Sep 11, 2025
Generation of human gastroid with fundic-antral patterning using pluripotent stem cells
- Xia Li1,
- Feng Lin2,
- Shiyu Sun3,
- Yue Shao1
- 1Institute of Biomechanics and Medical Engineering, Department of Engineering Mechanics, School of Aerospace Engineering, Tsinghua University, Beijing 100084, China;
- 2Wenzhou Institute, University of Chinese Academy of Sciences, Wenzhou, Zhejiang 325001, China;
- 3Department of Mechanical Engineering, University of Michigan, Ann Arbor, MI 48109, USA

Protocol Citation: Xia Li, Feng Lin, Shiyu Sun, Yue Shao 2025. Generation of human gastroid with fundic-antral patterning using pluripotent stem cells. protocols.io https://dx.doi.org/10.17504/protocols.io.q26g7n873lwz/v1
License: This is an open access protocol distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Protocol status: Working
We use this protocol and it's working
Created: July 26, 2025
Last Modified: September 11, 2025
Protocol Integer ID: 223331
Keywords: generation of human gastric organoid, pluripotent stem cells current human gastric organoid, antral axis in early stomach organogenesi, fundic gastric organoid, human gastric organoid, current human gastric organoid, antral gastric organoid, higher fidelity gastric development model, early stomach organogenesi, generation of human gastroid, spheroids from definitive endoderm, engineered tissue assembly, human pluripotent stem cell, human gastroid, like posterior foregut, tissue assembly, using pluripotent stem cell, spheroid, definitive endoderm, gastroid
Abstract
Current human gastric organoids are commonly generated using a stepwise process in which human pluripotent stem cells (hPSCs) are differentiated to epithelial-like posterior foregut (PFG) spheroids from definitive endoderm (DE) monolayer, before further development towards either fundic gastric organoids (FGO) or antral gastric organoids (AGO). However, the generation of human gastric organoids with asymmetric fundic-antral patterning remains challenging, which is possibly due to the lack of non-endodermal cell populations. Herein, we adopted a multi-lineage co-development strategy through either self-organization or engineered tissue assembly, which enabled the generation of a higher fidelity gastric development model, the gastroid, that could recapitulate the regional patterning along the fundic-antral axis in early stomach organogenesis.
Attachments
Materials
**mTeSR1, Stem Cell, 85850
Y-27632, TargetMol, T1870
RPMI1640, Gibco, 11875093
NEAA (Non-Essential Amino Acids), Gibco, 11140050
Activin A, peprotech, 120-14E
BMP4 (Bone Morphogenetic Protein 4), peprotech, 120-05ET
SB431542, selleck, S1067
LDN193189, selleck, S2618
dFBS (Defined Fetal Bovine Serum), Hyclone, SH30070
FGF4 (Fibroblast growth factor 4), peprotech, 100-31
CHIR99021, sigma, SML1046-5MG
NOGGIN, peprotech, 120-10C
RA (Retinoic acid), sigma, R2625
EGF (Epidermal Growth Factor), peprotech, AF-100-15
FGF10 (Fibroblast Growth Factor 10), peprotech, 100-26
N2, INVITROGEN, 17502048
B27, INVITROGEN, 17504044
L-glutamine, Gibco, 25030081
Penicillin–streptomycin, Gibco, 15070063
DMEM/F-12, Gibco, C11330500BT
Advanced DMEM/F-12, Gibco, 12634010
Neurobasal, Gibco, 21103049
HEPES, Gibco, 5630080
Dispase, Stem Cell, 7923
Accutase, Gibco, A1110501
Geltrex, Gibco, A1413302**
Troubleshooting
Before start
WORKFLOW:
Culture Medium:
*Gut medium: Advanced DMEM-F12+ 1× N2+ 1× B27 (without vitamin A) + 2 mM L-glutamine+
100 units per ml (1×) penicillin–streptomycin+ 15 mM HEPES.
*N2B27: basal neural differentiation medium (N2B27): Advance DMEM/F12: Neurobasal medium
(1:1; Gibco), 0.5× N2, 0.5× B27, 1% NEAA, 2 mM L-glutamine, and 0.1 mM β-mercaptoethanol.
Fabrication of U-bottom microwell array
3d 8h
A customized aluminum mold composed of arrays of hemispherical-top micropillars was machined by a computer numerically controlled ultra-high precision lathe. The diameter and total height of micropillars were 400 μm.
