C Overnight (16-24 hours)
1d
Before removing slides from hybridisation oven, ensure the water bath is heated to 37 °C . Warm 100% formamide to Room temperature before opening. Once at room temperature, prepare Stringent Wash by mixing equal parts 100 % formamide with 4X SSC. Fill 2 staining jars with Stringent Wash and heat to 37 °C in the water bath
Once Stringent Wash reaches 37 °C , remove slides from hybridisation oven and dip into 2X SSC, allowing the HybriSlips to slide off, proceed to Stringent Wash after 00:05:00
5m
Wash slides 2X in Stringent Wash (00:25:00 each wash)
25m
Remove slides from Stringent Wash and wash 2X in Room temperature 2X SSC (00:02:00 each wash). Slides can be stored in 2X SSC for up to 01:00:00
1h 2m
Adding morphology markers
2h 15m
Prepare humidity chamber – clean with RNase AWAY. Line chamber with Kimwipes wetted with DEPC treated water
2m
NOTE – Morphology marker preparation will vary and should be empirically tested. Here we used unconjugated antibodies and thus modified the NanoString GeoMx protocol accordingly
Remove slides one at a time from 2X SSC, remove excess liquid with a Kimwipe and lay flat in humidity chamber
1m
Block sections in 200 µL Buffer W for 00:30:00 with the lid closed
30m
Prepare morphology marker solution as per the table below
| A | B | C | |
| Reagent | Dilution | Volume | |
| Buffer W | - | (n) x 220 µl | |
| GFAP | 1:1000 | (n) x 0.22 µl | |
| P2RY12 | 1:100 | (n) x 2.2 µl |
Morphology marker solution (n) indicates number of slides
Remove buffer W from slides by tapping on a Kimwipe, place back in humidity chamber and cover with 200 µL morphology marker solution. Close the lid and incubate in humidity chamber for 01:00:00
1h
During incubation, prepare secondary antibody mixture as per the table below
| A | B | C | |
| Reagent | Dilution | Volume | |
| Buffer W | - | (n) x 220 µl | |
| AF488 | 1:1000 | (n) x 0.22 µl | |
| AF647 | 1:500 | (n) x 0.44 µl |
Wash slides 2x in 2X SSC
2m
Return slides to humidity chamber, cover with 200 µL secondary antibody mixture and incubate for 00:20:00
20m
During incubation, prepare nuclear stain as per the table below
| A | B | C | |
| Reagent | Dilution | Volume | |
| Buffer W | - | (n) x 220 µl | |
| SYTO83 | 1:1000 | (n) x 0.22 µl |
Wash slides 2x in 2X SSC
2m
Return slides to humidity chamber, cover with nuclear stain mixture and incubate for 00:15:00
15m
Wash slides 2x in 2X SSC, proceed to load slides onto the GeoMx DSP (see the GeoMx DSP Instrument User Manual) - https://nanostring.com/wp-content/uploads/2022/06/MAN-10152-01-GeoMx-DSP-Instrument-User-Manual.pdf
If needed, slides can be stored in 2X SSC for 1 week at 4 °C
Once GeoMx DSP aspirates have been collected, proceed to library preparation as per the GeoMx DSP NGS Readout User Manual - https://nanostring.com/wp-content/uploads/2022/06/MAN-10153-01-GeoMx-DSP-NGS-Readout-User-Manual.pdf