3d
A 2 Mass / % volume solution of agarose (w/v, distilled water) was poured into the aluminum mold, before cooling and solidifying at Room temperature , and then carefully peeled off from the mold.
6h
The U-bottom agarose microwell arrays were cut to fit the culture area of a 24-well plate before use.
1h
U-bottom agarose microwell arrays were sterilized with UV light in PBS for at least 00:30:00 and then blocked with0.5 Mass / % volume F127 for at least 00:30:00 before seeding cells.
1h
Generation of posterior foregut (PFG) spheroids from multi-lineage monolayer (multi-lineage PFG spheroids)
1w
Before day 0, human pluripotent stem cells (hPSCs) were maintained in a standard feeder-free system using mTeSR1 medium (Stem Cell) at 37 °C , 5% CO₂. And plates were coated with 1 % volume in PBS Geltrex (Gibco) at 37 °C for 01:00:00 . hPSCs were passaged every 3-4 days with 1U dispase (Stem Cell).
At day 0, to generate multi-lineage monolayer using an embryoid body (EB)-like approach, hPSCs were dissociated into single cells using Accutase (Gibco) at 37 °C for 00:10:00 and then seeded into 24-well plate containing U-bottom agarose microwell array at a density of 150,000 cells cm-2 in mTeSR1 medium containing 10 micromolar (µM) Y27632.
1d
At day 1, the EBs were cultured in a differentiation medium (RPMI 1640 medium + 1 % (v/v) NEAA + 100 ng/mL Activin A + 50 ng/mL BMP4) for 24 h.
1d
At day 2, EBs were collected from the microwell array, then reattached onto a 1 % (v/v) Geltrex-coated 24-well culture plate with a ratio of 1: 1, and cultured with RPMI 1640 medium + 1 % (v/v) NEAA + 0.2 % (v/v) dFBS + 100 ng/mL Activin A for 24h.
1d
At day 3, the cells were cultured with RPMI 1640 medium + 1 % (v/v) NEAA + 2 % (v/v) dFBS + 100 ng/mL Activin A for another 24 h.
1d
To further induce PFG differentiation, above multi-lineage monolayer was cultured in RPMI 1640 medium supplemented with 1 % (v/v) NEAA, 2 % (v/v) dFBS, 500 ng/mL FGF4, 200 ng/mL NOGGIN, and 2 micromolar (µM) CHIR99021 from day 4 to 7, with another 2 micromolar (µM) RA added from day 6 to 7 to obtain multi-lineage PFG spheroids, which spontaneously bud-off from the multi-lineage monolayer on day 7 and were collected for further culture.
3d
Human gastroid induction in 3D embedded culture
6w 1d 0h 45m
At day 7, multi-lineage PFG spheroids were collected and resuspended in Geltrex, and plated as 3D gel droplets with a volume of 20 μL in center of polydimethylsiloxane (PDMS) coated 24 well culture plate.
30m
After solidification of Geltrex for 00:15:00 at 37 °C , the 3D culture was initiated using basal gut medium, which contains: Advanced DMEM/F12, 1× N2 supplements (Invitrogen), 1× B27 supplements (without vitamin A, Invitrogen), 2 millimolar (mM) L-glutamine, 15 millimolar (mM) HEPES, and 100 U penicillin/streptomycin in combination with growth factors and / or small molecules as specified below.
15m
PFG spheroids were first cultured in basal gut medium supplemented with 100 ng/mL EGF, 200 ng/mL NOGGIN, 2 micromolar (µM) RA, and 50 ng/mL FGF10 from day 7 to day 10, and with 100 ng/mL EGF and 50 ng/mL FGF10 from day 10 to day 13, and then with 100 ng/mL EGF from day 13 to day 16.
1w 2d
For extended culture beyond day 16, the gastroids were manually collected and individually embedded in Geltrex, and further cultured in basal gut medium supplemented with 100 ng/mL EGF until day 34. Medium was replaced every 3-4 days.
4w 6d
Generation of epithelial PFG spheroids from definitive endoderm monolayer
1w
For differentiation of definitive endoderm (DE) monolayer, hPSCs at around 80% confluence were dissociated into single cells using Accutase at 37 °C for 00:10:00 and then re-plated onto a 1 % (v/v) Geltrex-coated 24-well plate at 150,000 cells cm-2 in mTeSR1 medium containing 10 micromolar (µM) Y27632.
1d
At day 1, cells were cultured in a differentiation medium (RPMI 1640 medium + 1 % (v/v) NEAA (non-essential amino acids) + 100 ng/mL Activin A + 50 ng/mL BMP4) for 24 h.
1d
At day 2, cells were cultured with RPMI 1640 medium + 1 % (v/v) NEAA + 0.2 % (v/v) defined fetal bovine serum (dFBS; Hyclone) + 100 ng/mL Activin A for 24 h.
1d
At day 3, the cells were cultured with RPMI 1640 medium + 1 % (v/v) NEAA + 2 % (v/v) dFBS + 100 ng/mL Activin A for another 24 h.
1d
To generate PFG spheroids, above DE monolayer was further differentiated in RPMI 1640 medium supplemented with 1 % (v/v) NEAA, 2 % (v/v) dFBS, 500 ng/mL FGF4, 200 ng/mL NOGGIN, and 2 micromolar (µM) CHIR99021 from day 4 to 7, with another 2 micromolar (µM) Retinoic acid (RA) added from day 6 to 7. Epithelial PFG spheroids spontaneously bud-off from the culture surface on day 7 and were collected for further differentiation.
3d
Generation of neural spheroids
6d
To generate neural spheroids, hPSCs were seeded into a 1 % (v/v) Geltrex-coated 24-well plate at 50,000 cells cm-2 in mTeSR1 medium containing 10 micromolar (µM) Y27632.
1d
At day 1, cells were cultured in basal neural differentiation medium (Advance DMEM/F12: Neurobasal medium (1:1; Gibco) + 0.5× N2 + 0.5× B27 + 1 % (v/v) NEAA + 2 millimolar (mM) L-glutamine + 0.1 millimolar (mM) β-mercaptoethanol) supplemented with 10 micromolar (µM) SB431542, 0.1 micromolar (µM) LDN193189 from day 1 to day 5.
4d
At day 6, singly-dissociated neural cells were seeded into 24-well plate containing U-bottom agarose microwell array at a density of 50,000 cells cm-2 in neural differentiation medium supplemented with 10 micromolar (µM) SB431542, 0.1 micromolar (µM) LDN193189 and 10 micromolar (µM) Y27632 to generate neural spheroids.
1d
Generation of gastric assembloids
1d 4h
Neural spheroids and epithelial posterior foregut (PFG) spheroids are separately generated using above-mentioned methods before being assembled.
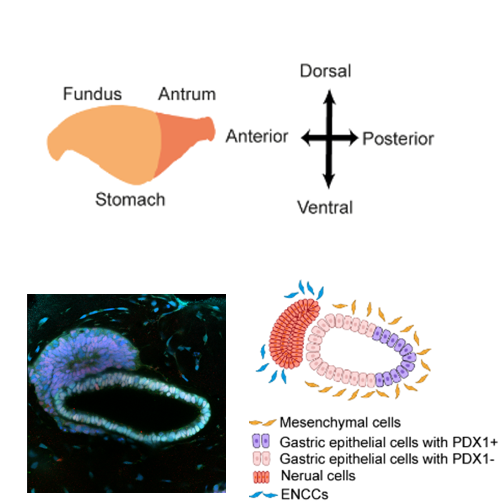
At day 7, epithelial PFG spheroids were collected and seeded into the U-bottom agarose microwell array that contains neural spheroids as described above to generate an asymmetric “gastric assembloid”, with neural population positioned in proximity to one side of the PFG spheroid.
2h
Then the assembloids were cultured in basal gut medium supplemented with 100 ng/mL EGF, 200 ng/mL NOGGIN, 2 micromolar (µM) RA, and 50 ng/mL FGF10.
1d
At day 8, the assembloids with 1:1 (PFG spheroid: neural spheroid) ratio were collected under a stereomicroscope and embedded in Geltrex as motioned perversely, further culture using the gastroid induction medium until day 16 (referred to human gastroid induction in 3D embedded culture section).
2h
Protocol references
1 Lin, F. et al. Mechanically enhanced biogenesis of gut spheroids with instability-driven morphomechanics. Nat. Commun. 14, 6016, doi:10.1038/s41467-023-41760-2 (2023).
2 Broda, T. R., McCracken, K. W. & Wells, J. M. Generation of human antral and fundic gastric organoids from pluripotent stem cells. Nat. Protoc. 14, 28-50, doi:10.1038/s41596-018-0080-z (2019).
3 Zheng, Y. et al. Dorsal-ventral patterned neural cyst from human pluripotent stem cells in a neurogenic niche. Sci. Adv. 5, eaax5933, doi:10.1126/sciadv.aax5933 (2019).

